Abstract
Aims and Methods
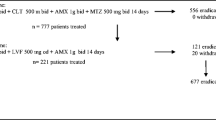
Our aim was to assess, in obese patients undergoing Roux-en Y gastric bypass surgery, the quadruple concomitant HP eradication rates at first line treatment as proposed by the Maastricht IV consensus in areas of high clarithromycin resistance rates—proton pump inhibitor bid, clarithromycin 500 mg bid, amoxicillin 1000 mg bid, and metronidazole 500 mg bid. This is a single center prospective study over a 3-year period. Endoscopy and HP assessment (by histology or C13 urea breath) were performed at baseline, and post treatment HP status was assessed by C13 urea breath test 4–6 weeks after the end of therapy.
Results
The study cohort consisted of 600 adult obese HP positive patients [19 % male/81 % female, age 40.7 (±10.4) years] consecutively scheduled for HP concomitant therapy. HP was eradicated in 416 patients [69.3 % (95% CI 65.5–72.9 %)] and the eradication was independent of gender, age, endoscopic diagnosis, and smoking status (p > 0.05).
Conclusions
Two weeks quadruple concomitant therapy did not achieve Maastricht recommended first line acceptable HP eradication rates (at least 80 %) in obese Portuguese patients undergoing GB.
Similar content being viewed by others
References
Quina M, Guerreiro A. Gastric cancer (carcinoma) and Helicobacter pylori: situation in Portugal. Hepato Gastroenterol. 2001;48:1565–8.
Ferlay J, Autier P, Boniol M, et al. Estimates of cancer incidence and mortality in Europe in 2006. Ann Oncology. 2007;18:581–92.
Cerqueira RM, Manso MC, Correia MR, et al. Helicobacter pylori eradication therapy in obese patients undergoing gastric by-pass surgery – fourteen days superior to seven days? Obes Surg. 2011;21(9):1377–81.
Watkins BJ, Blackmun S, Kuehner ME. Gastric adenocarcinoma after Roux-en-Y gastric bypass: access and evaluation of excluded stomach. Surg Obes Relat Dis. 2007;3:644–7.
Silechia G, Catalano C, Gentileschi P, et al. Virtual gastroduodenoscopy: a new look at the bypassed stomach and duodenum after laparoscopic Roux-en-Y gastric bypass for morbid obesity. Obes Surg. 2002;12:339–48.
Gill KR, McKinney JM, Stark ME, et al. Investigation of the excluded stomach after Roux-en-Y gastric bypass: the role of percutaneous endoscopy. World J Gastroenterol. 2008;14:1946–8.
Yamamoto H, Sekine Y, Sato Y, et al. Total enteroscopy with a nonsurgical steerable double-balloon method. Gastrointest Endosc. 2001;53:216–20.
Fried M, Hainer V, Basdevant A, et al. Inter-disciplinary European Guidelines on surgery of severe obesity. Int J Obes. 2007;31:569–77.
ASGE Guidelines. Role of endoscopy in the bariatric patient. Gastrointest Endosc. 2008;68:1–10.
Orlando G, Pilone V, Vittiello A, et al. Gastric cancer following bariatric surgery. Surg Laparosc Endosc Percutan Tech. 2014;24:400–5.
Malfertheiner P, Megraud F, O’Morain C, et al. Current concepts in the management of Helicobacter pylori infection: the Maastricht III Consensus Report. Gut. 2007;56:772–81.
Malfertheiner P, Megraud F, O’Morain C, et al. Management of Helicobacter pylori infection—the Maastricht IV /Florence Consensus Report. Gut. 2012;61:646–64.
Cerqueira RM, Correia MR, Fernandes CD, et al. Cumulative Helicobacter pylori eradication therapy in obese patients undergoing gastric bypass surgery. Obes Surg. 2013;23(2):145–8.
Graham DY, Fischbach L. Helicobacter pylori treatment in the era of increasing antibiotic resistance. Gut. 2010;59:1143–53.
Boyanova L. Prevalence of multi-drug resistant Helicobacter pylori in Bulgaria. J Med Microbiol. 2009;58(Pt 7):930–5.
Mathers CD, Loncar D. Projections of global mortality and burden of disease from 2002 to 2030. PLoS Med. 2006;3:e 442.
Mégraud F. H pylori antibiotic resistance: prevalence, importance and advances in testing. Gut. 2004;53:1374–84.
Wu DC, Hsu PI, Wu JY, et al. Sequential and concomitant therapy with 4 drugs are equally effective for eradication of H pylori infection. Clin Gastroenterol Hepatol. 2010;8:36–41.
Molina Infante J, Pazos Pacheco C, Vinagre Rodriguez G, et al. Non bismuth quadruple concomitant therapy versus standard triple therapy for clarithromycin susceptible H pylori and versus quadruple sequential therapy for clarithromycin resistant H pylori. Helicobacter. 2012;17(4):269–76.
Essa AS, Kramer JR, Graham DY, et al. Meta analysis: four drug, three antibiotic, non bismuth containing “concomitant therapy” versus triple therapy for Helicobacter pylori. Helicobacter. 2009;14:109–18.
Gisbert P, Calvet X. Update on non-bismuth quadruple (concomitant) therapy for eradication of Helicobacter pylori. Clin Exp Gastroenterol. 2012;5:23–34.
Greenberg ER, Anderson GL, Morgan DR, et al. 14 days triple, 5 days concomitant, and 10 days sequential therapies for Helicobacter pylori infection in seven Latin America sites: a randomised trial. Lancet. 2011;378:507–14.
Toros AB, Ince AT, Kesis B, et al. A new modified concomitant therapy for Helicobacter pylori eradication in Turkey. Helicobater. 2011;16:225–8.
Zhu R, Chen K, Zheng YY, et al. Meta-analysis of the efficacy of probiotics in Helicobacter pylori eradication therapy. W J Gastroenterol. 2014;20:18013–21.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethics Statement
These treatments were not prescribed as part of a clinical trial, and therefore, there was no need for study approval of the Hospital’s Ethics Committee.
Conflicts of Interest
The authors declare that they have no competing interests.
Financial Support
None.
Rights and permissions
About this article
Cite this article
Cerqueira, R.M., Correia, M.R., Vilar, H. et al. How Effective Is the Quadruple Concomitant Helicobacter Pylori Eradication Therapy for Obese Patients Undergoing Gastric Bypass Surgery?. OBES SURG 26, 1163–1166 (2016). https://doi.org/10.1007/s11695-015-1920-3
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11695-015-1920-3