Abstract
Background
Surgical treatment of obesity is characterized by both early and late complications, and thus, there is a need to develop safe and non-invasive techniques. Ghrelin is an orexigenic hormone produced by the fundus of the stomach, which may represent a novel target for obesity management. Unfortunately, numerous attempts to alter ghrelin levels have failed to present significant clinical results. We describe a novel procedure that involves modifying arterial blood flow to the gastric fundus for limiting plasma ghrelin levels.
Methods
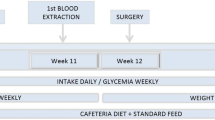
A gastroscope was advanced into the gastric fundus of 13 healthy Yorkshire swine, and the fundus was clipped under direct visualization to restrict left gastric artery blood flow. Body weights and ghrelin levels were recorded before and once a week for 4 weeks after the procedure.
Results
Compared to controls, gastroscopic clipping of the fundus decreased plasma ghrelin levels and prevented further weight gain in the 4 weeks of follow-up. Immunohistochemistry and histomorphometry revealed reduced numbers of ghrelin-positive cells in the fundus of experimental animals. We also observed thrombosis in submucosal arteries and submucosal fibrosis. Histological studies demonstrated minimal gastric mucosal injury.
Conclusion
Gastroscopic clipping of the fundus in an experimental porcine model resulted in sustained weight loss and a reduction in plasma ghrelin levels at 1 month post-procedure, with no adverse events. Further experimental studies in human patients are needed to examine the clinical utility of this procedure and to optimize a technique, which can facilitate adequate weight loss while minimizing the risk of mucosal injury.




Similar content being viewed by others
References
Ng M et al. Global burden of disease study 2013 collaborators. Global, regional, and national incidence, prevalence, and years lived with disability for 301 acute and chronic diseases and injuries in 188 countries, 1990–2013: a systematic analysis for the global burden of disease study 2013. Lancet. 2014;384(9945):766–81.
Güngör NK. Overweight and obesity in children and adolescents. J Clin Res Pediatr Endocrinol. 2014;6(3):129–43.
Haslam DW, James WP. Obesity. Lancet. 2005;366(9492):1197–209.
Kopelman PG. Obesity as a medical problem. Nature. 2000;404(6778):635–43.
Lean M. Cutting fat absorption to halt weight gain. Practitioner. 1998;242(1593):860–5.
Barness LA. Obesity in children. Fetal Pediatr Pathol. 2007;26(2):75–85.
Barness LA, Opitz JM, Gilbert-Barness E. Obesity: genetic, molecular, and environmental aspects. Am J Med Genet A. 2007;143A(24):3016–34.
Strader AD, Woods SC. Gastrointestinal hormones and food intake. Gastroenterology. 2005;128:175–91.
Cummings DE, Shannon MH. Ghrelin and gastric bypass: is there a hormonal contribution to surgical weight loss? J Clin Endocrinol Metab. 2003;88:2999–3002.
Hu Z, Cha SH, Haasteren G, et al. Effect of centrally administered C75, a fatty acid synthase inhibitor, on ghrelin secretion and its downstream effects. Proc Natl Acad Sci U S A. 2005;102:3972–7.
Murray CD, Kamm MA, Bloom SR, et al. Ghrelin for the gastroenterologist: history and potential. Gastroenterology. 2003;125:1492–502.
Flegal KM, Carroll MD, Ogden CL, et al. Prevalence and trends in obesity among US adults, 1999-2000. JAMA. 2002;288:1723–7.
Cigaina V. Long-term follow-up of gastric stimulation for obesity: the Mestre 8-year experience. Obes Surg. 2004;14(Suppl 1):S14–22.
Hanusch-Enserer U, Cauza E, Brabant G, et al. Plasma ghrelin in obesity before and after weight loss after laparoscopical adjustable gastric banding. J Clin Endocrinol Metab. 2004;89:3352–8.
Leonetti F, Silecchia G, Iacobellis G, et al. Different plasma ghrelin levels after laparoscopic gastric bypass and adjustable gastric banding in morbid obese subjects. J Clin Endocrinol Metab. 2003;88:4227–31.
Arepally A, Barnett BP, Patel TH, et al. Catheter-directed gastric artery chemical embolization suppresses systemic ghrelin levels in porcine model. Radiology. 2008;249:127–33.
Inui A, Asakawa A, Bowers CY, et al. Ghrelin, appetite, and gastric motility: the emerging role of the stomach as an endocrine organ. FASEB journal: official publication of the Federation of American Societies for Experimental Biology. 2004;18:439–56.
Druce MR, Small CJ, Bloom SR. Minireview: gut peptides regulating satiety. Endocrinology. 2004;145:2660–5.
Bawudun D, Xing Y, Liu WY, et al. Ghrelin suppression and fat loss after left gastric artery embolization in canine model. Cardiovasc Intervent Radiol. 2012;35(6):1460–6.
Paxton BE, Kim CY, Alley CL, et al. Bariatric embolization for suppression of the hunger hormone ghrelin in a porcine model. Radiology. 2013;266:471–9.
Kipshidze N, Archvadze A, Bertog S, et al. Endovascular bariatrics: first in humans study of gastric artery embolization for weight loss. JACC Cardiovasc Interv. 2015;8(12):1641–4.
Joseph B, Berishvili E, Benten D, et al. Isolated small intestinal segments support auxiliary livers with maintenance of hepatic functions. Nat Med. 2004;10(7):749–53.
Kakabadze A, Kakabadze Z. Prospect of using decellularized human placenta and cow placentome for creation of new organs: targeting the liver (part I: anatomic study. Transplant Proc. 2015;47(4):1222–7.
Angrisani L, Santonicola A, Iovino P, et al. Bariatric surgery worldwide 2013. Obes Surg. 2015;25(10):1822–32.
Kumar N. Endoscopic therapy for weight loss: Gastroplasty, duodenal sleeves, intragastric balloons, and aspiration. World J Gastrointest Endosc. 2015;7(9):847–59.
Dansinger ML, Tatsioni A, Wong JB, et al. Meta-analysis: the effect of dietary counseling for weight loss. Ann Intern Med. 2007;147(1):41–50.
Gersin KS, Rothstein RI, Rosenthal RJ, et al. Open-label, sham-controlled trial of an endoscopic duodenojejunal bypass liner for preoperative weight loss in bariatric surgery candidates. Gastrointest Endosc. 2010;71:976–82.
Sullivan S, Stein R, Jonnalagadda S, et al. Aspiration therapy leads to weight loss in obese subjects: a pilot study. Gastroenterology. 2013;145:1245–52.e1-5.
Ponce J, Quebbemann BB, Patterson EJ. Prospective, randomized, multicenter study evaluating safety and efficacy of intragastric dual-balloon in obesity. Surg Obes Relat Dis. 2013;9:290–5.
Mion F, Ibrahim M, Marjoux S, et al. Swallowable Obalon® gastric balloons as an aid for weight loss: a pilot feasibility study. Obes Surg. 2013;23:730–3.
Marinos G, Eliades C, Muthusamy V, et al. First clinical experience with the transpyloric shuttle device, a non-surgical endoscopic treatment for obesity: results from a 3-month and 6-month study. SAGES. 2013: Abstract.
Sauer N, Rösch T, Pezold J, et al. A new endoscopically implantable device (SatiSphere) for treatment of obesity—efficacy, safety, and metabolic effects on glucose, insulin, and GLP-1 levels. Obes Surg. 2013;23:1727–33.
Shikora S, Toouli J, Herrera MF, et al. Vagal blocking improves glycemic control and elevated blood pressure in obese subjects with type 2 diabetes mellitus. J Obes. 2013;2013:245683.
Shikora SA, Bergenstal R, Bessler M, et al. Implantable gastric stimulation for the treatment of clinically severe obesity: results of the SHAPE trial. Surg Obes Relat Dis. 2009;5(1):31–7.
Diana M, Halvax P, Pop R, et al. Gastric supply manipulation to modulate ghrelin production and enhance vascularization to the cardia: proof of the concept in a porcine model. Surg Innov. 2015;22(1):5–14.
Amesur NB, Zajko AB, Philip D, et al. Interventional radiology techniques for intra-arterial chemoembolization. Hepatocell Cancer Curr Clin Oncol. Springer 2005. 215–231.
Bradley EL, Goldman ML. Gastric infarction after therapeutic embolization. Surgery. 1976;79(4):421–4.
Brown KT, Friedman WN, Marks RA, et al. Gastric and hepatic infarction following embolization of the left gastric artery: case report. Radiology. 1989;172(3):731–2.
Bilbao JI, Martínez-Cuesta A, Urtasun F, et al. Complications of embolization. Semin intervent Radiol. 2006;23(2):126–42.
Prochaska JM, Flye MW, Johnsrude IS. Left gastric artery embolization for control of gastric bleeding: a complication. Radiology. 1973;107(3):521–2.
Kiyono S, Maruyama H, Kondo T, et al. Hemodynamic effect of the left gastric artery on esophageal varices in patients with cirrhosis. J Gastroenterol. 2016;51(9):900–9.
Waxman I, Saitoh Y, Raju GS, et al. High-frequency probe EUS-assisted endoscopic mucosal resection: a therapeutic strategy for submucosal tumors of the GI tract. Gastrointest Endosc. 2002;55:44–9.
Raju GS, Waxman I. High-frequency US probe sonography-assisted endoscopic mucosal resection. Gastrointest Endosc. 2000;52(Suppl 6):S39–49.
Acknowledgments
The authors would like to thank Endobar Solutions LLC for funding this research.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
Nickolas Kipshidze is the Founder and major shareholder of Endobar Solutions LLC; Dr. Kipshidze reports research grant from Endobar Solutions, LLC, during the conduct of the study. In addition, author 1 has a patent I.P. pending.
Anaka Prakash—no conflicts of interest.
Nodar Kipshidze—no conflicts of interest.
David Chakhunashvili—no conflicts of interest.
Zurab Kakabadze—no conflicts of interest
Rights and permissions
About this article
Cite this article
Kipshidze, N., Prakash, A., Kipshidze, N. et al. A Novel Endoscopic Bariatric Procedure: Results of an Experimental Study. OBES SURG 26, 3058–3065 (2016). https://doi.org/10.1007/s11695-016-2389-4
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11695-016-2389-4