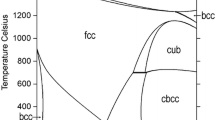
Abstract
Thermodynamic descriptions of the Fe-Nb-P system and its binary sub-system, Fe-Nb, are developed in the frame of a new Fe-X-P (X = Al, Cr, Cu, Mn, Mo, Nb, Ni, Si, Ti) database. The thermodynamic parameters of the binary sub-systems, Fe-P and Nb-P, are taken from earlier assessments and those of the Fe-Nb-P and Fe-Nb systems are optimized in this study using literature experimental thermodynamic and phase equilibrium data. The solution phases of the system (i.e. bcc_A2, fcc_A1, Liquid) are described with the substitutional solution model. The phase Mu (Fe7Nb6) has been modeled as (Fe,Nb)7(Nb)6, while Fe2P and FeNbP, have been modeled both as (Fe,Nb)2(P). All other phases (Fe2Nb, Fe3P, FeP, Nb3P, Nb7P4, NbP, NbP2, FeNb2P, FeNb4P, (P)) are treated as stoichiometric. The assessment of this system needs improvement but more experimental information has to be available.















Similar content being viewed by others
References
J. Miettinen and G. Vassilev, Thermodynamic Description of Ternary Fe-X-P Systems. Part 1: Fe-Cr-P, J. Phase Equil. Diff., 2014, 35, p 458-468. doi:10.1007/s11669-014-0314-x
J. Miettinen and G. Vassilev, Thermodynamic Description of Ternary Fe-X-P Systems. Part 2: Fe-Cu-P, J. Phase Equil. Diff., 2014, 35, p 469-475. doi:10.1007/s11669-014-0315-9
J. Miettinen and G. Vassilev, Thermodynamic Description of Ternary Fe-X-P Systems. Part 3: Fe-Mn-P. Submitted to J. Phase Equil. Diff.
J. Miettinen, S. Louhenkilpi, H. Kytönen, and J. Laine, IDS: Thermodynamic-Kinetic-Empirical Tool for Modeling of Solidification, Microstructure and Material Properties, Math. Comput. Simulat., 2010, 80, p 1536-1550
W. Huang, Thermodynamic Evaluation of the Fe–Nb–C System, Z. Metallkd., 1990, 81, p 397-404
C. Toffolon and C. Servant, Thermodynamic Assessment of the Fe-Nb System, CALPHAD., 2000, 24, p 97-112
B.-J. Lee, Thermodynamic Assessment of the Fe-Nb-Ti-C-N System, Metall. Mater. Trans. A., 2001, 32, p 2423-2439
A. Khvan and B. Hallstedt, Thermodynamic Description of the Fe–Mn–Nb–C System, CALPHAD., 2012, 39, p 62-69
S. Liu, B. Hallstedt, D. Music, and Y. Du, Ab initio Calculations and Thermodynamic Modeling for the Fe-Mn-Nb System, CALPHAD, 2012, 38, p 43-58
T. Tokunaga, N. Hanaya, H. Ohtani, and M. Hasebe, Thermodynamic Analysis of the Fe-Nb-P Ternary Phase Diagram, ISIJ Int., 2009, 49, p 947-956
J.-H. Shim, C.-S. Oh, and D.N. Lee, Thermodynamic Properties and Calculation of Phase Diagram of the Fe-P System, J. Korean Inst. Met. Mater., 1996, 34, p 385-393
H. Lukas, S. Fries, and B. Sundman, Computational Thermodynamics: The Calphad Method, Cambridge University Press, UK, 2007
V. Raghavan, Phase Diagrams for Ternary Iron Alloys, Part 3, Calcutta, Indian Institute of Metals, 1988
Z. Bejarano, S. Gama, C. Ribeiro, G. Effenberg, and C. Santos, On the Existence of the Fe2Nb3 Phase in the Fe-Nb System, Z. Metallkd., 1991, 82, p 615-620
Z. Bejarano, S. Gama, C. Ribeiro, G. Effenberg, and C. Santos, The Iron-Niobium Phase Diagram, Z. Metallkd., 1993, 84, p 160-164
N. Voronov, Splavy jeleza s niobiem (Alloys of Iron with Niobium). Izv. Akad. Nauk SSSR, Khim., 1937, 1, p 1369-1379.
H. Eggers and W. Peter, The Iron-Niobium Phase Diagram, Mitt. Kaiser-Wilhelm Inst. Eisenforsch., 1938, 20, p 199-203
R. Genders and R. Harrison, Niobium-Iron Alloys, J. Iron Steel Inst., 1939, 140, p 29-37
J. Goldschmidt, The Constitution of the Iron-Niobium-Silicon System, J. Iron Steel Inst., 1960, 194, p 169-180
W. Gibson, R. Lee, and W. Hume-Rothery, Liquidus-Solidus Relations in Iron-Rich Iron-Niobium and Iron-Molybdenum Alloys, J. Iron and Steel Inst., 1961, 198, p 64-66
A. Ferrier, E. Ubelacker, and E. Wachtel, Study of the Fe-Nb Phase Diagram Between 0 and 12 at% Nb from 1200 to 1535 °C, C.R. Acad. Sci. Paris., 1964, 258, p 5424-5427
E. Abrahamson and S. Lopata, The Lattice Parameters and Solubility Limits of Alpha Iron as Affected by Some Binary Transition-Element Additions, Trans. AIME., 1966, 236, p 76-87
A. Raman, Structural Study of Niobium-Iron Alloys, Proc. Indian Acad. Sci., 1967, A65, p 256-264
W. Fischer, K. Lorenz, H. Fabritius, and D. Schlegel, Examination of the α/γ Transformation in Very Pure Binary Alloys of Iron with Molybdenum, Vanadium, Tungsten, Niobium, Tantalum, Zirconium and Cobalt, Arch. Eisenhuttenwes., 1970, 41, p 489-498
S. Voss, M. Palm, F. Stein, and D. Raabe, Phase Equilibria in the Fe-Nb System, JPED., 2011, 32, p 97-104
E. Ichise and K. Horikawa, Thermodynamic Study of Fe-Ta and Fe-Nb Alloys by Means of Knudsen Cell Mass Spectrometry, ISIJ Int., 1989, 29, p 843-851
Y. Iguchi, S. Nosomi, K. Saito, and T. Fuwa, Experimental Determinations of Phase Equilibria in the Fe-V-X (X:Nb, Ti) Systems and Development of Thermodynamic Database of Microalloyed Steel and its Applications, Tetsu-to-Hagane., 1982, 68, p 633-640
V. Sudavtsova, V. Kurach, and G. Batalin, Термодинамические свойства жидких двойных сплавов Fe-(Y, Zr, Nb, Mo) (Thermodynamic Properties of the Liquid Binary Alloys Fe-(Y,Zr, Nb, Mo)), Izv. Akad. Nauk SSSR Metally., 1987, 3, p 60-61
L. Schaefers, M. Rösner-Kuhn, J. Qin, and M. Frohberg, Mixing Enthalpy and Heat Content Measurements of Liquid Binary Iron-Niobium Alloys, Steel Res., 1995, 66, p 183-187
V. Drobyshev and T. Rezukhina, X-ray Investigation of the Nb–Fe System and the Determination of the Thermodynamic Properties of the Compound NbFe2, Russ. Metall., 1966, 2, p 85-89
G. Barbi, High-Temperature Electrochemical Determination of the Themodynamic Stability of the Iron-Rich, Iron-Niobium Intermetallic Phase, Z. Naturforsch., 1969, 24A, p 1580-1585
A. Niessen, F. De Boer, R. Boom, P. De Châtel, W. Mattens, and A. Miedema, Model Predictions for the Enthalpy of Formation of Transition Metal Alloys II, CALPHAD, 1983, 7, p 51-70
R. Vogel and W. Bleichroth, Das Dreistoffsystem Eisen-Phosphor-Niob (The Ternary System Fe-P-Nb), Arch. Eisenhüttenwes., 1962, 33, p 195-210, in German
H. Kaneko, T. Nishizawa, K. Tamaki, and A. Tanifuji, Solubility of Phosphorus in α and γ Iron, Nippon Kinzoku Gakkai-Si., 1965, 29, p 166-170, in Japanese
K. Yamada and E. Kato, Effect of Dilute Concentrations of Si, Al, Ti, V, Cr Co, Ni, Nb and Mo on the Activity Coefficient of P in Liquid Iron, Trans. Iron Steel Inst. Jpn., 1983, 23, p 51-55
S. Ban-Ya, N. Maruyama, and Y. Kawase, Effects of Ti, V, Cr, Mn Co, Ni, Cu, Nb, Mo and W on the Activity of Phosphorus in Liquid Iron, J. Iron Steel Inst. Jpn., 1984, 70, p 65-72, in Japanese
J.-O. Andersson, T. Helander, L. Höglund, P. Shi, and B. Sundman, Thermo-Calc & DICTRA. Computational Tools for Materials Science, CALPHAD, 2002, 26, p 273-312
R. Elliot and W. Rostoker, The Occurrence of Laves-Type Phases Among Transition Elements, Trans. ASM., 1958, 50, p 617-633
K. Bewilogua, R. Reichelt, K. Wetzig, and H. Wittig, Emissionselektronenmikroskopische Untersuchungen am Zweistoffsystem Niob-Eisen (Studies of the Binary System Fe-Nb by Emission Electron Microscope), Cryst. Res. Technol., 1972, 7, p 601-609
A. Dinsdale, SGTE Data for Pure Elements, CALPHAD, 1991, 15, p 317-425
Acknowledgment
Financial support of the Finnish Funding Agency for Technology and Innovation (TEKES) is gratefully acknowledged by Dr J. Miettinen. The research was carried out as part of the Finnish Metals and Engineering Competence Cluster (FIMECC)’s SIMP program.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Miettinen, J., Vassilev, G. Thermodynamic Description of Ternary Fe-X-P systems. Part 5: Fe-Nb-P. J. Phase Equilib. Diffus. 36, 68–77 (2015). https://doi.org/10.1007/s11669-014-0351-5
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11669-014-0351-5