Abstract
Purpose
The present research was taken to study the hospital-based incidence and clinico-pathological changes associated with naturally occurring trypanosomosis in dogs of Mizoram.
Methods
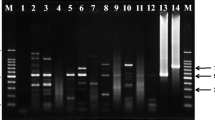
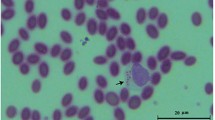
A 5-year prospective study on hospital-based incidence and clinico-pathological changes associated with naturally occurring trypanosomosis in dogs of Mizoram was carried out during the study period from April, 2015 to March, 2020. Trypanosoma evansi infection was confirmed by microscopic examination and polymerase chain reaction (PCR). Non-infected clinically healthy dogs (n = 6) served as control. Blood samples were collected to study the haemogram and serum samples were used for the evaluation of serum biochemical parameters and oxidant-antioxidant parameters.
Results
During the study period, an overall incidence of 0.25% was recorded for trypanosomosis in dogs. The most consistent clinical findings noticed were anorexia/inappetence, pyrexia, depression/lethargy, pale mucous membrane, dehydration and lymphadenomegaly. Anaemia, granulocytopenia, lymphocytosis and thrombocytopenia were the major findings noticed in trypanosomosis affected dogs. The profile of vital organ function revealed that the mean values of total protein, albumin and random blood glucose were significantly (P < 0.05) lower, whereas the mean values of alanine aminotransferase (ALT), aspartate aminotransferase (AST), alkaline phosphatase (ALP), total bilirubin, blood urea nitrogen (BUN) and creatinine were significantly (P < 0.05) higher in dogs affected with trypanosomosis. The mean value of lipid hydroperoxide (LPO) was significantly (P < 0.05) higher, whereas the mean values of glutathione (GSH), superoxide dismutase (SOD) and total antioxidant activity (TAOA) were significantly (P < 0.05) lower in trypanosomosis affected dogs. When total erythrocyte count (TEC) was correlated with LPO (r = − 0.631, P < 0.05), a negative correlation was found, while in case of GSH (r = 0.757, P < 0.05), SOD (r = 0.767, P < 0.05) and TAOA (r = 0.713, P < 0.05), it was positively correlated.
Conclusion
A negative correlation of TEC count with LPO, while a positive correlation with GSH, SOD and TAOA signify the role of oxidative stress in the pathogenesis of anaemia induced by T. evansi infection in dogs. The present study findings might be helpful to clinicians when treating clinical cases of this kind. Incorporation of organ protective drugs and antioxidants in the treatment schedule may result in better prognosis.





Similar content being viewed by others
Abbreviations
- ALT:
-
Alanine aminotransferase
- AST:
-
Aspartate aminotransferase
- ALP:
-
Alkaline phosphatase
- BUN:
-
Blood urea nitrogen
- CAT:
-
Catalase
- GPx:
-
Glutathione peroxidase
- GR:
-
Glutathine reductase
- GSH:
-
Glutathione
- LPO:
-
Lipid hydroperoxide
- SOD:
-
Superoxide dismutase
- TAOA:
-
Total antioxidant activity
References
Sumbria D, Singla LD, Sharma A, Moudgil AD, Bal MS (2014) Equine trypanosomosis in central and western Punjab: prevalence, haemato-biochemical response and associated risk factors. Acta Trop 138:44–50. https://doi.org/10.1016/j.actatropica.2014.06.003
Parashar R, Singla LD, Gupta M, Sharma SK (2018) Evaluation and correlation of oxidative stress and haemato-biochemical observations in horses with natural patent and latent trypanosomosis in Punjab state of India. Acta Parasitol 63(4):733–743. https://doi.org/10.1515/ap-2018-0087
Rjeibi MR, Hamida TB, Dalgatova Z, Mahjoub T, Rejeb A, Dridi W, Gharbi M (2015) First report of surra (Trypanosoma evansi infection) in a Tunisian dog. Parasite. https://doi.org/10.1051/parasite/2015004
Mulandane FC, Snyman LP, Brito DR, Bouyer J, Fafetine J, Van Den Abbeele J, Oosthuizen M, Delespaux V, Neves L (2020) Evaluation of the relative roles of the Tabanidae and Glossinidae in the transmission of trypanosomosis in drug resistance hotspots in Mozambique. Parasit Vectors 13:1–16. https://doi.org/10.1186/s13071-020-04087-1
Desquesnes M, Dargantes A, Lai DH, Lun ZR, Holzmuller P, Jittapalapong S (2013) Trypanosoma evansi and surra: a review and perspectives on transmission, epidemiology and control, impact, and zoonotic aspects. BioMed Res Int. https://doi.org/10.1155/2013/321237
Ramírez-Iglesias JR, Eleizalde MC, Reyna-Bello A, Mendoza M (2017) Molecular diagnosis of cattle trypanosomes in Venezuela: evidences of Trypanosoma evansi and Trypanosoma vivax infections. J Parasit Dis 41(2):450–458. https://doi.org/10.1007/s12639-016-0826-x
Götsch S, Leschnik M, Duscher G, Burgstaller JP, Wille-Piazzai W, Joachim A (2009) Ticks and haemoparasites of dogs from Praia, Cape Verde. Vet Parasitol 166(1–2):171–174. https://doi.org/10.1016/j.vetpar.2009.08.009
Abdullah INCI, Yazar S, Tuncbilek AS, Canhilal R, Doganay M, Aydin L, Aktas M, Vatansever Z, Ozdarendeli A, Ozbel Y, Yildirim A (2013) Vectors and vector-borne diseases in Turkey. Ankara Üniv Vet Fak Derg 60(4):281–296. https://doi.org/10.1501/vetfak_0000002593
Pamplona R, Costantini D (2011) Molecular and structural antioxidant defenses against oxidative stress in animals. Am J Physiol Regul Integr Comp Physiol 301(4):R843–R863. https://doi.org/10.1152/ajpregu.00034.2011
Hou X, Zhang J, Ahmad H, Zhang H, Xu Z, Wang T (2014) Evaluation of antioxidant activities of ampelopsin and its protective effect in lipopolysaccharide-induced oxidative stress piglets. PLoS ONE 9(9):e108314. https://doi.org/10.1371/journal.pone.0108314
Chethan GE, Garkhal J, De UK (2016) Disturbance of thyroid function in canine ehrlichiosis and babesiosis associated with oxidative stress. Comp Clin Path 25(5):987–992. https://doi.org/10.1007/s00580-016-2291-4
Gaykwad C, Garkhal J, Chethan GE, Nandi S, De UK (2018) Amelioration of oxidative stress using N-acetylcysteine in canine parvoviral enteritis. J Vet Pharmacol Ther 41(1):68–75. https://doi.org/10.1111/jvp.12434
Jain NC (1986) Schalm’s veterinary hematology, 4th edn. Lea & Febiger
Doumas BT (1975) Standards for total serum protein assays—a collaborative study. Clin Chem 21(8):1159–1166. https://doi.org/10.1093/clinchem/21.8.1159
Doumas BT, Watson WA, Biggs HG (1971) Albumin standards and the measurement of serum albumin with bromcresol green. Clin Chim Acta 31(1):87–96. https://doi.org/10.1016/0009-8981(71)90365-2
Reitman S, Frankel S (1957) A colorimetric method for the determination of serum glutamic oxalacetic and glutamic pyruvic transaminases. Am J Clin Pathol 28(1):56–63. https://doi.org/10.1093/ajcp/28.1.56
Kind PR, King EJ (1954) Estimation of plasma phosphatase by determination of hydrolysed phenol with amino-antipyrine. J Clin Pathol 7(4):322. https://doi.org/10.1136/jcp.7.4.322
Wybenga DR, Di Giorgio J, Pileggi VJ (1971) Manual and automated methods for urea nitrogen measurement in whole serum. Clin Chim Acta 17(9):891–895. https://doi.org/10.1093/clinchem/17.9.891
Toro C, Ackermann PG (1975) Practical clinical chemistry. Little Brown & Co., Boston, p 154
Jendrassik L, Grof P (1938) Colorimetric method of determination of bilirubin. Biochem Z 297:81–82
Trinder P (1969) Determination of glucose in blood using glucose oxidase with an alternative oxygen acceptor. Ann Clin Biochem 6(1):24–27. https://doi.org/10.1177/000456326900600108
Singh B, Kalra IS, Gupta MP, Nauriyal DC (1993) Trypanosoma evansi infection in dogs: seasonal prevalence and chemotherapy. Vet Parasitol 50(1–2):137–141. https://doi.org/10.1016/0304-4017(93)90014-E
Chowdhury P, Biswas U, Guha C, Jana PS (2005) Prevalence of canine trypanosomosis in and around Kolkata city. Indian Vet J 82(7):797–798
Prasad KL, Kondaiah PM, Rayulu VC, Srilatha C (2015) Prevalence of canine trypanosomiasis in certain areas of Andhra Pradesh. J Parasit Dis 39(2):238–240. https://doi.org/10.1007/s12639-013-0326-1
Ramesh P, Chowdary CSR, Chaitanya Y (2016) Diagnosis and treatment of canine trypanosomiasis—a case study. Int J Sci Environ Technol 5:3387–3393
Masake RA, Njuguna JT, Brown CC, Majiwa PAO (2002) The application of PCR–ELISA to the detection of Trypanosoma brucei and T. vivax infections in livestock. Vet Parasitol 105(3):179–189. https://doi.org/10.1016/S0304-4017(02)00020-1
Mugasa CM, Adams ER, Boer KR, Dyserinck HC, Büscher P, Schallig HD, Leeflang MM (2012) Diagnostic accuracy of molecular amplification tests for human African trypanosomiasis—systematic review. PLoS Negl Trop Dis 6(1):e1438. https://doi.org/10.1371/journal.pntd.0001438
Ahmed HA, Picozzi K, Welburn SC, MacLeod ET (2013) A comparative evaluation of PCR-based methods for species-specific determination of African animal trypanosomes in Ugandan cattle. Parasit Vectors 6(1):316. https://doi.org/10.1186/1756-3305-6-316
Nwoha RI, Anene BM (2011) Changes in packed cell volume and hemoglobin concentration in dogs with single and conjunct experimental infections of Trypanosoma brucei and Ancylostoma caninum. Philippine J Vet Anim Sci 37(2):151–158
Ogbu KI, Anene BM, Nweze NE, Danladi MA (2017) Trypanosomosis in dogs: a review. Int J Sci Appl Res 2(2):144–174
Reddy SB, Sivajothi S (2017) Corneal opacity due to trypanosomosis in buffaloes-need of topical medication. Open Access J Sci 1(6):155–156. https://doi.org/10.15406/oajs.2017.01.00031
Nwoha RIO (2013) A review on trypanosomosis in dogs and cats. Afr J Biotechnol 12(46):6432–6442. https://doi.org/10.5897/AJB2013.12093
Nongo NN, Tion MT, Apaa TT, Ogunro BN (2015) A case of canine trypanosomosis with epistaxis in a two-year old alsatian dog. J Agric Vet Sci 8(11):68–72. https://doi.org/10.9790/2380-081116872
Kjos SA, Snowden KF, Craig TM, Lewis B, Ronald N, Olson JK (2008) Distribution and characterization of canine Chagas disease in Texas. Vet Parasitol 152(3–4):249–256. https://doi.org/10.1016/j.vetpar.2007.12.021
Rossi SMS, Boada-Sucre AA, Simoes MT, Boher Y, Rodriguez P, Moreno M, de Ruiz ML, Marquez ML, Finol HJ, Sanoja C, Payares G (2017) Adhesion of Trypanosoma evansi to red blood cells (RBCs): Implications in the pathogenesis of anaemia and evasion of immune system. Diagn Pathol Open. https://doi.org/10.4172/2476-2024.1000122
Igbokwe IO (1994) Mechanisms of cellular injury in African trypanosomiasis. Vet Bull 64(7):611–620
Sivajothi S, Rayulu VC, Reddy BS (2015) Haematological and biochemical changes in experimental Trypanosoma evansi infection in rabbits. J Parasit Dis 39(2):216–220. https://doi.org/10.1007/s12639-013-0321-6
Allam L, Ogwu D, Agbede RI, Sackey AK (2011) Hematological and serum biochemical changes in gilts experimentally infected with Trypanosoma brucei. Vet Arh 81(5):597–609
Reddy BS, Kumari KN, Sivajothi S, Rayulu VC (2016) Haemato-biochemical and thyroxin status in Trypanosoma evansi infected dogs. J Parasit Dis 40(2):491–495. https://doi.org/10.1007/s12639-014-0531-6
Sulaiman FA, Adeyemi OS (2010) Changes in haematological indices and protein concentrations in Trypanosoma brucei infected rats treated with homidium chloride and diminazene aceturate. EXCLI J 9:39
Edozie Agu W, Nwachinemelu Egbuji A (2002) Urine albumin level in mice infected with Trypanosoma brucei. Vet Arh 72(2):101–108
Nwoha RIO, Eze IO, Anene BM (2013) Serum biochemical and liver enzymes changes in dogs with single and conjunct experimental infections of Trypanosoma brucei and Ancylostoma caninum. Afr J Biotechnol 12(6):618–624. https://doi.org/10.5897/AJB10.2594
Kadima KB, Gyang EO, Saror DI, Esievo KA (2000) Serum biochemical values of Trypanosoma vivax-infected cattle and the effects of lactose in saline infusion. Vet Arh 70(2):67–74
Baldissera MD, Souza CF, Grando TH, da Silva AS, Monteiro SG (2016) Involvement of oxidative stress, cholinergic and adenosinergic systems on renal damage caused by Trypanosoma evansi infection: relationship with lipid peroxidation. Microb Pathog 99:191–195. https://doi.org/10.1016/j.micpath.2016.08.028
Abenga JN, Anosa VO (2007) Serum biochemical changes in experimental gambian trypanosomosis. II. Assessing hepatic and renal dysfunction. Turk J Vet Anim Sci 31(5):293–296
Gunaseelan L, Senthil Kumar K, Selvaraj P, Kathiresan D (2009) Haemato biochemical changes in a case of canine trypanosomiasis. Tamilnadu J Vet Anim Sci 5(3):122–123
Cadioli FA, Marques LC, Machado RZ, Alessi AC, Aquino LPCT, Barnabé PA (2006) Experimental Trypanosoma evansi infection in donkeys: hematological, biochemical and histopathological changes. Arq Bras Med Vet Zootec 58(5):749–756. https://doi.org/10.1590/S0102-09352006000500008
Saleh MA, Al-Salahy MB, Sanousi SA (2009) Oxidative stress in blood of camels (Camelus dromedaries) naturally infected with Trypanosoma evansi. Vet Parasitol 162(3–4):192–199. https://doi.org/10.1016/j.vetpar.2009.03.035
Ranjithkumar M, Kamili NM, Saxena A, Dan A, Dey S, Raut SS (2011) Disturbance of oxidant/antioxidant equilibrium in horses naturally infected with Trypanosoma evansi. Vet Parasitol 80(3–4):349–353. https://doi.org/10.1016/j.vetpar.2011.03.029
Pandey V, Nigam R, Jaiswal AK, Sudan V, Singh RK, Yadav PK (2015) Haemato-biochemical and oxidative status of buffaloes naturally infected with Trypanosoma evansi. Vet Parasitol 212(3–4):118–122. https://doi.org/10.1016/j.vetpar.2015.07.025
Mishra RR, Senapati SK, Sahoo SC, Das MR, Sahoo G, Patra RC (2017) Trypanosomiasis induced oxidative stress and hemato-biochemical alteration in cattle. J Entomol Zool Stud 5(6):721–727
Fang YZ, Yang S, Wu G (2002) Free radicals, antioxidants, and nutrition. Nutrition 18(10):872–879. https://doi.org/10.1016/S0899-9007(02)00916-4
Ozden S, Catalgol B, Gezginci-Oktayoglu S, Arda-Pirincci P, Bolkent S, Alpertunga B (2009) Methiocarb-induced oxidative damage following subacute exposure and the protective effects of vitamin E and taurine in rats. Food Chem Toxicol 47(7):1676–1684. https://doi.org/10.1016/j.fct.2009.04.018
Akanji MA, Adeyemi OS, Oguntoye SO, Sulyman F (2009) Psidium guajava extract reduces trypanosomosis associated lipid peroxidation and raises glutathione concentrations in infected animals. EXCLI J 8:148–154. https://doi.org/10.17877/DE290R-8903
I Eze J, Ajanwachukwu N, C Animoke P, O Onoja S, N Anosa G, U Eze U (2016) Immune response, anaemia and oxidative stress in Trypanosoma brucei brucei infected rats fed vitamin E supplemented diet. Antiinfect Agents 14(1):28–37. https://doi.org/10.2174/221135251401160302122153
Harvey JW (1997) The erythrocyte: physiology, metabolism, and biochemical disorders. In: Clinical biochemistry of domestic animals. Academic Press, pp 157–203
Clemens MR, Waller HD (1987) Lipid peroxidation in erythrocytes. Chem Phys Lipids 45(2–4):251–268. https://doi.org/10.1016/0009-3084(87)90068-5
De UK, Dey S, Banerjee PS, Sahoo M (2012) Correlations among Anaplasma marginale parasitemia and markers of oxidative stress in crossbred calves. Trop Anim Health Pro 44(3):385–388. https://doi.org/10.1007/s11250-011-9938-6
Acknowledgements
The authors are highly thankful to the Vice Chancellor, Central Agricultural University and Dean, College of Veterinary Sciences and Animal Husbandry, Selesih, Aizawl, for providing the requisite facilities to carry out the present research work.
Author information
Authors and Affiliations
Contributions
KS, CGE, PR, SKB, VJ, HP, SKB, NB, and DD: conducting the research and investigation process, specifically performing the experiments, or data/evidence collection. KS, CGE, and NT: preparation, creation and/or presentation of the published work, specifically writing the initial draft (including substantive translation)
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical statement
This article does not contain any studies with human or animal participants performed by any of the authors. The article reports clinical cases presented to TVCC, College of Veterinary Sciences and Animal husbandry, Central Agricultural University, Selesih, Aizawl, Mizoram. All protocols followed were as per the guidelines from the standard textbooks in Veterinary Medicine and were in compliance with ethical standards of the institute.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Sarma, K., Eregowda, C.G., Roychoudhury, P. et al. A 5-Year Prospective Study on Incidence and Clinico-pathological Changes Associated with Naturally Occurring Trypanosomosis in Dogs of Mizoram, India. Acta Parasit. 67, 61–71 (2022). https://doi.org/10.1007/s11686-021-00425-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11686-021-00425-0