Abstract
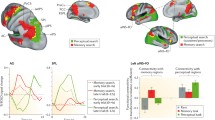
The rostral prefrontal cortex (rPFC) is crucial in prospective memory (PM) behavior. Several functional magnetic resonance imaging studies showed that its medial (mrPFC) and lateral (lrPFC) portions dissociate during PM tasks. In light of the Multiprocess theory (McDaniel and Einstein 2000), here we tested whether the two portions of the rPFC are dissociable by modulating strategic vs. spontaneous processes during a PM task. We investigated these two processes by means of a 2 × 2 experimental design in which focal vs. non-focal conditions were modulated by varying the conjoint nature of the ongoing task (i.e., lexical decision vs. syllable matching) and the PM cue (words vs. syllables). Using the two portions of the rPFC as regions of interest, we found an effect of the non-focal condition in the lrPFC and, conversely, an effect of the focal condition in the mrPFC. In the whole-brain analysis we found an effect of the non-focal condition in the bilateral intraparietal sulcus, the bilateral middle frontal gyrus, the supplementary motor areas and the vermis of the cerebellum, whereas we found an effect of the focal condition in the ventromedial PFC. Overall, our results show that different brain regions are involved when multiple processes underlying PM behavior are modulated.




Similar content being viewed by others
Notes
Here we provided a description of the localizer task, for more detailed information the reader is referred to the original paper (Barban et al. 2014). The localizer task comprised 7 ONG and 7 PM blocks of 10 trials each. PM and ONG blocks were presented randomly but, in order to prevent contamination, each PM block was followed by a block of the ONG task using geometrical shapes instead of letters (wash-out block). ONG blocks started with a green X (5 s); participants executed the ONG task alone. PM blocks started with a red fixation X (5 s) with above two letters as PM cues. For each ONG trial, participants were administered a single target letter immediately below a fixation cross for 1 s. After its offset, a horizontal four-letters string appeared for 1.5 s. The string always contained the target letter. Participants were required to press a first button with their forefinger if the target letter appeared in one of the two left positions of the string and to press a second button with their middle finger if the target letter appeared in one of the two right positions of the string. The PM task consisted in pressing a third button with the ring finger whenever a PM cue letter was present in a letter string of the PM task.
Delta RT of PM trials reflected the differences between the time occurring for executing the PM action (i.e., the second pressing on the button-pad) and the time occurring for executing the ONG task during the BAS blocks.
References
Barban, F., Carlesimo, G. A., Macaluso, E., Caltagirone, C., & Costa, A. (2014). Functional interplay between stimulus-oriented and stimulus-independent attending during a prospective memory task. Neuropsychologia, 53, 203–212.
Beck, S. M., Ruge, H., Walser, M., & Goschke, T. (2014). The functional neuroanatomy of spontaneous retrieval and strategic monitoring of delayed intentions. Neuropsychologia, 52, 37–50.
Benoit, R. G., Gilbert, S. J., Frith, C. D., & Burgess, P. W. (2012). Rostral prefrontal cortex and the focus of attention in prospective memory. Cerebral Cortex, 22, 1876–1886.
Bisiacchi, P. S., Schiff, S., Ciccola, A., & Kliegel, M. (2009). The role of dual-task and task-switch in prospective memory: Behavioural data and neural correlates. Neuropsychologia, 47, 1362–1373.
Brandimonte, M., Einstein, G. O., & McDaniel, M. A. (Eds.). (1996). Prospective memory: Theory and application. Mahwah: Erlbaum.
Brewer, G. A., Knight, J. B., Marsh, R. L., & Unsworth, N. (2010). Individual differences in event-based prospective memory: Evidence for multiple processes supporting cue detection. Memory & Cognition, 38, 304–311.
Buckner, R. L., Krienen, F. M., Castellanos, A., Diaz, J. C., & Yeo, B. T. T. (2011). The organization of the human cerebellum estimated by intrinsic functional connectivity. Journal of Neurophysiology, 106, 2322–2345.
Burgess, P. W., Quayle, A., & Frith, C. D. (2001). Brain regions involved in prospective memory as determined by positron emission tomography. Neuropsychologia, 39, 545–555.
Burgess, P. W., Scott, S. K., & Frith, C. D. (2003). The role of the rostral frontal cortex (area 10) in prospective memory: A lateral versus medial dissociation. Neuropsychologia, 41, 906–918.
Burgess, P. W., Simons, J. S., Dumontheil, I., & Gilbert, S. J. (2005). The gateway hypothesis of rostral prefrontal cortex (area 10) function. In J. Duncan, L. Phillips, & P. Mcleod (Eds.), Measuring the mind: Speed, control and age (pp. 217–248). Oxford: Oxford University Press.
Burgess, P. W., Gonen-Yaacovi, G., & Volle, E. (2011). Functional neuroimaging studies of prospective memory: What have we learnt so far? Neuropsychologia, 49, 2246–2257.
Ciaramelli, E., Grady, C. L., & Moscovitch, M. (2008). Top-down and bottom-up attention to memory: A hypothesis (AtoM) on the role of the posterior parietal cortex in memory retrieval. Neuropsychologia, 46, 1828–1851.
Cohen, A.-L., Jaudas, A., & Gollwitzer, P. M. (2008). Number of cues influence the cost of remembering to remember. Memory & Cognition, 36, 149–156.
Cona, G., Bisiacchi, P. S., & Moscovitch, M. (2014). The effects of focal and non-focalcues on the neural correlates of prospective memory: Insights from ERPs. Cerebral Cortex, 24, 2630–2646.
Cona, G., Scarpazza, C., Sartori, G., Moscovitch, M., & Bisiacchi, P. S. (2015). Neural bases of prospective memory: A meta-analysis and the "attention to delayed intention" (AtoDI) model. Neuroscience and Biobehavioral Reviews, 52, 21–37.
Cona, G., Bisiacchi, P. S., Sartori, G., & Scarpazza, C. (2016). Effects of cue focality on the neural mechanisms of prospective memory: A meta-analysis of neuroimaging studies. Scientific Reports, 6, 25983.
Costa, A., Peppe, A., Serafini, F., Zabberoni, S., Barban, F., Caltagirone, C., & Carlesimo, G. A. (2014). Prospective memory performance of patients with Parkinson's disease depends on shifting aptitude: Evidence from cognitive rehabilitation. Journal of the International Neuropsychological Society, 20, 717–726.
Costa, A., Peppe, A., Zabberoni, S., Serafini, F., Barban, F., Scalici, F., Caltagirone, C., & Carlesimo, G. A. (2015). Prospective memory performance in individuals with Parkinson's disease who have mild cognitive impairment. Neuropsychology, 29, 782–791.
Dehaene, S., Jobert, A., Naccache, L., Ciuciu, P., Poline, J. B., Le Bihan, D., & Cohen, L. (2004). Letter binding and invariant recognition of masked words: Behavioral and neuroimaging evidence. Psychological Science, 15, 307–313.
Einstein, G. O., & McDaniel, M. A. (2005). Prospective memory: Multiple retrieval processes. Current Directions in Psychological Science, 14, 286–290.
Einstein, G. O., & McDaniel, M. A. (2010). Prospective memory and what costs do not reveal about retrieval processes: A commentary on Smith, Hunt, McVay, and McConnell (2007). Journal of experimental psychology. Learning. Memory and Cognition, 36, 1082–1088 discussion 1089–1095.
Einstein, G. O., Holland, L. J., McDaniel, M. A., & Guynn, M. J. (1992). Age-related deficits in prospective memory: The influence of task complexity. Psychology and Aging, 7, 471–478.
Einstein, G. O., McDaniel, M. A., Thomas, R., Mayfield, S., Shank, H., Morrisette, N., & Breneiser, J. (2005). Multiple processes in prospective memory retrieval: Factors determining monitoring versus spontaneous retrieval. Journal of Experimental Psychology. General, 134, 327–342.
Gilbert, S. J., Gollwitzer, P. M., Cohen, A. L., Burgess, P. W., & Oettingen, G. (2009). Separable brain systems supporting cued versus self-initiated realization of delayed intentions journal of experimental psychology. Learning, Memory and Cognition, 35, 905–915.
Gilbert, S. J., Gonen-Yaacovi, G., Benoit, R. G., Volle, E., & Burgess, P. W. (2010). Distinct functional connectivity associated with lateral versus medial rostral prefrontal cortex: A meta-analysis. Neuroimage, 53, 1359–1367.
Gordon, B. A., Shelton, J. T., Bugg, J. M., McDaniel, M. A., & Head, D. (2011). Structural correlates of prospective memory. Neuropsychologia, 49, 3795–3800.
Greicius, M. D., & Menon, V. (2004). Default-mode activity during a passive sensory task: Uncoupled from deactivation but impacting activation. Journal of Cognitive Neuroscience, 16, 1484–1492.
Harrison, T. L., & Einstein, G. O. (2010). Prospective memory: Are preparatory attentional processes necessary for a single focal cue? Memory & Cognition, 38, 860–867.
Hefer, C., Cohen, A. L., Jaudas, A., & Dreisbach, G. (2017). The flexible engagement of monitoring processes in non-focal and focal prospective memory tasks with salient cues. Acta Psychologica, 179, 4253.
Henseler, I., Krüger, S., Dechent, P., & Gruber, O. (2011). A gateway system in rostral PFC? Evidence from biasing attention to perceptual information and internal representations. Neuroimage, 56, 1666–1676.
Hicks, J. L., Franks, B. A., & Spitler, S. N. (2017). Prior task experience and comparable stimulus exposure nullify focal and non-focal prospective memory retrieval differences. The Quarterly Journal of Experimental Psychology, 70, 1997–2006.
Landsiedel, J., & Gilbert, S. J. (2015). Creating external reminders for delayed intentions: Dissociable influence on “task-positive” and “task-negative” brain networks. Neuroimage, 104, 231–240.
Lau, H. C., Rogers, R. D., Haggard, P., & Passingham, R. E. (2004). Attention to intention. Science, 303, 1208–1210.
McDaniel, M. A., & Einstein, G. O. (2000). Strategic and automatic processes in prospective memory retrieval: A multiprocess framework. Applied Cognitive Psychology, 14, S127–S144.
McDaniel, M. A., Guynn, M. J., Einstein, G. O., & Breneiser, J. (2004). Cue-focused and reflexive-associative processes in prospective memory retrieval. Journal of Experimental Psychology: Learning Memory Cognition, 30, 605–614.
McDaniel, M. A., Lamontagne, P., Beck, S. M., Scullin, M. K., & Braver, T. S. (2013). Dissociable neural routes to successful prospective memory. Psychological Science, 24, 1791–1800.
McDaniel, M. A., Umanath, S., Einstein, G. O., & Waldum, E. R. (2015). Dual pathways to prospective remembering. Frontiers in Human Neuroscience, 9, 232.
Meeks, J. T., & Marsh, R. L. (2010). Implementation intentions about non-focal event-based prospective memory tasks. Psychological Research, 74, 82–89.
Miall, R. C., & Robertson, E. M. (2006). Functional imaging: Is the resting brain resting? Current Biology, 16, 998–1000.
Moscovitch, M. (1994). Memory and working with memory: Evaluation of a component process model and comparison with other models. In D. L. Schacter & E. Tulving (Eds.), Memory systems (pp. 269–310). Cambridge: MIT Press.
Okuda, J., Gilbert, S. J., Burgess, P. W., Frith, C. D., & Simons, J. S. (2011). Looking to the future: Automatic regulation of attention between current performance and future plans. Neuropsychologia, 49, 2258–2271.
Oldfield, R. C. (1971). The assessment and analysis of handedness: The Edinburgh inventory. Neuropsychologia, 9, 97–113.
O'Reilly, J. X., Beckmann, C. F., Tomassini, V., Ramnani, N., & Johansen-Berg, H. (2010). Distinct and overlapping functional zones in the cerebellum defined by resting state functional connectivity. Cerebral Cortex, 20, 953–965.
Penny, W. D., & Holmes, A. P. (2004). Random effects analysis. In R. S. J. Frackowiak, K. J. Friston, C. Frith, R. J. Dolan, C. J. Price, S. Zeki, J. Ashburner, & W. D. Penny (Eds.), Human brain function (2nd ed., pp. 843–851). San Diego: Elsevier.
Raichle, M. E., MacLeod, A. M., Snyder, A. Z., Powers, W. J., Gusnard, D. A., & Shulman, G. L. (2001). A default mode of brain function. Proceedings of the National Academy of Sciences of the United States of America, 98, 676–682.
Reynolds, J. R., West, R., & Braver, T. (2009). Distinct neural circuits support transient and sustained processes in prospective memory and working memory. Cerebral Cortex, 19, 1208–1221.
Scullin, M. K., McDaniel, M. A., Shelton, J. T., & Lee, J. H. (2010). Focal/non-focal cue effects in prospective memory: Monitoring difficulty or different retrieval processes? Journal of experimental psychology. Learning, Memory and Cognition, 36, 736–749.
Simons, J. S., Scholvinck, M. L., Gilbert, S. J., Frith, C. D., & Burgess, P. W. (2006). Differential components of prospective memory? Evidence from fMRI. Neuropsychologia, 44, 1388–1397.
Smith, R. E. (2003). The cost of remembering to remember in event-based prospective memory: Investigating the capacity demands of delayed intention performance. Journal of experimental psychology. Learning. Memory and Cognition, 29, 347–361.
Smith, R. E. (2010). What Costs Do Reveal and Moving Beyond the Cost Debate: Reply to Einstein and McDaniel (in press). Journal of Experimental Psychology. Learning, Memory and Cognition, 36, 1089–1095.
Smith, R. E., Hunt, R. R., McVay, J. C., & McConnell, M. D. (2007). The cost of event-based prospective memory: Salient target events. Journal of experimental psychology. Learning. Memory and Cognition, 33, 734–746.
Strickland, L., Heathcote, A., Remington, R. W., & Loft, S. (2017). Accumulating evidence about what prospective memory costs actually reveal. Journal of experimental psychology. Learning. Memory and Cognition, 43, 1616–1629.
Umeda, S., Kurosaki, Y., Terasawa, Y., Motoichiro, K., & Miyahara, Y. (2011). Deficits in prospective memory following damage to the prefrontal cortex. Neuropsychologia, 49, 2178–2184.
Uretzky, S., & Gilboa, A. (2010). Knowing your lines but missing your cue: Rostral prefrontal lesions impair prospective memory cue detection, but not action-intention superiority. Journal of Cognitive Neuroscience, 22, 2745–2757.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
No author declared a conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the standards of the Ethics Committee of IRCCS Santa Lucia Foundation (Rome, IT) committee and with the 1964 Helsinki declaration and its later amendments.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Barban, F., Scalici, F., Carlesimo, G.A. et al. Medio-lateral functional dissociation of the rostral prefrontal cortex with focal/non-focal cues during a prospective memory task. Brain Imaging and Behavior 14, 1175–1186 (2020). https://doi.org/10.1007/s11682-019-00063-1
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11682-019-00063-1