Abstract
Purpose
Obesity is a highly prevalent condition with severe clinical burden. Bariatric procedures are an important and expanding treatment option. This study compared short-(30-day composite adverse events) and long-term (intervention/operation, endoscopy, hospitalization, and mortality up to 5 years) safety outcomes associated with three bariatric surgical procedures.
Materials and Methods
This observational cohort study replicated an electronic health record study comparing short- and long-term problems associated with three bariatric surgical procedures between January 1, 2006, and September 30, 2015, within a Health Plan Research Network.
Results
Of 95,251 adults, 34,240 (36%) underwent adjustable gastric banding (AGB), 36,206 (38%) Roux-en-Y gastric bypass (RYGB), and 24,805 (26%) sleeve gastrectomy (SG). Median (interquartile range) years of follow-up was 3.3 (1.4–5.0) (AGB), 2.5 (1.0–4.6) (RYGB), and 1.1 (0.5–2.1) (SG). Overall mean (SD) age was 44.2 (11.4) years. The cohort was predominantly female (76%). Thirty-day composite adverse events occurred more frequently following RYGB (3.8%) than AGB (3.1%) and SG (2.8%). Operation/intervention was less likely in SG than in RYGB (adjusted hazard ratio (AHR), 0.87; 95%CI, 0.80–0.96; P=0.003), and more likely in AGB than in RYGB (AHR, 2.10; 95%CI, 2.00–2.21; P<0.001). Hospitalization was less likely after ABG and SG than after RYGB: AGB vs. RYGB, AHR=0.73; 95%CI, 0.71–0.76; P<0.001; SG vs. RYGB, AHR=0.79; 95%CI, 0.76–0.83; P<0.001. Mortality was most likely for RYGB (SG vs. RYGB: AHR, 0.76; 95%CI, 0.64–0.92; P=0.004; AGB vs. RYGB: AHR, 0.49; 95%CI, 0.43–0.56; P=0.001).
Conclusions
Interventions, operations, and hospitalizations were more often associated with AGB and RYGB than SG while RYGB had the lowest risk for revision.
Graphical abstract

Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
An estimated 93.3 million American adults were affected by obesity in 2015–2016; the estimated prevalence rate in adults was 39.8%, and 18.5% in youths, with a substantial portion severe enough (body mass index > 35 kg/m2 ) to merit consideration of bariatric surgery [1]. Because of its already high and increasing prevalence rate, obesity is deemed an epidemic by the Centers of Disease Control and Prevention [2]. Obesity is linked to several leading causes of preventable death including cardiovascular disease, type 2 diabetes, and some cancers, exacting a severe clinical burden [3].
As the prevalence of obesity increased, bariatric procedures became an important and rapidly expanding part of the treatment arsenal [4,5,6], with evidence shown to be superior to medical and lifestyle interventions for weight loss and glycemic control [7,8,9,10,11,12]. Roux-en-Y gastric bypass (RYGB) was predominant in the early 2000s; however, by the late 2000s, adjustable gastric banding (AGB) procedure had become more widely used. Both RYGB and AGB appear to have waned in popularity following the emergence of sleeve gastrectomy (SG), which is the most commonly used bariatric procedure currently[13, 14].
Information on the long-term comparative outcomes of these common bariatric procedures is accumulating [4, 6, 15, 16]. Pieces of evidence are wanted to address the existing variation in clinical utilization and insurance coverage of the surgical procedures [17, 18]. To help generate new knowledge and insights on their comparative long-term safety and effectiveness, the National Patient-Centered Clinical Research Network (PCORnet), using electronic health record (EHR) data, analyzed outcomes from three of the most commonly performed bariatric procedures: AGB, RYGB, and SG [19, 20], and have recently reported their results [21,22,23,24].
The purpose of the present study was to compare the short- and long-term safety outcomes across the three common bariatric surgery procedures, AGB, RYGB, and SG, using administrative claims data and the methodology of the PCORnet Bariatric Study (PBS). This study complements other real-world data to compare the safety of different bariatric procedures in the United States.
Methods
Study Design and Data Source
This observational cohort study simulated the research plan and employed the sample selection criteria described in the PBS [19,20,21,22]. This study first ran the PBS computable phenotype in the HealthCore-Anthem Research Network (HCARN) claims data stored in the PCORnet common data model. The HCARN data is a proprietary and longitudinal claims database containing medical and pharmacy information from enrollees within 14 US-based regional health plans. The computable phenotype retrospectively identified health plan enrollees who received bariatric surgery (see Supplementary Table 1 for codes) between January 1, 2006, and September 30, 2015. The initial bariatric procedure was defined as the index procedure, and the procedure date was defined as the index date. Adopting the PBS population definition, we evaluated adult patients (20–79 years old at the index date) and stratified them by index procedure type [19,20,21,22].
Outcomes of Interest
We used HCARN claims data in the PCORnet common data model to identify adverse events of interest delineated in the PBS [19,20,21,22]. Short-term safety included 30-day composite outcome, including venous thromboembolism, percutaneous or operative intervention, not being discharged within 30 days, or death. The long-term safety outcomes were evaluated up to 5 years after the index procedure. The primary long-term adverse event included subsequent operation or intervention encompassing any additional bariatric procedure and abdominal procedures. The secondary long-term adverse events consisted of subsequent endoscopy, revision (a component subcategory of operations), all-cause hospitalization, and all-cause death. Patients were censored once they were no longer enrolled with health plans or death or as of September 30, 2015, whichever was first. This study used a limited, deidentified dataset and was exempted from informed consent requirements by the New England Independent Review Board.
Statistical Analysis
Multivariable logistic regression analysis was used to estimate adjusted odds ratios (AORs) to compare the three types of bariatric surgeries for the 30-day composite outcome. Cox proportional hazard models were applied to estimate adjusted hazard ratios (AHRs) and cumulative probability of long-term outcomes for the three surgery types. All baseline demographic and clinical characteristics were adjusted in all models. For exploratory purposes, heterogeneity in treatment effects was tested across gender and age (<65, or ≥65 years), and a subgroup analysis was conducted on those aged 20 to 64 years old. All analyses were conducted with R Studio Pro, version 3.6.3 software.
Results
There were 95,251 adults that met the eligibility criteria for this study, as shown in Supplementary Figure 1. Overall, the mean (SD) age was 44.2 (11.4) years. The cohort was predominantly female (75.8%). Among eligible patients, 34,240 (35.9%) underwent AGB, 36,206 (38.0%) underwent RYGB, and 24,805 (26.0%) underwent SG. The RYGB group was slightly older (mean: 44.9 vs. 43.7 years for SG, and 43.9 years for AGB) (Table 1), with the highest rates of diabetes (42.4%) and hypertension (70.4%). A total of 1072 deaths occurred during the following and 42,574 patients had 5 years’ follow-up and 51,605 either disenrolled from the health plan or reached the end of the study period (09/30/2015) during the follow-up. The median (interquartile range) follow-up was 3.3 (1.4–5.0) years for AGB, 2.5 (1.0–4.6) years for RYGB, and 1.1 (0.5–2.1) years for SG.
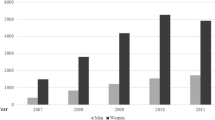
Figure 1 shows the temporal trend in bariatric surgeries performed from 2006 to 2015. We observed the peak proportions of patients with AGB in 2009 (63.6%) and a substantial decline to 9.1% in 2014. The proportion undergoing SG increased dramatically from 0.8% in 2006 to 62.6% in 2014. The trend for RYGB was relatively stable.
30-Day Composite Outcome
Within 30 days after the index procedure, 3.05% of AGB, 3.80% of RYGB, and 2.78% of SG had the composite outcome (Table 2). Larger proportions required intervention: 2.62% for AGB, 2.14% for RYGB, and 1.71% for SG. The AORs for the composite outcome were significantly lower for AGB relative to RYGB (AOR, 0.81; 95% CI, 0.72–0.92; P<0.001) and SG (AOR, 0.80; 95% CI, 0.73–0.87; P<0.001). Compared to the RYGB group, the SG group had similar probability (AOR, 0.98; 95% CI, 0.88–1.10; P=0.08).
Primary Long-Term Outcome
Operation or intervention was more likely following AGB compared to RYGB (AHR, 2.10; 95%CI, 2.00–2.21; P <0.001) but less likely for SG than RYGB (AHR, 0.87; 95%CI, 0.80–0.96; P=.003) (Table 3). Accordingly, the estimated cumulative probability (95% CI) of operation or intervention was higher for AGB, followed by RYGB and then SG. The probability for AGB was 7.0% (6.7–7.3%) at 1 year, 12.6% (12.1–3.1%) at 3 years, and 18.3% (17.6–19.0%) at 5 years (Table 4, Fig. 2a).
Heterogeneity of Treatment Effects for Primary Outcome and Subgroup Analysis
Heterogeneity of treatment effects was examined across gender (female, male) and age (<65, ≥65 years) (Table 5). There was no evidence of heterogeneity of treatment effects for SG vs. RYGB for gender and age. However, the increased risk of operation or intervention for AGB, compared to RYGB, was lower for males than females (male: AHR, 1.7; 95%CI, 1.6–1.9; and female: 2.2; 95% CI, 2.1–2.3; P < 0.001), and lower for age ≥65 than age <65 (age ≥65: 1.2; 95%CI, 1.0–1.6; and age <65: 2.2; 95% CI, 2.0–2.3; P < 0.001). Similarly, the increased risk for operation or intervention for AGB, compared to SG, was also lower for males than females and for age ≥65 than age <65 (Table 5). For the subgroup analysis, 91,854 (96.4% of the overall 95,251 patients) aged 20–64 years old, and the results for short- and long-term safety outcomes were similar to the main analysis (Supplementary Table 2).
Secondary Long-Term Outcomes
Endoscopy
Endoscopy for any reason (diagnostic or therapeutic) was less likely for SG vs. RYGB (AHR, 0.43; 95% CI, 0.38–0.48; P < 0.001) and also less likely for AGB vs. RYGB (AHR, 0.36; 95% CI, 0.33–0.39; P < .001) (Table 3). The corresponding cumulative rate of endoscopy (95% CI) was highest for RYGB: 3.7% (3.5–3.8%) at 1 year, 6.0% (5.7–6.3%) at 3 years, and 8.3% (7.9–8.7%) at 5 years (Table 4, Fig. 2c).
Revision
Revisional procedures appeared to be most common after AGB, followed by SG and then RYGB (AHR of AGB vs. RYGB, 11.3; 95% CI, 10.2–12.5; P <0.001; AHR of SG vs. RYGB, 2.9; 95% CI, 2.5–3.3; P <0.001) (Table 3). The highest estimated cumulative probability of revision (95% CI) were on AGB patients: 5.5% (5.2–5.8%) at 1 year, 9.3% (8.8–9.8%) at 3 years, and 14.9% (14.1–15.7%) at 5 years.
Hospitalization
Hospitalization was less likely after ABG and SG than after RYGB: AGB vs. RYGB, AHR=0.73; 95%CI, 0.71–0.76; P <0.001; SG vs. RYGB, AHR=0.79; 95%CI, 0.76–0.83; P <0.001 (Table 3). The estimated cumulative incidence rates of hospitalization (95% CI) for RYGB were 14.3% (14.0–14.7%) at 1 year, 30.0% (29.4–30.6%) at 3 years, and 42.3% (41.5–43.0%) at 5 years (Table 4, Fig. 2d).
Mortality
For time to all-cause mortality, the AHR was significantly lower after SG than RYGB: AHR, 0.76; 95%CI, 0.64–0.92; P =0.004. Compared to RYGB, AGB was associated with lower mortality risk (AHR, 0.49; 95%CI, 0.43–0.56; P =0.001) (Table 3). The estimated cumulative risk of all-cause mortality (95% CI) for RYGB was 0.34% (0.30–0.38%) at 1 year, 0.64% (0.57–0.71%) at 3 years, and 0.98% (0.88–1.09%) at 5 years (Table 4).
Discussion
Our study assessed short- and long-term bariatric surgery risks for adults in a real-world setting utilizing administrative claims data. Patients who underwent RYGB tended to have higher risks for major adverse events as compared to AGB or SG.
Consistent with reported patterns [13, 14, 20], we found a shift away from RYGB toward the use of AGB by the mid-to-late 2000s, while, since 2011, SG rose in preference and became predominant. In the recently published PBS utilizing the same cohort selection criteria, Arterburn et al. reported 5.5% of patients had AGB in their adult sample [21] compared to our finding of 36.0%. This is likely explained by our longer study period and ability to capture bariatric procedures in broader representation of healthcare settings (the EHR data from PBS over-represented academic medical settings). This might also explain the higher percutaneous or operative intervention rate of AGB observed within 30 days after the surgery.
Regarding long-term adverse events, our results show directional similarities with prior studies. Similar to the PBS [22] and other studies [25, 26] that compared the risk of RYGB versus SG, we found RYGB patients were more likely to require operation or intervention and hospitalization than SG patients in the long run. However, different from the PBS, the risks of AGB were smaller in our current study, and we observed a significant association between AGB and lower hospitalization and mortality when comparing to RYGB and SG. This difference may in part reflect a sampling difference in AGB patients. In our study, the AGB patients tended to be followed longer (median 3.3 vs. 1.1 years for SG) and the SG patients had a shorter time to hospitalization (median 0.9 vs. 2.4 years) at follow-up.
Although the estimated cumulative mortality risk was consistently less than 1% up to 5 years after three procedure types, we observed that RYGB patients had a significant higher death rate. Differently, the PBS showed similar mortality rates for SG and RYGB [22]. Of note, the RYGB and SG patients in our study had shorter follow-up time compared to the PBS cohort (median 2.5 vs. 3.4 years for RYGB; median 1.1 vs. 2.2 years for SG). This should be considered in interpreting the results. Additional follow-up time is necessary to examine the long-term mortality differences between bariatric procedures, particularly for SG.
Regarding the risk of revisional procedures, we found AGB patients had higher risk than RYGB and SG patients, consistent with the fact that ABG has been largely abandoned as a bariatric procedure owing to concerns about insufficient weight loss and the need for reoperation due to band failures and slippage. The estimated risk of revision was 14.9% at year 5 for AGB compared to 1.4% for RYGB and 4.0% for SG. This result is consistent with Ibrahim et al., in which 18.5% of Medicare beneficiaries who underwent AGB had reoperations over an average of a 4.5-year follow-up [27]. There was a significantly higher revision risk associated with SG (AHR of 2.88) when compared to RYGB. The corresponding AHR was 1.17 in the PBS but did not reach statistical significance.24 Similar to our result, Lewis et al. reported SG patients were more likely to undergo bariatric conversion or revision (AHR, 1.83; 95% CI, 1.19–2.80) [25]. A prior study showed more weight regain after SG at 5 years [21], which could contribute to the higher likelihood of revision associated with SG. Other explanations could be including the planned second-stage conversion (for early-stage SG) and the continuing or worsen of GERD associated with SG [28].
In the heterogeneity test, we found the harm of AGB over RYGB and SG for operation or intervention was greater with females and for those less than 65 years old. This finding suggests that females and younger population do better with RYGB or SG rather than AGB. Our study confirms the heterogeneity findings regarding gender and age in the PBS [22].
When interpreting the results, the different follow-up time of each procedure group is worthy of note. AGB patients had the longest follow-up with a median of 3.3 years, and SG patients had the least with a median of 1.1 years. The difference was tied to the time trends in bariatric procedure evolution. Given the rise of SG towards the end of the study period, our analysis did not have a large number of SG patients followed for 5 years, which could impair the likelihood to distinguish the difference for rare and late-onset outcomes such as revision and mortality. Studies with more longer-term follow-up will be important to examine our findings for SG populations.
Prior studies have presented the comparative effectiveness and safety of bariatric procedures on obese patients and the subset with diabetes [21,22,23,24,25, 29, 30]. Our study adds safety information for bariatric procedures using a large national health plan data. Our results clarify that AGB is associated with high risk of subsequent operation/intervention and revision and becomes less common in practice. Regarding SG and RYGB, our results show that RYGB has a higher risk of subsequent operation or intervention, but SG has a higher revision rate possibly due to more weight regain and reflux. The findings of this study will undoubtedly contribute to the larger understanding needed in making clinical decisions, and also help payers to decide on the best investments in this area.
Limitations
This observational study relied on secondary data repurposed for research from their original transactional functions. The HCARN data does not provide information on race and ethnicity, nor do they include risk factors such as family history, diet, and exercise regimen—all of which could influence outcomes substantively. The data does not capture the member’s healthcare utilization if they disenroll from the health plans, which leads to loss of follow-up. More directly, anthropometric measures, such as body mass index, were not available in claims data, and comorbidity resolution data was not examined during the follow-up. Additionally, as aforementioned, the SG group had a shorter follow-up time. As a result, these findings must be interpreted with caution. While the study population was reflective of working-age adult patients in commercial health plans, the results may not be generalizable to patients enrolled in different types of health plans, those outside the United States, or those without health insurance.
Conclusion
This study compared the risks of the three commonly performed bariatric procedures in a large and broadly representative sample. This study showed that AGB patients experienced the most risk of adverse events in general, and RYGB had a higher risk of operation and intervention but sustainable less need for revision comparing to SG after the initial bariatric procedure. Our results extend the PBS results by adding claims data representative of academic and non-academic medical centers, which could provide additional perspectives and guidance, and a better understanding of the range of longer-term outcomes from bariatric procedures.
References
Pories WJ. Bariatric surgery: risks and rewards. J Clin Endocrinol Metab. 2008;93(11 Suppl 1):S89–96.
Bolen SD, Chang HY, Weiner JP, et al. Clinical outcomes after bariatric surgery: a five-year matched cohort analysis in seven US states. Obes Surg. 2012;22(5):749–63.
U.S. Department of Health & Human Services. Managing overweight and obesity in adults: systematic evidence review from the obesity expert panel. Accessed at https://www.nhlbi.nih.gov/health-topics/managing-overweight-obesity-in-adultson08-22-18.
Nguyen NT, Masoomi H, Magno CP, et al. Trends in use of bariatric surgery, 2003-2008. J Am Coll Surg. 2011;213(2):261–6.
Sturm R. Increases in clinically severe obesity in the United States, 1986-2000. Arch Intern Med. 2003;163(18):2146–8.
Courcoulas AP, Yanovski SZ, Bonds D, et al. Long-term outcomes of bariatric surgery: a National Institutes of Health symposium. JAMA Surg. 2014;149(12):1323–9.
Adams TD, Davidson LE, Litwin SE, et al. Health benefits of gastric bypass surgery after 6 years. JAMA. 2012;308(11):1122–31.
Ikramuddin S, Korner J, Lee WJ, et al. Roux-en-Y gastric bypass vs intensive medical management for the control of type 2 diabetes, hypertension, and hyperlipidemia: the Diabetes Surgery Study randomized clinical trial. JAMA. 2013;309(21):2240–9.
Mingrone G, Panunzi S, De Gaetano A, et al. Bariatric surgery versus conventional medical therapy for type 2 diabetes. N Engl J Med. 2012;366(17):1577–85.
Schauer PR, Kashyap SR, Wolski K, et al. Bariatric surgery versus intensive medical therapy in obese patients with diabetes. N Engl J Med. 2012;366(17):1567–76.
Arterburn DE, Courcoulas AP. Bariatric surgery for obesity and metabolic conditions in adults. BMJ. 2014;349:g3961.
Reames BN, Finks JF, Bacal D, et al. Changes in bariatric surgery procedure use in Michigan, 2006-2013. JAMA. 2014;312(9):959–61.
Sarwer DB, Ritter S, Wadden TA, et al. Attitudes about the safety and efficacy of bariatric surgery among patients with type 2 diabetes and a body mass index of 30-40 kg/m2. Surg Obes Relat Dis. 2013;9(5):630–5.
Sikorski C, Luppa M, Dame K, et al. Attitudes towards bariatric surgery in the general public. Obes Surg. 2013;23(3):338–45.
Ferrante JM, Piasecki AK, Ohman-Strickland PA, et al. Family physicians' practices and attitudes regarding care of extremely obese patients. Obesity (Silver Spring). 2009;17(9):1710–6.
Finkelstein EA, Allaire BT, Burgess SM, et al. Financial implications of coverage for laparoscopic adjustable gastric banding. Surg Obes Relat Dis. 2011;7(3):295–303.
Karlsson J, Taft C, Ryden A, et al. Ten-year trends in health-related quality of life after surgical and conventional treatment for severe obesity: the SOS intervention study. Int J Obes (Lond). 2007;31(8):1248–61.
Kohn GP, Galanko JA, Overby DW, et al. Recent trends in bariatric surgery case volume in the United States. Surgery. 2009;146(2):375–80.
Comparing the Benefits and Harms of Three Types of Weight Loss Surgery -- The PCORnet Bariatric Study. Patient-Centered Outcomes Research Institute. https://www.pcori.org/research-results/2015/comparing-benefits-and-harms-three-types-weight-loss-surgery-pcornet-bariatric (accessed 08-17-2018).
Toh S, Rasmussen-Torvik LJ, Harmata EE, et al. The National Patient-Centered Clinical Research Network (PCORnet) Bariatric Study Cohort: rationale, methods, and baseline characteristics. JMIR Res Protoc. 2017;6(12):e222.
Arterburn D, Wellman R, Emiliano A, et al. Comparative effectiveness and safety of bariatric procedures for weight loss: a PCORnet cohort study. Annals of internal medicine. 2018;169(11):741–50.
Courcoulas A, Coley RY, Clark JM, et al. Interventions and operations 5 years after bariatric surgery in a cohort from the US National Patient-Centered Clinical Research Network Bariatric Study. JAMA Surg. 2020;155:194–204.
Arterburn DE, Johnson E, Coleman KJ, Herrinton LJ, Courcoulas AP, Fisher D, Li RA, Theis MK, Liu L, Fraser JR, Haneuse S. Weight outcomes of sleeve gastrectomy and gastric bypass compared to nonsurgical treatment. Ann Surg. 2020. https://doi.org/10.1097/SLA.0000000000003826.
McTigue KM, Wellman R, Nauman E, et al. Comparing the 5-year diabetes outcomes of sleeve gastrectomy and gastric bypass: the National Patient-Centered Clinical Research Network (PCORNet) Bariatric Study. JAMA Surg. 2020;155:e200087.
Lewis KH, Arterburn DE, Callaway K, et al. Risk of operative and nonoperative interventions up to 4 years after Roux-en-Y gastric bypass vs vertical sleeve gastrectomy in a nationwide US commercial insurance claims database. JAMA Netw Open. 2019;2(12):e1917603.
Li RA, Liu L, Arterburn D, et al. Five-year longitudinal cohort study of reinterventions after sleeve gastrectomy and Roux-en-Y Gastric Bypass. Ann Surg. 2019;273:758–65.
Ibrahim AM, Thumma JR, Dimick JB. Reoperation and medicare expenditures after laparoscopic gastric band surgery. JAMA Surg. 2017;152(9):835–42.
Arterburn D, Gupta A. Comparing the outcomes of sleeve gastrectomy and Roux-en-Y gastric bypass for severe obesity. JAMA. 2018;319(3):235–7.
Coleman KJ, Schlundt DG, Bonnet KR, et al. Understanding the bariatric patient perspective in the National Patient-Centered Clinical Research Network (PCORnet) bariatric study. Obes Surg. 2020;30(5):1837–47.
Coleman KJ, Schlundt DG, Bonnet KR, et al. Correction to: understanding the bariatric patient perspective in the National Patient-Centered Clinical Research Network (PCORnet) bariatric study. Obes Surg. 2020;30(5):1848.
Funding
This study was funded by the Patient-Centered Outcomes Research Institute, Contract for Advancement of PCORnet Infrastructure: Health Plan Data Project, HealthCore Inc., HealthCore-Anthem Research Network (HCARN), under award number of HP-1510-32545.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical Approval Statement
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. This study was conducted in full compliance with relevant provisions of the Health Insurance Portability and Accountability Act. Researchers had access to only a limited dataset that contained no patient identifiers and no patients were contacted directly.
Informed Consent Statement
This is a retrospective observational study, and for this type of study, formal consent is not required.
Conflict of Interest
Dr. Arterburn and Dr. McTigue both participated in the bariatric study funded by PCORI: an initiative funded by the Patient-Centered Outcomes Research Institute (grant OBS-1505-30683). For the remaining authors, no conflicts of interest were declared.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Key Points
1. Gastric banding is associated with high risk for operation and revision and has become less common in practice.
2. Operations or interventions, hospitalization, and endoscopy are more commonly associated with gastric bypass than sleeve gastrectomy.
3. The risk of revision is less for gastric bypass than for sleeve gastrectomy.
4. The results contribute to the larger understanding needed in making clinical and policy decisions on bariatric procedures.
Supplementary Information
ESM 1
(DOCX 79 kb)
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Ma, Q., Shambhu, S., Arterburn, D.E. et al. Interventions and Operations after Bariatric Surgery in a Health Plan Research Network Cohort from the PCORnet, the National Patient-Centered Clinical Research Network. OBES SURG 31, 3531–3540 (2021). https://doi.org/10.1007/s11695-021-05417-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11695-021-05417-7