Abstract
Introduction
Sarcopenic obesity (SO) is characterised by the confluence of muscle deterioration and high adiposity. When non-surgical interventions prove insufficient, bariatric surgery (BS) becomes the primary approach. This study aimed to address BS effects on SO outcomes 1 year post-surgery among middle-aged women, also considering physical exercise’s impact.
Methods
Prospective single-centre study of 140 patients who underwent Roux-en-Y gastric bypass or sleeve gastrectomy between November 2019 and December 2022. Participants were categorised into tertiles according to SO’s diagnosis and severity (group 1—patients with the most severe SO; group 2—intermediate; group 3—the least severe or without SO), calculated considering the consensus issued by ESPEN and EASO in 2022. Evaluations of clinical and biochemical parameters were conducted before and 12 months after BS, and the variation was used for comparative purposes. Body composition was assessed using bone density scans. Linear regression analysis accounted for both surgery type and baseline body mass index (BMI).
Results
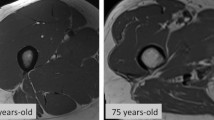
Before BS, SO prevalence in the overall sample was 89.3%, decreasing to 2.9% after BS. Group 1 had more body fat mass (56.9 vs 54.8 vs 50.7 kg, p < 0.001), total, trunk and leg fat at baseline and a significantly lower total skeletal muscle mass (47.2 vs 49.4 vs 51.8 kg, p < 0.001). One year post-BS, group 1 presented more weight loss (− 39.8 ± 11.4 kg, p = 0.031), BMI reduction (− 15.9 ± 4.6 kg/m2, p = 0.005) and lost more fat mass (− 32.6 vs − 30.5 vs − 27.9 kg, p = 0.005), but not total skeletal muscle mass (− 5.8 vs − 5.9 vs − 6.8 kg, p = 0.130). Remission rates for comorbidities were substantial among all groups, but more marked among patients within group 1 (type 2 diabetes mellitus 75%, hypertension 47.1% and dyslipidemia 52.8%). Engagement in physical exercise of any kind has increased post-BS (33.1% vs 79.1%).
Conclusion
Despite concerns about malabsorptive mechanisms potentially worsening muscle loss, patients with the most severe SO undergoing BS lost more fat mass while experiencing the smallest reduction in total skeletal muscle mass. Remission rates for comorbidities following BS were notable among all groups.
Graphical abstract

Similar content being viewed by others
References
Baumgartner R. Body composition in healthy aging. Ann N Y Acad Sci. 2006;994(1):437–48.
Hsu K, Liao C, Tsai M, et al. Effects of exercise and nutritional intervention on body composition, metabolic health, and physical performance in adults with sarcopenic obesity: a meta-analysis. Nutrients. 2019;11(9):2163.
Donini LM, Busetto L, Bischoff SC, et al. Definition and Diagnostic Criteria for Sarcopenic Obesity: ESPEN and EASO Consensus Statement. Obes Facts. 2022;15(3):321–35.
Cruz-Jentoft AJ, Bahat G, Bauer J, et al. Sarcopenia: revised European consensus on definition and diagnosis. Age Ageing. 2019;48(1):16–31.
Mastino D, Robert M, Betry C, et al. Bariatric surgery outcomes in sarcopenic obesity. Obes Surg. 2016;26(10):2355–62.
Wang M, Tan Y, Shi Y, et al. Diabetes and sarcopenic obesity: pathogenesis, diagnosis, and treatments. Front Endocrinol (Lausanne). 2020;25:11.
Lu C, Yang K, Chang H, et al. Sarcopenic obesity is closely associated with metabolic syndrome. Obes Res Clin Pract. 2013;7(4):301–7.
Kalinkovich A, Livsgits G. Sarcopenic obesity or obese sarcopenia: a cross talk between age-associated adipose tissue and skeletal muscle inflammation as a main mechanism of the pathogenesis. Ageing Res Rev. 2017;1(35):200–21.
Ciudin A, Simó-Servat A, Palmas F, et al. Sarcopenic obesity: a new challenge in the clinical practice. Endocrinol Diabetes Nutr. 2020;67(10):672–81.
Wei S, Nguyen TT, Zhang Y, et al. Sarcopenic obesity: epidemiology, pathophysiology, cardiovascular disease, mortality, and management. Front Endocrinol (Lausanne). 2023;14:1185221.
Fried M, Yumuk V, Oppert JM, et al. Interdisciplinary European guidelines on metabolic and bariatric surgery. Obes Facts. 2013;6(5):449–68.
Gallagher D, Heymsfield SB, Heo M, et al. Healthy percentage body fat ranges: an approach for developing guidelines based on body mass index. Am J Clin Nutr. 2000;72(3):694–701.
Poggiogalle E, Lubrano C, Sergi G, et al. Sarcopenic obesity and metabolic syndrome in adult Caucasian subjects. J Nutr Health Aging. 2016;20(9):958–63.
Petermann-Rocha F, Balntzi V, Gray SR, et al. Global prevalence of sarcopenia and severe sarcopenia: a systematic review and meta-analysis. J Cachexia Sarcopenia Muscle. 2022;13(1):86–99.
Kim T, Yang S, Yoo H, et al. Prevalence of sarcopenia and sarcopenic obesity in Korean adults: the Korean sarcopenic obesity study. Int J Obes. 2009;33:885–92.
Vieira FT, Godziuk K, Lamarca F, et al. Sarcopenic obesity diagnosis by different criteria mid-to long-term post-bariatric surgery. Clin Nutr. 2022;41(9):1932–41.
Mijnarends D, Meijers J, Halfens R, et al. Validity and reliability of tools to measure muscle mass, strength, and physical performance in community-dwelling older people: a systematic review. J Am Med Dir Assoc. 2013;14(3):170–8.
Prado C, Wells J, Smith S, et al. Sarcopenic obesity: a critical appraisal of the current evidence. Clin Nutr. 2012;31(5):583–601.
Sousa-Santos AR, Afonso C, Borges N, et al. Factors associated with sarcopenia and undernutrition in older adults. Nutr Diet. 2019;76(5):604–12.
Esposito L, Valeriani L, Anzolin F, et al. A valid screening tool of sarcopenic obesity in patients candidates to bariatric surgery. Clin Nutr ESPEN. 2023;1(54):506.
Sjöström L, Lindroos AK, Peltonen M, et al. Lifestyle, diabetes, and cardiovascular risk factors 10 years after bariatric surgery. N Engl J Med. 2004;351(26):2683–93.
Ciangura C, Bouillot J, Lloret-Linares C, et al. Dynamics of change in total and regional body composition after gastric bypass in obese patients. Obesity. 2010;18(4):760–5.
Sivakumar J, Chen Q, Sutherland TR, et al. Body composition differences between excess weight loss ≥ 50% and < 50% at 12 months following bariatric surgery. Obes Surg. 2022;32(8):2556–66.
Crispim Carvalho NN, Baccin Martins VJ, da Nóbrega VA, et al. Effects of preoperative sarcopenia-related parameters on cardiac autonomic function in women with obesity following bariatric surgery: a one-year prospective study. Nutrients. 2023;15(12):2656.
Crispim Carvalho NN, Martins VJB, Filho JM, et al. Effects of preoperative sarcopenia-related parameters on the musculoskeletal and metabolic outcomes after bariatric surgery: a one-year longitudinal study in females. Sci Rep. 2023;13(1):13373.
Lynch DH, Spangler HB, Franz JR, et al. Multimodal diagnostic approaches to advance precision medicine in sarcopenia and frailty. Nutrients. 2022;14(7):1384.
Coral RV, Bigolin AV, Machry MC, et al. Improvement in muscle strength and metabolic parameters despite muscle mass loss in the initial six months after bariatric surgery. Obes Surg. 2021;31(10):4485–91.
Brocca L, Toniolo L, Reggiani C, et al. FoxO-dependent atrogenes vary among catabolic conditions and play a key role in muscle atrophy induced by hindlimb suspension. J Physiol. 2017;595(4):1143–58.
Alizadeh PH. Exercise therapy for people with sarcopenic obesity: myokines and adipokines as effective actors. Front Endocrinol (Lausanne). 2022;17:13.
Piché ME, Tardif I, Auclair A, Poirier P. Effects of bariatric surgery on lipid-lipoprotein profile. Metabolism. 2021;1:115.
Brethauer S, Aminian A, Resenthal R, et al. Bariatric surgery improves the metabolic profile of morbidly obese patients with type 1 diabetes. Diab Care. 2014;37(3):51–2.
Tumova E, Sun W, Jones PH, et al. The impact of rapid weight loss on oxidative stress markers and the expression of the metabolic syndrome in obese individuals. J Obes. 2013; Available from: /pmc/articles/PMC3880717/
Wahlroos S, Phillips ML, Lewis MC, et al. Rapid significant weight loss and regional lipid deposition: implications for insulin sensitivity. Obes Res Clin Pract. 2007;1(1):7–16.
Harder H, Dinesen B, Astrup A. The effect of a rapid weight loss on lipid profile and glycemic control in obese type 2 diabetic patients. Int J Obes Relat Metab Disord. 2004;28(1):180–2. Available from: https://pubmed.ncbi.nlm.nih.gov/14610532/.
Pasdar Y, Darbandi M, Rezaeian S, et al. Association of obesity, sarcopenia, and sarcopenic obesity with hypertension in adults: a cross-sectional study from Ravansar, Iran During 2014–2017. Front Public Health. 2022;2:9.
Park SH, Park JH, Song PS, et al. Sarcopenic obesity as an independent risk factor of hypertension. J Am Soc Hypertens. 2013;7(6):420–5.
Coelho Júnior HJ, Aguiar SDS, Gonçalves IDO, et al. Sarcopenia is associated with high pulse pressure in older women. J Aging Res. 2015.
Ittermann T, Markus MRP, Bahls M, et al. Low serum TSH levels are associated with low values of fat-free mass and body cell mass in the elderly. Sci Rep. 2021;11(1):10547.
Nyrnes A, Jorde R, Sundsfjord J. Serum TSH is positively associated with BMI. Int J Obes. 2006;30(1):100–5.
Tiller D, Ittermann T, Greiser KH, et al. Association of serum thyrotropin with anthropometric markers of obesity in the general population. Thyroid. 2016;26(9):1205–14.
Svare A, Nilsen TIL, Bjøro T, et al. Serum TSH related to measures of body mass: longitudinal data from the HUNT Study, Norway. Clin Endocrinol (Oxf). 2011;74(6):769–75.
Petroni ML, Caletti MT, Dalle Grave R, et al. Prevention and treatment of sarcopenic obesity in women. Nutrients. 2019;11(6):1302.
Stuck AK, Tsai LT, Freystaetter G, et al. comparing prevalence of sarcopenia using twelve sarcopenia definitions in a large multinational European population of community-dwelling older adults. J Nutr Health Aging. 2023;27(3):205–12.
Author information
Authors and Affiliations
Consortia
Corresponding author
Ethics declarations
Ethics Approval
All procedures performed in this study involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Informed Consent
Written informed consent was obtained from all individual participants included in the study.
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Key Points
• Bariatric surgery emerges as both a risk factor and a management approach in sarcopenic obesity.
• Patients with severe sarcopenic obesity lost more fat mass and less muscle mass after bariatric surgery.
• Bariatric surgery reduces sarcopenic obesity’s prevalence and improves comorbidities.
Supplementary Information
ESM 1
(DOCX 21 kb)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Rodrigues, P.S., Mendonça, F.M., Neves, J.S. et al. Effects of Bariatric Surgery on Sarcopenic Obesity Outcomes: A One-Year Prospective Study in Middle-Aged Women. OBES SURG 34, 1674–1683 (2024). https://doi.org/10.1007/s11695-024-07164-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11695-024-07164-x