Abstract
Introduction
About 20% of patients operated with Roux-en-Y gastric bypass (RYGBP) experience poor long-term weight result. This study compared levels of leptin and gut hormones in long-term weight responders with non-responders after RYGBP. In a subgroup analysis, hormone levels were assessed in T2DM (type 2 diabetes mellitus) and normoglycemic participants.
Methods
Insulin, glucose, leptin, acyl-ghrelin, total PYY, active GLP-1, and GIP were measured during an oral glucose tolerance test (OGTT) in post-RYGBP subjects: 22 non-responders (BMI 40.6 ± 6.0 kg/m2 after an excess BMI loss [EBMIL] of 26.0 ± 15.9%) and 18 responders (BMI 29.5 ± 3.5 kg/m2 after an EBMIL of 74.9 ± 18.2%). Subjects were matched for preoperative age, BMI, and years of follow-up. Measures of glucose homeostasis were calculated, and body composition was measured.
Results
Fat mass–adjusted fasting leptin correlated negatively with %EBMIL (r = − 0.57, p < 0.01). Non-responders presented higher levels of leptin during the OGTT. Leptin decreased and ghrelin returned to baseline levels earlier in non-responders. Despite having higher insulin resistance than responders, non-responders demonstrated similar OGTT responses of GLP-1, GIP, and PYY. T2DM participants demonstrated lower GLP-1 levels than normoglycemic participants of similar weight.
Conclusion
Fasting leptin is associated with weight result after RYGBP, and hormonal responses to a glucose oral load might work towards promoting obesity in long-term non-responders after RYGBP. Poor long-term weight result and glycemic status after RYGBP are each associated with differences in peptide hormone levels.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Bariatric surgery is considered to be a safe and reliable method to achieve substantial weight loss and relief of obesity-related comorbidities, with superior outcomes compared with non-surgical alternatives [1, 2]. Roux-en-Y gastric bypass (RYGBP) [3] results in durable weight loss for most patients [4], with marked hormonal changes occurring promptly after surgery, even before tangible weight loss ensues [5]. However, some degree of postsurgical weight regain is common, as noted in one-half of patients 2 years after surgery [6]. After 5 years, about 20% of patients will have lost less than half of their preoperative overweight [7], a hallmark of failed bariatric surgery [8].
To study mechanisms involved in weight loss and weight maintenance, research has been aimed at investigating various peptide hormones that regulate appetite and glucose homeostasis. Leptin is an adipocyte-derived hormone involved in satiety sensing and energy balance regulation that mainly changes in tandem with fat mass [9]. Leptin decreases after diet-induced weight loss and remains low during weight maintenance [10]. In obese subjects, decreased sensitivity to circulating leptin is suspected [11]. Amongst endocrine signaling hormones in the upper gastrointestinal tract, the orexigenic hormone ghrelin [12] and gastric inhibitory peptide (GIP) [13] are higher in obese individuals than in normal-weight individuals. After increasing during diet-induced weight loss, ghrelin and GIP both return to baseline levels during weight maintenance [14, 15]. These changes speak in favor of a transient regulatory role of ghrelin and GIP in diet-induced short-term weight loss.
Glucagon-like peptide-1 (GLP-1) is an insulinogenic and anorexigenic hormone from the lower gastrointestinal tract. Fasting GLP-1 levels increase after diet-induced weight loss and after 1 year of weight maintenance [15], and postprandial GLP-1 levels are lower in obese subjects than in lean subjects [16]. Peptide YY (PYY) reduces the postprandial insulin response and postprandial PYY was found to increase after diet-induced weight loss and remained higher than baseline 1 year after maintenance of reduced body weight [15]. GLP-1 and PYY are expected to dampen hunger signaling and promote weight loss, and thus, changes in these hormones after diet-induced weight loss are believed to contribute to long-term weight maintenance.
To identify hormonal differences associated with disparate long-term outcomes of bariatric surgery, the primary aim of this study was to compare circulating leptin and peptide hormones during an oral glucose tolerance test (OGTT) in subjects with and without satisfactory long-term weight loss after RYGBP. In a subgroup analysis, hormone levels in post-RYGBP subjects with type 2 diabetes mellitus (T2DM) were compared with weight-similar normoglycemic subjects.
Material and Methods
Participant Selection
Female subjects operated with RYGBP were screened for inclusion amongst 474 subjects answering our follow-up questionnaire regarding body weight 5 years after surgery. Selection criterion for non-responders was less than 50% excess BMI loss (%EBMIL), while responders were recruited from the remaining patients. Excess BMI was defined as BMI over 25 kg/m2, and 22.8% of answering subjects reported an EBMIL of < 50%. Non-responders (n = 22) and responders (n = 18) were matched at inclusion for preoperative age, weight, and height, as well as number of years between surgery and examination (Table 1). After a median of 11 years after surgery, non-responders had a BMI of 40.6 ± 6.0 kg/m2 and responders had a BMI of 29.5 ± 3.5 kg/m2, corresponding to an EBMIL of 26.0% ± 15.9% and 74.9% ± 18.2%, means ± SD, respectively. Because the majority of patients having bariatric surgery are female, male participants were excluded from the present study for the sake of uniformity.
T2DM Subgroup
T2DM subjects were identified according to the diagnostic criteria of the OGTT: fasting blood glucose values ≥ 7.0 mmol/L and/or blood glucose ≥ 11.0 mmol/L at the 120-min checkpoint of the test. Normoglycemic subjects were identified as having fasting blood glucose of < 6.1 mmol/L. To better define the groups, four participants with impaired glucose tolerance were excluded from the subgroup analysis. Thus, seven subjects with T2DM (BMI 34.7 ± 5.3 following EBMIL 46.2 ± 26.3%, 11.7 ± 4.5 years after surgery) were compared with 29 normoglycemic subjects (BMI 34.3 ± 6.5 following EBMIL 52.9 ± 27.9%, 11.7 ± 3.8 years after surgery, means ± SD respectively) regarding glucose homeostasis and hormone levels. There were 2 responders and 5 non-responders in the diabetic group, and 15 responders and 14 non-responders in the normoglycemic group. T2DM subjects did not significantly differ from normoglycemic participants in preoperative age, BMI, %EBMIL, or years of follow-up.
Surgical Technique
The main part of the stomach and duodenum were excluded from the passage of ingested nutrients by the construction of a proximal gastric pouch. The jejunum was divided 30 cm after the ligament of Treitz, and the distal end was anastomosed to the gastric pouch. Biliary continuity was restored by anastomosing the proximal end to the jejunum, 70 cm distal to the gastroenteroanastomosis. All participants were operated upon using the same technique. All participants followed the same postoperative course and follow-up.
Oral Glucose Tolerance Test
Participants were asked to fast overnight and to take no medications on the day of the examination. On arriving to the laboratory at 08:30 AM, they were informed about the OGTT details. An intravenous line was inserted into an antecubital vein, and anthropometric data (length and weight) were collected. Fasting blood samples were collected before ingestion of 75 g of glucose dissolved in 250 mL water to start the OGTT. Additional blood samples were drawn at 30, 60, 90, and 120 min after the oral glucose load. Samples were later assayed for leptin and acyl-ghrelin (hereinafter ghrelin), the incretins GIP and active GLP-1, as well as total PYY, insulin, and glucose.
Blood Sample Handling
A protease inhibitor cocktail was prepared, as described in earlier studies [17,18,19], consisting of 5.5 μL 10 mM KR-62436 [20] (DPP4 inhibitor) in DMSO and a SIGMAFAST® protease inhibitor tablet [21] (both produced by Sigma-Aldrich Corp., St. Louis, MO, USA, cat no. K4264 and S8830) dissolved in 2100 μL of distilled water (50× stock). Blood samples were immediately put on ice, and 160 μL of the 50× protease inhibitor cocktail was added. Tubes were vortexed for 10 s and centrifuged at 4 °C, 10 min, and 2500 RCF. The resulting supernatant (plasma) was immediately frozen at − 25 °C until assayed. Blood was also drawn into 6-mL tubes in parallel for HbA1c, insulin, and glucose analyses. These were sent at room temperature for analysis at the hospital clinical chemistry laboratory immediately after the examination was completed.
Multiplex ELISA by Electrochemiluminescence
Plasma concentrations of ghrelin, GIP, active GLP-1, leptin, and total PYY were assayed by multiplex ELISA using electrochemiluminescence detection. Plasma samples were thawed and vortexed. Samples were then analyzed in duplicate on 96-well multispot plates (Meso Scale Diagnostics, Rockville, MD, USA) coated with capture antibodies against ghrelin, GIP, active GLP-1, leptin, and total PYY according to manufacturer’s instructions. The Meso Scale Diagnostics QuickPlex SQ120 imager was used to read the plates. Resulting duplicate sample values were used to calculate percent coefficient of variation (CV%) values, which were as follows (intra/inter-assay): ghrelin 19.8/3.5; GIP 3.6/6.9; active GLP-1 8.8/27.5; leptin 4.4/9.1; total PYY 5.1/4.0. Intra-assay CV% was possible using a quality control plasma sample on all plates. Lower limits of detection calculated from 5 standard curves were as follows (ng/L): ghrelin 6.4; GIP 4.0; active GLP-1 0.5; leptin 65; total PYY 12.5. Reference fasting values for these assays obtained from 41 healthy humans were as follows (mean ± SD, ng/L): ghrelin 118 ± 93; GIP 204 ± 197; active GLP-1 2.3 ± 2.5; leptin 4202 ± 8520; total PYY 38.5 ± 15.8.
Measures of Glucose Homeostasis
Whole-body insulin sensitivity was estimated using the Matsuda Index [22]. β cell function was estimated by the insulinogenic Index, using the formula: Insulinogenic Index = (30 min insulin − fasting insulin)/((30 min glucose − fasting glucose) × 18) [23]. Glucose and insulin responses were calculated as the incremental area under the curve (iAUC) by use of the trapezoid rule on values obtained by calculating the difference between measures in each control point and baseline (fasting values).
Body Composition
As leptin is known to be produced in the adipose tissue, it was considered necessary to adjust leptin values for fat mass. For this purpose, body composition was measured in all participants at the examination using a combination of bioelectrical impedance analysis (InBody S20, Biospace, Seoul, Korea) and air displacement plethysmography (BodPod, Life Measurements Inc., Concord, CA, USA).
Statistics
Data were investigated for normality using the Shapiro-Wilk test. Parametric variables were described as mean ± standard deviation and non-parametric variables as median with interquartile range. Correlations between %EBMIL, and fasting leptin and ghrelin (non-normally distributed) were done using Spearman’s product-moment correlation. The ratio between ghrelin and fat mass–adjusted leptin was calculated by dividing the former with the latter. Hormone data, ghrelin/leptin ratio, glucose, and insulin (non-normally distributed) during the OGTT were investigated for differences between groups using the Wilcoxon test and Holm’s method for correction for multiple comparisons. Differences in hormone levels within groups between baseline and different time points were examined with the Friedman test followed by Dunn’s test to correct for multiple comparisons.
For all analyses, p values < 0.05 were considered significant. Statistical analyses were conducted using R software (R version 3.2.5, The R Foundation for Statistical Computing, Vienna, Austria) and GraphPad Prism 8.1 software (GraphPad Software Inc., San Diego, CA, USA).
Results
Leptin
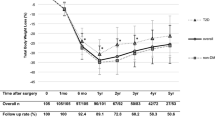
Postsurgical weight loss, measured as %EBMIL, correlated negatively with fat mass–adjusted fasting plasma leptin (r = − 0.57, p < 0.001, Fig. 1). This finding was consistent in the normoglycemic subgroup (r = − 0.61, p < 0.001). Non-responders showed higher levels of fat mass–adjusted leptin than responders during fasting and throughout the 60-, 90-, and 120-min time points. During the OGTT, non-responders showed lower-than-fasting leptin levels at the 90- and 120-min time points. Responders showed no differences in leptin from fasting levels throughout the OGTT (Fig. 2).
Ghrelin
There was a tendency for a positive correlation between %EBMIL and fasting plasma ghrelin (r = 0.30, p = 0.06, Fig. 1), but this correlation was not found in the normoglycemic subgroup. Ghrelin levels did not differ between responders and non-responders during fasting or throughout the OGTT. Ghrelin returned to baseline levels in non-responders at the 120-min time point, whereas responders showed lower-than-baseline ghrelin levels throughout the OGTT (Fig. 2).
Ghrelin/Leptin Ratio
Responders showed a higher ghrelin to leptin ratio during fasting and at the 60- and 90-min time points compared with non-responders (Fig. 2).
GIP, GLP-1, and PYY
Fasting levels of GIP, GLP-1, and PYY did not differ between non-responders and responders. For all three hormones, both groups demonstrated increased plasma levels in response to the OGTT at all times. There was a tenfold increase in GLP-1 from baseline to the 30-min time point in both groups (Fig. 2).
Glucose Homeostasis
Non-responders presented with a higher fasting blood glucose and lower insulin sensitivity than responders. No differences were observed between responders and non-responders regarding fasting serum insulin, β cell function, and glucose or insulin response to the OGTT (Table 2, Fig. 3).
T2DM Subgroup Analysis
As expected, the T2DM group presented higher levels of fasting glucose, higher glucose response, and lower insulin response to the OGTT than the normoglycemic group. T2DM subjects also had lower insulin sensitivity and β cell function compared with normoglycemic subjects. T2DM subjects showed lower GLP-1 levels compared at the 90- and 120-min time points compared with normoglycemic participants, but comparable levels of leptin, ghrelin, PYY, and GIP (Fig. 4). There were no differences between groups regarding the ratio between ghrelin and leptin throughout the OGTT.
Discussion
Fasting plasma levels of leptin and ghrelin correlated with weight result more than 5 years after surgery, and hormonal response to glucose differed between weight responders and non-responders during an OGTT. Furthermore, differences in gut hormones known to change after RYGBP were associated with glucose homeostasis in post-RYGBP participants.
There is no single best method with which to evaluate postoperative weight result. Changes in excess BMI, total body weight, and degree of weight regain have all been used for this purpose [24]. The use of %EBMIL, although reported to yield greater results in lighter subjects compared with heavier ones [25], is considered safe in this study as the presented groups were matched for preoperative BMI. Beyond changes in excess BMI, a definition of successful weight loss should also include remission of comorbidities and improvement of patients’ quality of life.
Circulating leptin is known to decrease by 50% during the first postoperative week after RYGBP surgery and to decrease further throughout the first postoperative year during weight loss [26]. Long-term weight result correlated negatively with fat mass–adjusted fasting leptin levels, with non-responders showing higher levels of leptin than responders during the OGTT as well as lower-than-baseline levels at the end of the OGTT. This is in line with earlier findings where leptin levels in morbidly obese subjects (BMI 45 kg/m2) were higher than in lean subjects [27]. The present data suggests that short-term RYGBP-induced leptin decreases are not sustained in subjects with poor weight result many years after surgery, with leptin levels and response to OGTT resembling those of non-operated obese subjects.
Although a tendency for positive correlation was found between fasting ghrelin and %EBMIL, no differences in fasting or glucose-stimulated ghrelin were found between weight responders and non-responders, which agrees with earlier studies where no change in fasting ghrelin was observed in weight-reduced patients up to 2 years after RYGBP surgery [28, 29]. Non-responders rather showed fasting ghrelin levels similar to those found in overweight non-operated subjects with similar fasting blood glucose levels [30]. Interestingly, variations in ghrelin levels occurred in both groups during the OGTT, where non-responders returned earlier to baseline levels. Diurnal and postprandial variations of ghrelin are otherwise known to disappear in obese subjects, and postprandial levels of ghrelin to be suppressed up to 2 years after RYGBP [29, 31]. These findings suggest that, early postoperative changes notwithstanding, leptin and ghrelin levels in non-responders many years after surgically induced weight loss resemble those of non-operated obese individuals, probably due to preserved or regained adiposity. The earlier rise of ghrelin to baseline level combined with leptin decreasing below baseline level at the end of the OGTT in non-responders would presumably have a stimulating effect on appetite, thus favoring weight gain.
Leptin is known to cross the blood-brain barrier, acting on hypothalamic neurons. The ghrelin assay in this study is highly selective for acyl-ghrelin, which preferentially (compared with other forms of ghrelin) crosses the blood-brain barrier, engaging GHS1R receptors of hypothalamic neurons [32]. It is intriguing that, although there were clear differences in both leptin and ghrelin, the ratios between the two hormones were preserved between responders and non-responders. The net consequences of these different permutations of ghrelin and leptin for hunger and reward systems of the hypothalamus are unclear. The neural circuitry of these systems is complex, involving several types of neurons [33]. Cross-talk between ghrelin and leptin at different concentrations through, for example, AgRP/NPY neurons when their ratios are clamped may be a fruitful line of future research.
Fasting levels of GLP-1, PYY, and GIP, as well as their responses to an oral glucose load, were similar in responders and non-responders. After RYGBP, GLP-1 increases progressively during the first postoperative year, albeit with considerable delay after improvement in glucose homeostasis and weight loss [5], and therefore, the causative effect of GLP-1 on these changes has been challenged. As reported earlier, the plasma levels of GLP-1 in the short term after RYGBP [34], both our groups present a 30-min peak, with GLP-1 levels increasing tenfold compared with baseline. Similar to GLP-1, RYGBP surgery increases fasting [35] as well as postprandial PYY levels postoperatively compared with lean and obese control subjects [36, 37]. This study shows none of the short-term changes in these two hormones reported to be associated with the long-term weight result, despite typical postoperative gut hormone patterns [36]. Moreover, GLP-1 analogues have been introduced as a pharmacological therapy for weight loss [38]. The similar GLP-1 responses in responders and non-responders can be interpreted to indicate that use of GLP-1 analogues in non-responders to improve weight loss after RYGBP probably increases the circulating pool of GLP-1 agonists to super-physiological levels, as opposed to restoring sub-physiological levels to normal levels.
The prevalence of T2DM in subjects with morbid obesity undergoing RYGBP, about 16%, is reduced postsurgically [39]. In the subgroup analysis, the T2DM group showed lower fasting ghrelin and PYY than the weight-similar normoglycemic group. Lower levels of GLP-1 in response to an oral glucose load could explain relapse to diabetes years after RYGBP in individuals with T2DM prior to their surgery.
Limitations
This study is limited by the lack of longitudinal data, like the participants’ preoperative hormone levels or preoperative hormonal responses to an OGTT, nor were non-hormonal factor, e.g., eating habits and physical exercise studied. Also, while providing a degree of uniformity, the inclusion of only female participants operated with shorter limb lengths than those used nowadays may be a limiting factor. In addition, there is a large variation in follow-up years within each group but as all participants were operated more than 5 years prior to the present investigations, this study represents long-term outcomes of bariatric surgery.
Conclusion
Poor long-term weight result and glycemic status after RYGBP are each associated with differences in peptide hormone levels. Although correlations between weight result and fasting leptin and ghrelin levels may in part be attributed to adiposity in long-term non-responders after RYGBP, the temporal pattern of these hormonal responses to an oral glucose load suggests they might promote obesity by way of hunger/satiety signaling. This stresses the importance of early identification of non-responders to RYGBP surgery. Lower GLP-1 levels seen in this postoperative study in subjects that had T2DM prior to surgery could explain their hyperglycemic status years after RYGBP.
References
Gloy VL, Briel M, Bhatt DL, et al. Bariatric surgery versus non-surgical treatment for obesity: a systematic review and meta-analysis of randomised controlled trials. BMJ (Clinical research ed). 2013;347:f5934.
Colquitt JL, Pickett K, Loveman E, et al. Surgery for weight loss in adults. Cochrane Database Syst Rev. 2014;8:Cd003641.
Angrisani L, Santonicola A, Iovino P, et al. Bariatric surgery worldwide 2013. Obes Surg. 2015;25(10):1822–32.
Maciejewski ML, Arterburn DE, Van Scoyoc L, et al. Bariatric surgery and long-term durability of weight loss. JAMA Surg. 2016;151(11):1046–55.
Falken Y, Hellstrom PM, Holst JJ, et al. Changes in glucose homeostasis after Roux-en-Y gastric bypass surgery for obesity at day three, two months, and one year after surgery: role of gut peptides. J Clin Endocrinol Metab. 2011;96(7):2227–35.
Magro DO, Geloneze B, Delfini R, et al. Long-term weight regain after gastric bypass: a 5-year prospective study. Obes Surg. 2008;18(6):648–51.
SOReg. Year report 2015 - Part 2. Scandinavian Obesity Surgery Registry (SOReg) [homepage on the Internet] Sweden: Year report 2015 [in Swedish] [cited 2016 October] Available from: http://www.ucruuse/soreg/component/edocman/2016-10-19-arsrapport-2015-del-2. 2016 2016-10-19.
Reinhold RB. Critical analysis of long term weight loss following gastric bypass. Surg Gynecol Obstet. 1982;155(3):385–94.
Mantzoros CS. The role of leptin in human obesity and disease: a review of current evidence. Ann Intern Med. 1999;130(8):671–80.
Sumithran P, Prendergast LA, Delbridge E, et al. Long-term persistence of hormonal adaptations to weight loss. N Engl J Med. 2011;365(17):1597–604.
Pan H, Guo J, Su Z. Advances in understanding the interrelations between leptin resistance and obesity. Physiol Behav. 2014;130:157–69.
Klok MD, Jakobsdottir S, Drent ML. The role of leptin and ghrelin in the regulation of food intake and body weight in humans: a review. Obes Rev. 2007;8(1):21–34.
Rao RS, Kini S. GIP and bariatric surgery. Obes Surg. 2011;21(2):244–52.
Garcia JM, Iyer D, Poston WSC, et al. Rise of plasma ghrelin with weight loss is not sustained during weight maintenance. Obesity. 2006;14(10):1716–23.
Iepsen EW, Lundgren J, Holst JJ, et al. Successful weight loss maintenance includes long-term increased meal responses of GLP-1 and PYY3-36. Eur J Endocrinol. 2016;174(6):775–84.
Adam TCM, Westerterp-Plantenga MS. Glucagon-like peptide-1 release and satiety after a nutrient challenge in normal-weight and obese subjects. Br J Nutr. 2005;93(6):845–51.
Gibbons C, Finlayson G, Caudwell P, et al. Postprandial profiles of CCK after high fat and high carbohydrate meals and the relationship to satiety in humans. Peptides. 2016;77:3–8.
Halim MA, Gillberg L, Boghus S, et al. Nitric oxide regulation of migrating motor complex: randomized trial of N(G)-monomethyl-L-arginine effects in relation to muscarinic and serotonergic receptor blockade. Acta Physiol (Oxford, England). 2015;215(2):105–18.
Lee I, Shi L, Webb DL, et al. Effects of whole-grain rye porridge with added inulin and wheat gluten on appetite, gut fermentation and postprandial glucose metabolism: a randomised, cross-over, breakfast study. Br J Nutr. 2016;116(12):2139–49.
Kim KR, Rhee SD, Kim HY, et al. KR-62436, 6-{2-[2-(5-cyano-4,5-dihydropyrazol-1-yl)-2-oxoethylamino]ethylamino} nicotinonitr ile, is a novel dipeptidyl peptidase-IV (DPP-IV) inhibitor with anti-hyperglycemic activity. Eur J Pharmacol. 2005;518(1):63–70.
Lodeiro M, Theodoropoulou M, Pardo M, et al. c-Src regulates Akt signaling in response to ghrelin via beta-arrestin signaling-independent and -dependent mechanisms. PLoS One. 2009;4(3):e4686.
Matsuda M, DeFronzo RA. Insulin sensitivity indices obtained from oral glucose tolerance testing: comparison with the euglycemic insulin clamp. Diabetes Care. 1999;22(9):1462–70.
Kadowaki T, Miyake Y, Hagura R, et al. Risk factors for worsening to diabetes in subjects with impaired glucose tolerance. Diabetologia. 1984;26(1):44–9.
Karmali S, Brar B, Shi X, et al. Weight recidivism post-bariatric surgery: a systematic review. Obes Surg. 2013;23(11):1922–33.
van de Laar A. Bariatric Outcomes Longitudinal Database (BOLD) suggests excess weight loss and excess BMI loss to be inappropriate outcome measures, demonstrating better alternatives. Obes Surg. 2012;22(12):1843–7.
Woelnerhanssen B, Peterli R, Steinert RE, et al. Effects of postbariatric surgery weight loss on adipokines and metabolic parameters: comparison of laparoscopic Roux-en-Y gastric bypass and laparoscopic sleeve gastrectomy--a prospective randomized trial. Surg Obes Relat Dis. 2011;7(5):561–8.
Carlson JJ, Turpin AA, Wiebke G, et al. Pre- and post- prandial appetite hormone levels in normal weight and severely obese women. Nutr Metab (Lond). 2009;6:32.
Karamanakos SN, Vagenas K, Kalfarentzos F, et al. Weight loss, appetite suppression, and changes in fasting and postprandial ghrelin and peptide-YY levels after Roux-en-Y gastric bypass and sleeve gastrectomy: a prospective, double blind study. Ann Surg. 2008;247(3):401–7.
Malin SK, Samat A, Wolski K, et al. Improved acylated ghrelin suppression at 2 years in obese patients with type 2 diabetes: effects of bariatric surgery vs standard medical therapy. Int J Obes (2005). 2014;38(3):364–70.
Gibbons C, Caudwell P, Finlayson G, et al. Comparison of postprandial profiles of ghrelin, active GLP-1, and total PYY to meals varying in fat and carbohydrate and their association with hunger and the phases of satiety. J Clin Endocrinol Metab. 2013;98(5):E847–55.
Cummings DE, Weigle DS, Frayo RS, et al. Plasma ghrelin levels after diet-induced weight loss or gastric bypass surgery. N Engl J Med. 2002;346(21):1623–30.
Banks WA, Tschop M, Robinson SM, et al. Extent and direction of ghrelin transport across the blood-brain barrier is determined by its unique primary structure. J Pharmacol Exp Ther. 2002;302(2):822–7.
Cassidy RM, Tong Q. Hunger and satiety gauge reward sensitivity. Front Endocrinol. 2017;8:104.
Anandhakrishnan A, Korbonits M. Glucagon-like peptide 1 in the pathophysiology and pharmacotherapy of clinical obesity. World J Diabetes. 2016;7(20):572–98.
Ballantyne GH. Peptide YY(1-36) and peptide YY(3-36): Part II. Changes after gastrointestinal surgery and bariatric surgery. Obes Surg. 2006;16(6):795–803.
Jacobsen SH, Olesen SC, Dirksen C, et al. Changes in gastrointestinal hormone responses, insulin sensitivity, and beta-cell function within 2 weeks after gastric bypass in non-diabetic subjects. Obes Surg. 2012;22(7):1084–96.
Chan JL, Mun EC, Stoyneva V, et al. Peptide YY levels are elevated after gastric bypass surgery. Obesity. 2006;14(2):194–8.
Webb DL, Abrahamsson N, Sundbom M, et al. Bariatric surgery - time to replace with GLP-1? Scand J Gastroenterol. 2017;52(6–7):635–40.
Sundbom M, Hedberg J, Marsk R, et al. Substantial decrease in comorbidity 5 years after gastric bypass: a population-based study from the Scandinavian Obesity Surgery Registry. Ann Surg. 2017;265(6):1166–71.
Acknowledgements
Open access funding provided by Uppsala University.
Funding
ES, DLW, PH, and MS—ALF funds, Sweden. DLW—Bengt Ihre Foundation, Swedish Medical Association, Gastroenterology Research Foundation.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Ethics Statement
The local ethics review board (EPN Uppsala) approved this study, diary number 2013/047/1.
Consent Statement
All participants submitted written consent prior to the examinations.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Sima, E., Webb, DL., Hellström, P.M. et al. Non-responders After Gastric Bypass Surgery for Morbid Obesity: Peptide Hormones and Glucose Homeostasis. OBES SURG 29, 4008–4017 (2019). https://doi.org/10.1007/s11695-019-04089-8
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11695-019-04089-8