Abstract
Background
Overweight and obesity dramatically increased in the last years. Hepatic complication of obesity, integrated in the term of non-alcoholic fatty liver disease (NAFLD), is a spectrum of abnormality ranging from steatosis to non-alcoholic steatohepatitis (NASH), potentially leading to cirrhosis. Liver biopsy remains the gold standard to evaluate the stage of NAFLD; however, the procedure is invasive. The indocyanine green (ICG) clearance test is performed since years to assess hepatic function before partial hepatectomy, or after liver transplantation. This study was designed to detect liver complications with the ICG clearance test in a population of obese patients scheduled for bariatric surgery.
Methods
In a prospective cohort study, morbidly obese individuals receiving bariatric surgery with scheduled hepatic biopsies were investigated. Liver function was determined by the ICG test preoperatively, and blood samples were collected. Liver biopsy specimens were obtained for each patient and classified according to the NAFLD activity score (NAS) by a single pathologist that was blinded to the results of the ICG test.
Results
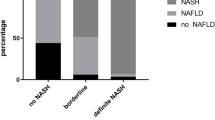
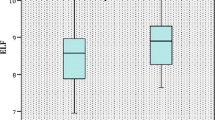
Twenty-six patients were included (7 male and 19 female). The mean age of participants was 45.8 years; the mean body mass index was 41.4 kg/m2. According to the NAS, 6 (23.1%) patients revealed manifest NASH, and 5 patients were considered borderline (19.2%). A closed correlation was observed between the ICG clearance test and hepatic steatosis (r = 0.43, p = 0.03), NAS (r = 0.44, p = 0.025), and fibrosis (r = 0.49, p = 0.01).
Conclusions
In obese patients, non-invasive evaluation of liver function with the indocyanine green clearance test correlated with histological features of NAFLD. This may detect non-invasively hepatopathy in obese population and could motive biopsy.

Similar content being viewed by others
References
Kelly T, Yang W, Chen C-S, et al. Global burden of obesity in 2005 and projections to 2030. Int J Obes. 2008;32:1431–7.
Alberti KGMM, Eckel RH, Grundy SM, et al. Harmonizing the metabolic syndrome: a joint interim statement of the International Diabetes Federation Task Force on Epidemiology and Prevention; National Heart, Lung, and Blood Institute; American Heart Association; World Heart Federation; International Atherosclerosis Society; and International Association for the Study of Obesity. Circulation. 2009;120:1640–5.
Angulo P. Nonalcoholic fatty liver disease. N Engl J Med. 2002;346:1221–31.
Younossi ZM, Koenig AB, Abdelatif D, et al. Global epidemiology of nonalcoholic fatty liver disease—meta-analytic assessment of prevalence, incidence, and outcomes. Hepatology; 2016;64:73–84.
Sanyal AJ, Banas C, Sargeant C, et al. Similarities and differences in outcomes of cirrhosis due to nonalcoholic steatohepatitis and hepatitis C. Hepatology. 2006;43:682–9.
Anty R, Iannelli A, Patouraux S, et al. A new composite model including metabolic syndrome, alanine aminotransferase and cytokeratin-18 for the diagnosis of non-alcoholic steatohepatitis in morbidly obese patients. Aliment Pharmacol Ther. 2010;32:1315–22.
Machado MV, Cortez-Pinto H. Non-invasive diagnosis of non-alcoholic fatty liver disease. A critical appraisal. J. Hepatol. 2013;58:1007–19.
Brunt EM, Kleiner DE, Wilson LA, et al. Nonalcoholic fatty liver disease (NAFLD) activity score and the histopathologic diagnosis in NAFLD: distinct clinicopathologic meanings. Hepatology. 2011;53:810–20.
Chalasani N, Younossi Z, Lavine JE, et al. The diagnosis and management of non-alcoholic fatty liver disease: practice guideline by the American Gastroenterological Association, American Association for the Study of Liver Diseases, and American College of Gastroenterology. Gastroenterology. 2012;142:1592–609.
Bravo AA, Sheth SG, Chopra S. Liver biopsy. N Engl J Med. 2001;344:495–500.
Piccinino F, Sagnelli E, Pasquale G, et al. Complications following percutaneous liver biopsy. A multicentre retrospective study on 68,276 biopsies. J. Hepatol. 1986;2:165–73.
Dyson JK, McPherson S, Anstee QM. Non-alcoholic fatty liver disease: non-invasive investigation and risk stratification. J Clin Pathol. 2013;66:1033–45.
Janssen MWW, Druckrey-Fiskaaen KT, Omidi L, et al. Indocyanine green R15 ratio depends directly on liver perfusion flow rate. J Hepato-Biliary-Pancreat Sci. 2010;17:180–5.
Levesque E, Martin E, Dudau D, et al. Current use and perspective of indocyanine green clearance in liver diseases. Anaesth Crit Care Pain Med. 2016;35:49–57.
Imamura H, Sano K, Sugawara Y, et al. Assessment of hepatic reserve for indication of hepatic resection: decision tree incorporating indocyanine green test. J Hepato-Biliary-Pancreat Surg. 2005;12:16–22.
de Liguori CN, O’Reilly DA, Dajani K, et al. Perioperative use of the LiMON method of indocyanine green elimination measurement for the prediction and early detection of post-hepatectomy liver failure. Eur. J. Surg. Oncol. J. Eur. Soc. Surg. Oncol. Br. Assoc. Surg. Oncologia. 2009;35:957–62.
Thomas MN, Weninger E, Angele M, et al. Intraoperative simulation of remnant liver function during anatomic liver resection with indocyanine green clearance (LiMON) measurements. HPB. 2015;17:471–6.
Haegele S, Reiter S, Wanek D, et al. Perioperative non-invasive Indocyanine green-clearance testing to predict postoperative outcome after liver resection. PLoS One. 2016;11:e0165481.
National Task Force on the Prevention and Treatment of Obesity. Overweight, obesity, and health risk. Arch. Intern. Med. 2000;160:898–904.
Vos JJ, Wietasch JKG, Absalom AR, et al. Green light for liver function monitoring using indocyanine green? An overview of current clinical applications. Anaesthesia. 2014;69:1364–76.
Kleiner DE, Brunt EM, Van Natta M, et al. Design and validation of a histological scoring system for nonalcoholic fatty liver disease. Hepatology. 2005;41:1313–21.
Iannelli A, Schneck A-S, Hébuterne X, et al. Gastric pouch resizing for Roux-en-Y gastric bypass failure in patients with a dilated pouch. Surg Obes Relat Dis Off J Am Soc Bariatr Surg. 2013;9:260–7.
Ratziu V, Charlotte F, Heurtier A, et al. Sampling variability of liver biopsy in nonalcoholic fatty liver disease. Gastroenterology. 2005;128:1898–906.
Goossens N, Hoshida Y, Song WM, et al. Nonalcoholic steatohepatitis is associated with increased mortality in obese patients undergoing bariatric surgery. Clin Gastroenterol Hepatol. 2016 ;14:1619-1628.
Machado M, Marques-Vidal P, Cortez-Pinto H. Hepatic histology in obese patients undergoing bariatric surgery. J Hepatol. 2006;45:600–6.
Fracanzani AL, Valenti L, Bugianesi E, et al. Risk of severe liver disease in nonalcoholic fatty liver disease with normal aminotransferase levels: a role for insulin resistance and diabetes. Hepatology. 2008;48:792–8.
Chalasani N. Nonalcoholic fatty liver disease liver fat score and fat equation to predict and quantitate hepatic steatosis: promising but not prime time! Gastroenterology. 2009;137:772–5.
Cusi K, Chang Z, Harrison S, et al. Limited value of plasma cytokeratin-18 as a biomarker for NASH and fibrosis in patients with non-alcoholic fatty liver disease. J Hepatol. 2014;60:167–74.
Mehta SR, Thomas EL, Patel N, et al. Proton magnetic resonance spectroscopy and ultrasound for hepatic fat quantification. Hepatol Res. 2010;40:399–406.
Hernaez R, Lazo M, Bonekamp S, et al. Diagnostic accuracy and reliability of ultrasonography for the detection of fatty liver: a meta-analysis. Hepatology. 2011;54:1082–90.
Tsochatzis EA, Gurusamy KS, Ntaoula S, et al. Elastography for the diagnosis of severity of fibrosis in chronic liver disease: a meta-analysis of diagnostic accuracy. J Hepatol. 2011;54:650–9.
Castéra L, Foucher J, Bernard P-H, et al. Pitfalls of liver stiffness measurement: a 5-year prospective study of 13,369 examinations. Hepatology. 2010;51:828–35.
Stern C, Castera L. Non-invasive diagnosis of hepatic steatosis. Hepatol Int. 2017;11:70–8.
Hedderich DM, Hasenberg T, Haneder S, et al. Effects of bariatric surgery on non-alcoholic fatty liver disease: magnetic resonance imaging is an effective, non-invasive method to evaluate changes in the liver fat fraction. Obes Surg. 2017;27:1755-1762.
Dulai PS, Sirlin CB, Loomba R. MRI and MRE for non-invasive quantitative assessment of hepatic steatosis and fibrosis in NAFLD and NASH: clinical trials to clinical practice. J Hepatol. 2016;65:1006–16.
Loomba R, Wolfson T, Ang B, et al. Magnetic resonance elastography predicts advanced fibrosis in patients with nonalcoholic fatty liver disease: a prospective study. Hepatology. 2014;60:1920–8.
Sakka SG. Assessing liver function. Curr Opin Crit Care. 2007;13:207–14.
Shigefuku R, Takahashi H, Nakano H, et al. Correlations of hepatic hemodynamics, liver function, and fibrosis markers in nonalcoholic fatty liver disease: comparison with chronic hepatitis related to hepatitis C virus. Int J Mol Sci. 2016;17(9). https://doi.org/10.3390/ijms17091545.
Yang Y-L, Di L, Duan Y-Y, et al. A prospective experimental study of liver fibrosis with ultrasound and its correlation with hepatic reserve function and hemodynamics. BMC Gastroenterol. 2012;12:168.
Angulo P, Kleiner DE, Dam-Larsen S, et al. Liver fibrosis, but no other histologic features, is associated with long-term outcomes of patients with nonalcoholic fatty liver disease. Gastroenterology. 2015;149:389–397.e10.
Ekstedt M, Hagström H, Nasr P, et al. Fibrosis stage is the strongest predictor for disease-specific mortality in NAFLD after up to 33 years of follow-up. Hepatology. 2015;61:1547–54.
Pind M-LL, Bendtsen F, Kallemose T, et al. Indocyanine green retention test (ICG-r15) as a noninvasive predictor of portal hypertension in patients with different severity of cirrhosis. Eur J Gastroenterol Hepatol. 2016;28:948–54.
Fisher CD, Lickteig AJ, Augustine LM, et al. Experimental non-alcoholic fatty liver disease results in decreased hepatic uptake transporter expression and function in rats. Eur J Pharmacol. 2009;613:119–27.
Alizai PH, Wendl J, Roeth AA, et al. Functional liver recovery after bariatric surgery—a prospective cohort study with the LiMAx test. Obes Surg. 2015;25:2047–53.
Lassailly G, Caiazzo R, Buob D, et al. Bariatric surgery reduces features of nonalcoholic steatohepatitis in morbidly obese patients. Gastroenterology. 2015;149:379–88.
Schneck AS, Anty R, Patouraux S, et al. Roux-en Y gastric bypass results in long-term remission of hepatocyte apoptosis and hepatic histological features of non-alcoholic steatohepatitis. Front Physiol. 2016;7:344.
Financial Support
This work was supported by grants from INSERM (France), the University of Nice, the Programme Hospitalier de Recherche Clinique (Centre Hospitalier Universitaire of Nice), and charities (Association Française pour l’Etude du Foie (AFEF)/LFB to PG, Société Francophone du Diabète (SFD) to PG). This work was also funded by the French Government (National Research Agency, ANR) through the “Investments for the Future” LABEX SIGNALIFE: program reference #ANR-11-LABX-0028-01 and #ANR-15-CE14-0016-01.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Written consent was obtained from each patient before enrolment. All procedures performed in this study were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Conflict of Interest
Pierre Eric DANIN has been a consultant for PULSION (GETINGE).
Additional information
Philippe Gual and Antonio Iannelli jointly supervised this work.
Rights and permissions
About this article
Cite this article
Danin, P.E., Anty, R., Patouraux, S. et al. Non-invasive Evaluation of NAFLD with Indocyanine Green Clearance Test: a Preliminary Study in Morbidly Obese Patients Undergoing Bariatric Surgery. OBES SURG 28, 735–742 (2018). https://doi.org/10.1007/s11695-017-2914-0
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11695-017-2914-0