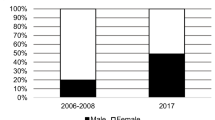
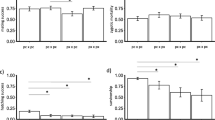
Abstract
Understanding the relative importance of various reproductive barriers to the early stages of speciation is an essential question in evolutionary biology. The closely related killifishes Fundulus heteroclitus and F. grandis occasionally hybridize in a small region in coastal Northeastern Florida showing that while barriers to reproduction exist, they are incomplete. The objective of this study was to elucidate barriers to reproduction between F. heteroclitus and F. grandis in the lab, as well as to quantify their strengths and relative contributions to reproductive isolation. Pre-zygotic (mating and fertilization) and post-zygotic (hatching) barriers were investigated by performing a variety of choice and no-choice laboratory mating experiments. Under no-choice conditions, barriers to mating had the greatest influence on hybrid production in F. grandis, whereas hatching barriers contributed to the majority of reproductive isolation in F. heteroclitus. Under choice conditions, however, pre-zygotic barriers had the greatest influence on hybrid production in both species. The total reproductive isolation that was observed in females of each species was stronger in F. heteroclitus than in F. grandis, and was nearly complete in F. heteroclitus females under choice conditions and was of moderate strength in F. grandis females. These results reveal an asymmetry in the potential gene flow between these two species, with F. grandis being more likely to hybridize than F. heteroclitus in the absence of environmental influences. No-choice backcrosses were also conducted and showed that at least some F1 hybrids are fertile. The observation that pre-zygotic barriers tend to be stronger than post-zygotic barriers in the early stages of speciation is consistent with similar studies in other organisms.


Similar content being viewed by others
References
Able, K., & Hagan, S. (2003). Impact of common reed, Phragmites australis, on essential fish habitat: Influence on reproduction, embryological development, and larval abundance of Mummichog (Fundulus heteroclitus). Estuaries, 26, 40–50.
Able, K., & Hata, D. (1984). Reproductive behavior in the Fundulus heteroclitus-F. grandis complex. Copeia, 1984, 820–825.
Berdan, E. L., & Fuller, R. C. (2012). A test for environmental effects on behavioral isolation in two species of killifish. Evolution, 66, 3224–3237.
Bernardi, G., & Powers, D. A. (1995). Phylogenetic relationships among nine species from the genus Fundulus (Cyprinodontiformes, Fundulidae) inferred from sequences of the cytochrome B gene. Copeia, 1995, 469–473.
Case, T., & Taper, M. (2000). Interspecific competition, environmental gradients, gene flow, and the coevolution of species’ borders. The American Naturalist, 155, 583–605.
Coyne, J. A., & Orr, A. H. (2004). Speciation. Sunderland: Sinauer Associates, Inc.
Coyne, J. A., & Orr, H. A. (1989). Patterns of speciation in Drosophila. Evolution, 43, 362–381.
Coyne, J. A., & Orr, H. A. (1998). The evolutionary genetics of speciation. Philosophical Transactions of the Royal Society of London B, 353, 287–305.
Dobzhansky, T. (1937). Genetics and the origin of species. New York: Columbia University Press.
Dobzhansky, T. (1940). Speciation as a stage in evolutionary divergence. The American Naturalist, 74, 312–321.
Ellis, W., & Bell, S. (2004). Conditional use of mangrove habitats by fishes: Depth as a due to avoid predators. Estuaries, 27, 966–976.
Elmer, K. R., Lehtonen, T. K., & Meyer, A. (2009). Color assortative mating contributes to sympatric divergence of neotropical cichlid fish. Evolution, 63, 2750–2757.
Foster, N. R. (1967). Trends in the evolution of reproductive behavior in killifishes. In: F. M. Bayer et al., (Eds.), Proceedings of the international conference on tropical oceanography (pp. 549–566). Miami, FL: University of Miami Institute of Marine Sciences.
Fricke, C., & Arnqvist, G. (2004). Conspecific sperm precedence in flour beetles. Animal Behaviour, 67, 729–732.
Galleher, S. N., Gilg, M. R., & Smith, K. J. (2010). Comparison of larval thermal maxima between Fundulus heteroclitus and F. grandis. Fish Physiology and Biochemistry, 36, 731–740.
Galleher, S. N., Gonzalez, I., Gilg, M. R., & Smith, K. J. (2009). Larvae and juvenile Fundulus heteroclitus abundance and distribution in Northeast Florida salt marshes. Southeastern Naturalist, 8, 495–502.
Geyer, L. B., & Palumbi, S. R. (2005). Conspecific sperm precedence in two species of tropical sea urchins. Evolution, 59, 97–105.
Gonzalez, I., Levin, M., Jermanus, S., Watson, B., & Gilg, M. R. (2009). Genetic assessment of species ranges in Fundulus heteroclitus and F. grandis in northeastern Florida salt marshes. Southeastern Naturalist, 8, 227–243.
Grady, J. M., Coykendall, D. K., Collette, B. B., & Quattro, J. M. (2001). Taxonomic diversity, origin, and conservation status of Bermuda killifishes (Fundulus) based on mitochondrial cytochrome b phylogenies. Conservation Genetics, 2, 41–52.
Gregorio, O., Berdan, E. L., Kozak, G. M., & Fuller, R. C. (2012). Reinforcement of male mate preference in sympatric killifish species Lucania goodie and Lucania parva. Behavioral Ecology and Sociobiology, 66, 1429–1436.
Hipperson, H., Dunning, L. T., Baker, W. J., Butlin, R. K., Hutton, I., Papadopulos, A. S. T., et al. (2016). Ecological speciation in sympatric palms: 2. Pre- and post-zygotic isolation. Journal of Evolutionary Biology, 29, 2143–2156.
Howard, D., Gregory, P., Chu, J., & Cain, M. (1998). Conspecific sperm precedence is an effective barrier to hybridization between closely related species. Evolution, 52, 511–516.
Howard, D. J. (1993). Reinforcement: Origin, dynamics and fate of an evolutionary hypothesis. In R. G. Harrison (Ed.), Hybrid zones and the evolutionary process (pp. 46–69). New York: Oxford University Press.
Hsiao, S., & Meier, A. (1989). Comparison of semilunar cycles of spawning activity in Fundulus grandis and F. heteroclitus held under constant laboratory conditions. Journal of Experimental Zoology, 252, 213–218.
Hsiao, S. M., Limesand, S. W., & Wallace, R. A. (1996). Semilunar follicular cycle of an intertidal fish: The Fundulus model. Biology of Reproduction, 54, 809–818.
Hsiao, S. -M., & Meier, A. H. (1986). Spawning cycles of the Gulf killifish, Fundulus grandis, in closed circulation systems. Journal of Experimental Zoology, 240, 105–112.
Jordan, D., & Evermann, B. (1898). The fishes of North and Middle America: A descriptive catalogue of the species of fish-like vertebrates found in the waters of North America, north of the Isthmus of Panama, 47th ed. US Government Printing Office, Washington, District of Columbia.
Kneib, R. (1986). The role of Fundulus heteroclitus in salt marsh trophic dynamics. American Zoologist, 26, 259–269.
Kneib, R. T. (1984). Patterns in the utilization of the intertidal salt marsh by larvae and juveniles of Fundulus heteroclitus (Linnaeus) and Fundulus luciae (Baird). Journal of Experimental Marine Biology and Ecology, 83, 41–51.
Lackey, A. C. R., & Boughman, J. W. (2017). Evolution of reproductive isolation in stickleback fish. Evolution, 71, 357–372.
Laurie, C. C. (1997). The weaker sex is heterogametic: 75 years of Haldane’s rule. Genetics, 147, 937–951.
Lowry, D. B., Modliszewski, J. L., Wright, K. M., Wu, C. A., & Willis, J. H. (2008). The strength and genetic basis of reproductive isolating barriers in flowering plants. Philosophical Transactions of the Royal Society of London B, 363, 3009–3021.
Ludlow, A. M., & Magurran, A. E. (2006). Gametic isolation in guppies (Poecilia reticulata). Proceedings of the Royal Society of London B, 273, 2477–2482.
Martin, M. D., & Mendelson, T. C. (2016). The accumulation of reproductive isolation in early stages of divergence supports a role for sexual selection. Journal of Evolutionary Biology, 29, 676–689.
Martin, M. D., & Mendelson, T. C. (2018). Hybrid sterility increases with genetic distance in snubnose darters (Percidae: Etheostoma). Environmental Biology of Fishes, 101, 215–221.
Martin, N. H., & Willis, J. H. (2007). Ecological divergence associated with mating system causes nearly complete reproductive isolation between sympatric Mimulus species. Evolution, 61, 68–82.
Martin, N. H., & Willis, J. H. (2010). Geographical variation in postzygotic isolation and its genetic basis within and between two Mimulus species. Philosophical Transactions of the Royal Society of London B, 365, 2469–2478.
Martín-Coello, J., Benavent-Corai, J., Roldan, E. R. S., & Gomendio, M. (2009). Sperm competition promotes asymmetries in reproductive barriers between closely related species. Evolution, 63, 613–623.
Matsubayashi, K. W., & Katakura, H. (2009). Contribution of multiple isolating barriers to reproductive isolation between a pair of phytophagous ladybird beetles. Evolution, 63, 2563–2580.
Mayr, E. (1940). Speciation phenomena in birds. The American Naturalist, 74, 249–278.
Mayr, E. (1942). Systematics and the origin of species, from the viewpoint of a zoologist. Cambridge: Harvard University Press.
Mayr, E. (2000). The biological species concept. In Q. D. Wheeler & R. Meier (Eds.), Species concepts and phylogenetic theory: A debate (pp. 17–29). New York: Columbia University Press.
Mendelson, T. C. (2003). Sexual isolation evolves faster than hybrid inviability in a diverse and sexually dimorphic genus of fish (Percidae: Etheostoma). Evolution, 57, 317–327.
Mendelson, T. C., Imhoff, V. E., & Venditti, J. J. (2007). The accumulation of reproductive barriers during speciation: Postmating barriers in two behaviorally isolated species of darters (Percidae: Etheostoma). Evolution, 61, 2596–2606.
Muller, H. J. (1939). Reversibility in evolution considered from the standpoint of genetics. Biological Reviews, 14, 261–280.
Muller, H. J. (1942). Isolating mechanisms, evolution, and temperature. Biology Symposium, 6, 71–125.
Naisbit, R. E., Jiggins, C. D., Linares, M., Salazar, C., & Mallet, J. (2002). Hybrid sterility, Haldane’s rule and speciation in Heliconius cydno and H. melpomene. Genetics, 161, 1517–1526.
Newman, H. (1907). Spawning behavior and sexual dimorphism in Fundulus heteroclitus and allied fish. Biological Bulletin, 12, 314–348.
Nosil, P. (2012). Ecological speciation. New York: Oxford University Press.
Orr, H. A. (1987). Genetics of male and female sterility in hybrids of Drosophila pseudoobscura and D. persimilis. Genetics, 116, 555–563.
Orti, G., Bell, M. A., Reimchen, T. E., & Meyer, A. (1994). Global survey of mitochondrial DNA sequences in the threespine Stickleback: Evidence for recent migrations. Evolution, 48, 608–622.
Ostevik, K. L., Andrew, R. L., Otto, S. P., & Rieseberg, L. H. (2016). Multiple reproductive barriers separate recently diverged sunflower ecotypes. Evolution, 70, 2322–2335.
Panhuis, T. M., Butlin, R., Zuk, M., & Tregenza, T. (2001). Sexual selection and speciation. Trends in Ecology & Evolution, 16, 364–371.
Pombi, M., Kengne, P., Gimonneau, G., Tene-Fossog, B., Ayala, D., Kamdem, C., et al. (2017). Dissecting functional components of reproductive isolation among closely related sympatric species of the Anopheles gambiae complex. Evolutionary Applications, 10, 1102–1120.
Presgraves, D. C. (2010). Darwin and the origin of interspecific genetic incompatibilities. The American Naturalist, 176, S45–S60.
Ramsey, J., Bradshaw, H. D., & Schemske, D. W. (2003). Components of reproductive isolation between the monkeyflowers Mimulus lewisii and M. cardinalis (Phrymaceae). Evolution, 57, 1520–1534.
Reynolds, J., & Gross, M. (1992). Female mate preference enhances offspring growth and reproduction in a fish, Poecilia reticulata. Proceedings of the Royal Society of London B, 250, 57–62.
Rundle, H. D., & Schluter, D. (1998). Reinforcement of stickleback mate preferences: Sympatry breeds contempt. Evolution, 52, 200–208.
Salzburger, W., Niederstätter, H., Brandstätter, A., Berger, B., Parson, W., Snoeks, J., et al. (2006). Colour-assortative mating among populations of Tropheus moorii, a cichlid fish from Lake Tanganyika, East Africa. Proceedings of the Royal Society of London B, 273, 257–66.
Schluter, D. (2001). Ecology and the origin of species. Trends in Ecology & Evolution, 16, 372–380.
Seehausen, O., van Alphen, J., & Witte, F. (1997). Cichlid fish diversity threatened by eutrophication that curbs sexual selection. Science, 277, 1808–1811.
Seehausen, O., & Van Alphen, J. J. M. (1998). The effect of male coloration on female mate choice in closely related Lake Victoria cichlids (Haplochromis nyererei complex). Behavioral Ecology and Sociobiology, 42, 1–8.
Sobel, J. M., & Chen, G. F. (2014). Unification of methods for estimating the strength of reproductive isolation. Evolution, 68, 1511–1522.
Sobel, J. M., Chen, G. F., Watt, L. R., & Schemske, D. W. (2010). The biology of speciation. Evolution, 64, 295–315.
Taylor, M., Leach, G., & DiMichele, L. (1979). Lunar Spawning Cycle in the Mummichog, Fundulus heteroclitus (Pisces: Cyprinodontidae). Copeia 1979, 291–297.
Vigueira, P. A., Schaefer, J. F., Duvernell, D. D., & Kreiser, B. R. (2007). Tests of reproductive isolation among species in the Fundulus notatus (Cyprinodontiformes: Fundulidae) species complex. Evolutionary Ecology, 22, 55–70.
Whitehead, A. (2010). The evolutionary radiation of diverse osmotolerant physiologies in killifish (Fundulus sp.). Evolution, 64, 2070–2085.
Williams, T. H., & Mendelson, T. C. (2014). Quantifying reproductive barriers in a sympatric pair of darter species. Evolutionary Biology, 41, 212–220.
Zeng, L. W., & Singh, R. S. (1993). The genetic basis of Haldane’s rule and the nature of asymmetric hybrid male sterility among Drosophila simulans, Drosophila mauritiana and Drosophila sechellia. Genetics, 134, 251–260.
Acknowledgements
We would like to thank the Lerner-Gray Memorial Fund of the American Museum of Natural History, the UNF Graduate School, and the UNF Coastal Biology Program for providing funding for this research. We thank Carlos Barbas, Jennifer Raabe, Victor Senf, Veronica Logue and Leigh Jordan for their help with collecting and caring for animals. Additionally, we would like to thank Dr. Kelly Smith and Dr. Eric Johnson for their comments on previous versions of this work and to Dr. Elena Buzaianu for statistical suggestions.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Barbas, R.E., Gilg, M.R. Quantification of Reproductive Isolating Barriers Between Two Naturally Hybridizing Killifish Species. Evol Biol 45, 425–436 (2018). https://doi.org/10.1007/s11692-018-9460-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11692-018-9460-0