Abstract
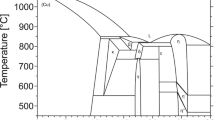
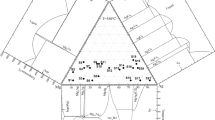
Sixteen alloys were prepared to determine the isothermal sections of the Ag–Cu–Nb system at 500, 600 and 700 °C using x-ray diffraction (XRD) and scanning electron microscopy with energy dispersive x-ray spectroscopy (SEM/EDS). Based on the thermodynamic descriptions of constitutive binary systems as well as the experimental phase equilibria data obtained from the present work and literatures, the Ag–Cu–M (M = Nb, Fe, Pb) ternary systems were thermodynamically evaluated by the CALPHAD (CALculation of PHAse Diagrams) method. The solution phases were described by the substitutional solution model and a set of self-consistent thermodynamic parameters was finally obtained for each of the ternary systems. The calculated isothermal sections are in good agreement with most of the reliable experimental data.









Similar content being viewed by others
References
J.W. Choi, S. Kwon, Y. Park, K. Kang, and M.H. Lee, In Silico High-Throughput Screening of Ag-Based Electrocatalysts for Anion-Exchange Membrane Fuel Cells, J. Phys. Chem. Lett., 2021, 12, p 5660–5667.
T.A. Kassam, N.H. Babu, N. Ludford, S. Yan, and A. Howkins, Secondary Phase Interaction at Interfaces of High-Strength Brazed Joints Made Using Liquid Phase Sintered Alumina Ceramics and Ag–Cu–Ti Braze Alloys, Sci. Rep., 2018, 8, p 3352.
X.X. Wang, J. Peng, D.T. Cui, P. Xue, H. Li, A.M. Hu, and G.Y. Sun, Research and Application of Silver-Based Brazing Alloys in Manufacturing Industries: A Review, Mater. Rep., 2018, 32, p 1477–1485.
Y. Zhang, B. Hu, G. Zeng, S.H. Liu, Y. Du, and H.Q. Yin, Experimental Investigation Thermodynamic Modeling and Solidified Microstructure of the Cu–Ti–Nb termary System, Calphad, 2022, 76, p 102395.
Y. Zhang, B. Hu, B.F. Li, M. Zhang, Q.P. Wang, and Y. Du, Experimental Investigation CALPHAD Modeling of the Cu–Cr–Si Termary System, Calphad, 2021, 74, p 102324.
Y. Zhang, B. Hu, Y.S. Shi, S.H. Liu, Y. Du, J.Q. Hu, Thermodynamic Modeling and Solidified Microstructure of the Ag–Sn–Zr Ternary System, T. Nonferr. Matel. Soc. (in press), 2022
R. Kieffer, S. Windisch, and H. Nowotny, Niobium-Tantalum, Metall., 1963, 17, p 669.
M.R. Baren, The Ag–Nb (silver-niobium) System, Bull. Alloy Phase Diagr., 1989, 10, p 640.
M. Aria, M. Tanaka, K.S. Goto, and M. Someno, Activity and Diffusivity Measurements of Copper in Gamma and Delta Fe by Equilibration between Solid Fe and liquid Ag, Metall. Mater. Trans. A, 1981, 12, p 497–504.
G. Petzow, and G. Effenberg, Silver-Copper-Lead, J. Mater. Res., 1988, 2, p 1–13.
E. Lüder, Das Dreistoff-System, Silber-Kupfer-Eisen, Z. Metallkd., 1924, 16, p 61–62.
T. Klassen, U. Herr, and R.S. Averback, Mechanisms of Phase Formation during Milling in the Ternary Immiscible Ag–Cu–Fesystem, Mat. Res. Soc. Symp. Proc., 1995, 400, p 25–30.
H.Y. Gao, J. Wang, D. Shu, and B.D. Sun, Effect of Ag on the Microstructure and Properties of Cu–Fe in Situ Composites, Scr. Mater., 2005, 53, p 1105–1109.
Y.F. Wang, H.Y. Gao, J. Wang, Y.F. Han, Y.B. Dai, and B.D. Sun, First-Principles Calculation of Ag Addition on the Diffusion Mechanisms of Cu–Fe Alloys, Solid State Commun., 2014, 183, p 60–63.
W.L. Jiang, C. Zhang, N. Xu, B. Yang, B.Q. Xu, D.C. Liu, and H.W. Yang, Experimental Investigation and Modelling of Phase Equilibria for the Ag–Cu–Pb System in Vacuum Distillation, Fluid Phase Equilib., 2016, 417, p 19–24.
Z.K. Liu, and Y. Wang, Computational thermodynamics of materials. Cambridge University Press, New York, 2016.
H. Lukas, S.G. Fries, and B. Sundman, Computational thermodynamics: the Calphad method. Cambridge University Press, New York, 2007.
A.T. Dinsdale, SGTE Data for Pure Elements, Calphad, 1991, 15, p 317–425.
X.C. He, H. Wang, H.S. Liu, and Z.P. Jin, Thermodynamic Description of the Cu–Ag–Zr System, Calphad, 2006, 30, p 367–374.
J.Q. Zhou, B. Hu, Y. Jiang, C.L. Qiu, Y. Du, and H.Q. Yin, Thermodynamic Modeling of the Ag–X (X=B, Fe, Sm, Pu) Binary System, J. Phase Equilib. Diffus., 2020, 41, p 257–268.
B.Z. Lee, C.S. Oh, and D.N. Lee, A thermodynamic Evaluation of the Ag–Pb–Sb System, J. Alloys Compd., 1994, 215, p 293–301.
G.I. Terekhov, and L.N. Aleksandrova, The Copper–Niobium Phase Diagram, Izv. Akad. Nauk. SSSR. Met., 1984, 4, p 210–213.
I. Ansara, and A. Jansson, System Cu-Fe, in COST 507, Vol. 2. I. Ansara, A.T. Dinsdale, and M.H. Rand, Eds., European Communities, Brussels, 1998, p 165–167
B. Onderka, and L. Zabdyr, A New Critical Assessment of the Copper-Lead System, Scand. J. Metall., 2001, 30, p 320–323.
O. Redlich, and A.T. Kister, Thermodynamics of Nonelectrolyte Solutions x–y–t Relations in a Binary System, Ind. Chem. Phys., 1948, 40, p 341–345.
Y.M. Muggianu, M. Gambino, and J.P. Bros, Enthalpies of Formation of Liquid Alloys Bismuth-Gallium-Tin at 723k - Choice of an Analytical Representation of Integral and Partial Thermodynamic Functions of Mixing for this Ternary-System, J. Chim. Phys. Phys. -Chim. Biol., 1975, 72, p 83–88.
B. Sundman, and J. Agren, A Regular Solution Model for Phases with Several Components and Sublattices Suitable for Computer Applications, J. Phys. Chem. Solids, 1981, 42, p 297–301.
B. Sundman, B. Jansson, and J.O. Andersson, The Thermo-Calc Databank System, Calphad, 1985, 9, p 153–190.
Acknowledgments
The work support from the Major Science and Technology Project of Precious Metal Materials Genetic Engineering in Yunnan Province (No. 202002AB080001-1), the National Natural Science Foundation of China (No. 52071002) and the National Natural Science Foundation of Anhui Province (No. 2008085QE200) are greatly acknowledged.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declared that there are no conflicts of interest for this work.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Zhang, Y., Hu, B., Yin, H. et al. Phase Equilibria of the Ag–Cu–M (M = Nb, Fe, Pb) Systems. J. Phase Equilib. Diffus. 43, 533–546 (2022). https://doi.org/10.1007/s11669-022-00998-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11669-022-00998-x