Abstract
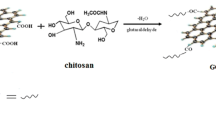
Composites containing magnetic chitosan chloride, graphene oxide and one of three different metal oxides (MnO2, Al2O3 and SiO2) were synthesised in the morphological form of beads and applied in removing Cr(VI) species in solution. The composites were successfully characterised using IR, XRD, TGA, DSC and SEM. Adsorption studies were carried out by varying pH, concentration, temperature and time. Maximum adsorptions of 78.2, 77.8 and 75.9 mg g−1 for each composite bead occurred at pH 2 and at 298 K. Adsorption followed the Langmuir isotherm, with a pseudo-second-order kinetic model. Thermodynamic studies proved that adsorption occurred spontaneously with the process being exothermic. The Cr(VI) species were desorbed from the beads using NaOH and the beads could be regenerated over six cycles.






Similar content being viewed by others
References
Aragay G, Pons J, Merkoçi A (2011) Recent trends in macro-, micro-, and nanomaterial-based tools and strategies for heavy-metal detection. Chem Rev 111:3433–3458. https://doi.org/10.1021/cr100383r
Azarmi F, Mironov E, Shakhova I, Safonov A (2018) Effect of Microstructural Characteristics on Thermal and Electrical Properties of Thermally Sprayed Ceramic Coatings. In: Proceedings of the 41st International Conference on Advanced Ceramics and Composites, vol 38. Wiley, pp 113–123. https://doi.org/10.1002/9781119474678.ch11
Azuaje J et al (2017) An efficient and recyclable 3D printed α-Al2O3 catalyst for the multicomponent assembly of bioactive heterocycles. Appl Catal A 530:203–210. https://doi.org/10.1016/j.apcata.2016.11.031
Barrera-Díaz CE, Lugo-Lugo V, Bilyeu B (2012) A review of chemical, electrochemical and biological methods for aqueous Cr(VI) reduction. J Hazard Mater 223–224:1–12. https://doi.org/10.1016/j.jhazmat.2012.04.054
Belgin Ergül NB, Mehmet SÖ (2014) The use of manganese oxide minerals for the removal arsenic and selenium anions from aqueous solutions. Energy Environ Eng 2:103–112. https://doi.org/10.13189/eee.2014.020501
Chen S, Zhu J, Wu X, Han Q, Wang X (2010) Graphene oxide − MnO2 nanocomposites for supercapacitors. ACS Nano 4:2822–2830. https://doi.org/10.1021/nn901311t
Costa M (1997) Toxicity and carcinogenicity of Cr(VI) in animal models and humans. Crit Rev Toxicol 27:431–442. https://doi.org/10.3109/10408449709078442
Devaraj S, Munichandraiah N (2008) Surfactant stabilized nanopetals morphology of α-MnO2 prepared by microemulsion method. J Solid State Electrochem 12:207–211
Donadel K, Felisberto MD, Fávere VT, Rigoni M, Batistela NJ, Laranjeira MC (2008) Synthesis and characterization of the iron oxide magnetic particles coated with chitosan biopolymer. Mater Sci Eng, C 28:509–514
Du Y, Wang L, Wang J, Zheng G, Wu J, Dai H (2015) Flower-, wire-, and sheet-like MnO2-deposited diatomites: highly efficient absorbents for the removal of Cr(VI). J Environ Sci 29:71–81. https://doi.org/10.1016/j.jes.2014.06.047
Elwakeel KZ (2009) Removal of reactive black 5 from aqueous solutions using magnetic chitosan resins. J Hazard Mater 167:383–392
Fan L, Luo C, Sun M, Li X, Lu F, Qiu H (2012a) Preparation of novel magnetic chitosan/graphene oxide composite as effective adsorbents toward methylene blue. Bioresour Technol 114:703–706. https://doi.org/10.1016/j.biortech.2012.02.067
Fan Y, Jiang W, Kawasaki A (2012b) Highly conductive few-layer graphene/Al2O3 nanocomposites with tunable charge carrier type. Adv Funct Mater 22:3882–3889. https://doi.org/10.1002/adfm.201200632
Ge J, Deng K, Cai W, Yu J, Liu X, Zhou J (2013) Effect of structure-directing agents on facile hydrothermal preparation of hierarchical γ-Al2O3 and their adsorption performance toward Cr(VI) and CO2. J Colloid Interface Sci 401:34–39. https://doi.org/10.1016/j.jcis.2013.03.028
Gheju M, Balcu I, Mosoarca G (2016) Removal of Cr(VI) from aqueous solutions by adsorption on MnO2. J Hazard Mater 310:270–277. https://doi.org/10.1016/j.jhazmat.2016.02.042
Glisoni RJ, Molina M, Calderón M, Moglioni AG, Sosnik A (2015) Chitosan-g-oligo (epsilon-caprolactone) polymeric micelles: microwave-assisted synthesis and physicochemical and cytocompatibility characterization. J Mater Chem B 3:4853–4864
Gupta VK, Gupta M, Sharma S (2001) Process development for the removal of lead and chromium from aqueous solutions using red mud—an aluminium industry waste. Water Res 35:1125–1134. https://doi.org/10.1016/S0043-1354(00)00389-4
Haeri SZ, Ramezanzadeh B, Asghari M (2017) A novel fabrication of a high performance SiO2-graphene oxide (GO) nanohybrids: characterization of thermal properties of epoxy nanocomposites filled with SiO2-GO nanohybrids. J Colloid Interface Sci 493:111–122. https://doi.org/10.1016/j.jcis.2017.01.016
Han G, Liu Y, Kan E, Tang J, Zhang L, Wang H, Tang W (2014) Sandwich-structured MnO2/polypyrrole/reduced graphene oxide hybrid composites for high-performance supercapacitors. RSC Adv 4:9898–9904
Hu X-j et al (2011) Adsorption of chromium (VI) by ethylenediamine-modified cross-linked magnetic chitosan resin: isotherms, kinetics and thermodynamics. J Hazard Mater 185:306–314
Huang G, Yang C, Zhang K, Shi J (2009) Adsorptive removal of copper ions from aqueous solution using cross-linked magnetic chitosan beads. Chin J Chem Eng 17:960–966. https://doi.org/10.1016/S1004-9541(08)60303-1
Hummers WS Jr, Offeman RE (1958) Preparation of graphitic oxide. J Am Chem Soc 80:1339–1339
Jastrzębska A et al (2016) Synthesis of the RGO/Al2O3 core–shell nanocomposite flakes and characterization of their unique electrostatic properties using zeta potential measurements. Appl Surf Sci 362:577–594
Jiang H, Yang L, Li C, Yan C, Lee PS, Ma J (2011) High-rate electrochemical capacitors from highly graphitic carbon-tipped manganese oxide/mesoporous carbon/manganese oxide hybrid nanowires. Energy Environ Sci 4:1813–1819. https://doi.org/10.1039/C1EE01032H
Jodeh S et al (2016) Using thiophene supported on SiO2 for removal Cr(VI) from water receptor. Appl J Envir Eng Sci 2:78–94
Karthikeyan T, Rajgopal S, Miranda LR (2005) Chromium(VI) adsorption from aqueous solution by Hevea Brasilinesis sawdust activated carbon. J Hazard Mater 124:192–199. https://doi.org/10.1016/j.jhazmat.2005.05.003
Kim BC, Park SW (2008) Fracture toughness of the nano-particle reinforced epoxy composite. Compos Struct 86:69–77
Kim M, Hwang Y, Kim J (2013) Graphene/MnO2-based composites reduced via different chemical agents for supercapacitors. J Power Sources 239:225–233
Kou L, Gao C (2011) Making silica nanoparticle-covered graphene oxide nanohybrids as general building blocks for large-area superhydrophilic coatings. Nanoscale 3:519–528
Kul AR, Koyuncu H (2010) Adsorption of Pb(II) ions from aqueous solution by native and activated bentonite: kinetic, equilibrium and thermodynamic study. J Hazard Mater 179:332–339. https://doi.org/10.1016/j.jhazmat.2010.03.009
Kumar S, Koh J (2014) Physiochemical and optical properties of chitosan based graphene oxide bionanocomposite. Int J Biol Macromol 70:559–564. https://doi.org/10.1016/j.ijbiomac.2014.07.019
Kurniawan TA, Chan GYS, Lo W-H, Babel S (2006) Physico–chemical treatment techniques for wastewater laden with heavy metals. Chem Eng J 118:83–98. https://doi.org/10.1016/j.cej.2006.01.015
Kyzas GZ, Kostoglou M, Lazaridis NK (2009) Copper and chromium(VI) removal by chitosan derivatives—equilibrium and kinetic studies. Chem Eng J 152:440–448. https://doi.org/10.1016/j.cej.2009.05.005
Lee H, Jeong Y, Min B, Lyoo W, Lee S (2009) Preparation and acid dye adsorption behavior of polyurethane/chitosan composite foams. Fibers Polym 10:636–642. https://doi.org/10.1007/s12221-010-0636-1
Li J, Zhang S, Chen C, Zhao G, Yang X, Li J, Wang X (2012) Removal of Cu(II) and fulvic acid by graphene oxide nanosheets decorated with Fe3O4 nanoparticles ACS. Appl Mater Interfaces 4:4991–5000. https://doi.org/10.1021/am301358b
Lin S, Kiang C (2003) Chromic acid recovery from waste acid solution by an ion exchange process: equilibrium and column ion exchange modeling. Chem Eng J 92:193–199
Liu L, Li C, Bao C, Jia Q, Xiao P, Liu X, Zhang Q (2012) Preparation and characterization of chitosan/graphene oxide composites for the adsorption of Au(III) and Pd(II). Talanta 93:350–357. https://doi.org/10.1016/j.talanta.2012.02.051
Liu M, Wen T, Wu X, Chen C, Hu J, Li J, Wang X (2013) Synthesis of porous Fe3O4 hollow microspheres/graphene oxide composite for Cr(VI) removal. Dalton Trans 42:14710–14717. https://doi.org/10.1039/c3dt50955a
Lu W, Luo Y, Chang G, Sun X (2011) Synthesis of functional SiO2-coated graphene oxide nanosheets decorated with Ag nanoparticles for H2O2 and glucose detection. Biosens Bioelectron 26:4791–4797. https://doi.org/10.1016/j.bios.2011.06.008
Luo X, Zeng J, Liu S, Zhang L (2015) An effective and recyclable adsorbent for the removal of heavy metal ions from aqueous system: magnetic chitosan/cellulose microspheres. Bioresour Technol 194:403–406
Ma W, Ya F-Q, Han M, Wang R (2007) Characteristics of equilibrium, kinetics studies for adsorption of fluoride on magnetic-chitosan particle. J Hazard Mater 143:296–302. https://doi.org/10.1016/j.jhazmat.2006.09.032
Maksin DD, Nastasović AB, Milutinović-Nikolić AD, Suručić LT, Sandić ZP, Hercigonja RV, Onjia AE (2012) Equilibrium and kinetics study on hexavalent chromium adsorption onto diethylene triamine grafted glycidyl methacrylate based copolymers. J Hazard Mater 209–210:99–110. https://doi.org/10.1016/j.jhazmat.2011.12.079
Martins AF, Bueno PV, Almeida EA, Rodrigues FH, Rubira AF, Muniz EC (2013) Characterization of N-trimethyl chitosan/alginate complexes and curcumin release. Int J Biol Macromol 57:174–184
Mckay G, Blair H, Findon A (1986) Sorption of metal ions by chitosan. In: Proc. Conf. Immobilisation of Ions by BiosorptionSoc. Chem. Ind., Lancaster University
Mourya VK, Inamdar NN (2008) Trimethyl chitosan and its applications in drug delivery. J Mater Sci 20:1057. https://doi.org/10.1007/s10856-008-3659-z
Ozaki H, Sharma K, Saktaywin W (2002) Performance of an ultra-low-pressure reverse osmosis membrane (ULPROM) for separating heavy metal: effects of interference parameters. Desalination 144:287–294. https://doi.org/10.1016/S0011-9164(02)00329-6
Palimi M, Rostami M, Mahdavian M, Ramezanzadeh B (2014) Application of EIS and salt spray tests for investigation of the anticorrosion properties of polyurethane-based nanocomposites containing Cr2O3 nanoparticles modified with 3-amino propyl trimethoxy silane. Prog Org Coat 77:1935–1945
Pugazhenthi G, Sachan S, Kishore N, Kumar A (2005) Separation of chromium (VI) using modified ultrafiltration charged carbon membrane and its mathematical modeling. J Membr Sci 254:229–239
Rozenberg BA, Tenne R (2008) Polymer-assisted fabrication of nanoparticles and nanocomposites. Prog Polym Sci 33:40–112. https://doi.org/10.1016/j.progpolymsci.2007.07.004
Sekine T, Nakatani K (2002) Intraparticle diffusion and adsorption isotherm for sorption in silica gel studied by single-microparticle injection and microabsorption methods. Langmuir 18:694–697. https://doi.org/10.1021/la0110500
Sharma YC, Srivastava V, Mukherjee AK (2010) Synthesis and application of nano-Al2O3 powder for the reclamation of hexavalent chromium from aqueous solutions. J Chem Eng Data 55:2390–2398. https://doi.org/10.1021/je900822j
Shaygan M, Davami K, Kheirabi N, Baek CK, Cuniberti G, Meyyappan M, Lee J-S (2014) Single-crystalline CdTe nanowire field effect transistors as nanowire-based photodetector. Phys Chem Chem Phys 16:22687–22693. https://doi.org/10.1039/C4CP03322A
Sieval AB, Thanou M, Kotze AF, Verhoef JC, Brussee J, Junginger HE (1998) Preparation and NMR characterization of highly substituted N-trimethyl chitosan chloride. Carbohydr Polym 36:157–165. https://doi.org/10.1016/S0144-8617(98)00009-5
Sun X, Yang L, Li Q, Zhao J, Li X, Wang X, Liu H (2014) Amino-functionalized magnetic cellulose nanocomposite as adsorbent for removal of Cr(VI): synthesis and adsorption studies. Chem Eng J 241:175–183. https://doi.org/10.1016/j.cej.2013.12.051
Tamura H, Katayama N, Furuichi R (1997) The Co2+ adsorption properties of Al2O3, Fe2O3, Fe3O4, TiO2, and MnO2 evaluated by modeling with the frumkin isotherm. J Colloid Interface Sci 195:192–202
Thinh NN et al (2013) Magnetic chitosan nanoparticles for removal of Cr(VI) from aqueous solution. Mater Sci Eng, C 33:1214–1218. https://doi.org/10.1016/j.msec.2012.12.013
Tiwari I, Singh M, Pandey CM, Sumana G (2015) Electrochemical genosensor based on graphene oxide modified iron oxide–chitosan hybrid nanocomposite for pathogen detection. Sens Actuators, B 206:276–283. https://doi.org/10.1016/j.snb.2014.09.056
Wan M-W, Kan C-C, Rogel BD, Dalida MLP (2010) Adsorption of copper (II) and lead (II) ions from aqueous solution on chitosan-coated sand. Carbohydr Polym 80:891–899. https://doi.org/10.1016/j.carbpol.2009.12.048
Weber W, Morris J (1962) Removal of biologically-resistant pollutants from waste waters by adsorption. Adv Water Pollut Res 2:231–266
Wu Y, Luo H, Wang H, Wang C, Zhang J, Zhang Z (2013) Adsorption of hexavalent chromium from aqueous solutions by graphene modified with cetyltrimethylammonium bromide. J Colloid Interface Sci 394:183–191. https://doi.org/10.1016/j.jcis.2012.11.049
Yang S et al (2015) Preparation of graphene oxide decorated Fe3O4@SiO2 nanocomposites with superior adsorption capacity and SERS detection for organic dyes. J Nanomater 16:337
Yu Z, Di H, Ma Y, Lv L, Pan Y, Zhang C, He Y (2015) Fabrication of graphene oxide–alumina hybrids to reinforce the anti-corrosion performance of composite epoxy coatings. Appl Surf Sci 351:986–996
Zhou F, Zhao X, Yuan C, Xu H (2007) Synthesis of γ-MnOOH nanorods and their isomorphous transformation into β-MnO2 and α-Mn2O3 nanorods. J Mater Sci 42:9978–9982
Zhu J et al (2012) One-pot synthesis of magnetic graphene nanocomposites decorated with core@double-shell nanoparticles for fast chromium removal. Environ Sci Technol 46:977–985. https://doi.org/10.1021/es2014133
Acknowledgements
This work was funded by the National Research Foundation (NRF), South Africa (Grant #95799), and the Eskom TESP Programme (Grant #P677). Microscopy images were taken by the Microscopy and Microanalysis Unit (MMU) at the University of Kwa Zulu-Natal.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Naicker, C., Nombona, N. & van Zyl, W.E. Fabrication of novel magnetic chitosan/graphene-oxide/metal oxide nanocomposite beads for Cr(VI) adsorption. Chem. Pap. 74, 529–541 (2020). https://doi.org/10.1007/s11696-019-00895-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11696-019-00895-7