Abstract
The main goal of this study was to achieve poreless titanium alloys with nanocrystalline structure. To this end, the influence of Ta, Mg and Zn content on the properties of Ti alloys was investigated. At first, nanocrystalline powders of TixTayMg and TixTayZn, where x = (30 and 40 wt.%) and y = (3 and 5 wt.%), were prepared using the mechanical alloying process at room temperature. Then, hot-pressing at 750 °C under vacuum was applied for 10 s to consolidate powders. The structure, microstructure, chemical composition, mechanical properties, corrosion resistance, wettability and MTT assay have been investigated. Alloys with Mg were allowed to undergo a shorter mechanical alloying process, achieve greater grain refinement after consolidation and improve mechanical properties. In all cases with increasing amounts of additives in titanium, more Ti-β phase was available. Furthermore, with increasing elements content, hot-pressed alloys consisting of Mg tended to have a more hydrophobic surface. According to the MTT test, all new alloys show non-toxic properties. Among all alloys tested in this study, Ti40Ta5Mg had the most interesting properties for biomedical applications, the highest content of Ti-β phase (81.3%), lowest porosity (0.07%), lowest Young modulus (102.1 GPa) and the lowest surface free energy (38.45 mN/m).
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
Titanium alloys, after stainless steel, are among the most commonly used metals for metallic biomaterials. Thanks to its properties such as excellent biocompatibility, self-passivation, high corrosion resistance, and great mechanical strength-to-density ratio compared to other metallic alloys, titanium is the most widely used metal in clinical practice (Ref 1, 2). Moreover, in a group of materials used for permanent implants, it shows the lowest Young modulus and density; however, the elastic modulus is still relatively high (65-110 GPa) in comparison with human bone (10-30 GPa) (Ref 1, 2). Such a large gap between these two values can induce stress shielding phenomenon and finally bone resorption, which in the worst scenario may lead to implant failure or bone fracture (Ref 1). Furthermore, commonly used biomedical titanium alloy (Ti6Al4V) contain toxic elements—vanadium and aluminum. In the implantation area, inflammation can appear, and extended exposure to these elements may cause Alzheimer’s disease, other neuropathies or osteomalacia (a neurodegenerative disease) (Ref 3, 4). Therefore, in order to prevent negative effects of the implant, the alloy applied must contain additives that are inert or well-tolerated by the human body. Tantalum, magnesium and zinc are alloying additives which, due to the intrinsic properties and properties of titanium alloys containing them, are potential candidates for applications in hard tissue implants (Ref 5,6,7,8,9,10,11).
Tantalum is inert, self-passivating and chemically resistant β-stabilizer, which may be used as an additive element to the titanium alloys (Ref 12). When compared to tantalum, titanium exhibits a notably reduced density (Ti: 4.51 g/cm3 versus Ta: 16.69 g/cm3), lower Young’s modulus (Ti: 103 GPa versus Ta: 200 GPa), and lower melting temperature (Ti: 1668 °C versus Ta: 3017 °C) (Ref 13, 14). Above-mentioned physical factors, such as differences in density and temperature, cause problems in manufacturing in a conventional way, leading to alternated distribution of Ti- and Ta-rich zones (Ref 14). Thus, it is suggested to use powder metallurgy to prepare alloys from these elements. With increasing concentrations of Ta, strength increases (Ref 7, 13,14,15,16,17,18,19) and corrosion resistance improves in comparison with pure titanium, due to the higher stability of Ta2O5 over TiO2 (Ref 7, 14, 19,20,21,22,23,24,25). However, Young modulus change is not linear, as it depends on factors such as chemical composition, phase composition, microstructure or processing conditions (Ref 7, 13,14,15,16,17,18,19, 21). In Bahador et al. (Ref 18), they achieved the lowest Young modulus of 61 GPa for Ti35Ta alloy using powder metallurgy, spark plasma sintering and hot forging. Zhou et al. (Ref 16) report the lowest modulus for Ti30Ta and Ti40Ta alloys, 69 and 81 GPa, respectively, using a tri-arc furnace and melting each alloy more than ten times to attain chemical homogenization. Liu et al. (Ref 14) focused on powder metallurgy. For Ti30Ta, they managed to get a significantly higher tensile strength (1056 MPa) over the ingot version (587 MPa) (Ref 16). In order to further decrease the elastic modulus (E), it is suggested to fabricate a porous structure. It is possible to reach E values close enough to a bone value. Soro et al. (Ref 26) used a mechanically alloyed Ti25Ta powders for selective laser melting (SLM) to get materials of 42% porosity and 23 GPa elastic modulus, which is favored for its interaction with tissue. With the introduction of porosity, it is possible to improve the osseointegration with the bone through the formation and growth of new tissue and vascularization process (Ref 20, 27). Some of the Ti-Ta alloys exhibit a super-elastic (Ref 18) or memory shape effect (Ref 28). Both porous structure and applying of Ti and Ta oxides improve osseointegration (Ref 14). Furthermore, tantalum oxide exhibits antibacterial properties, decreasing the risk of infection around the implantation area (Ref 5, 29).
Magnesium naturally occurs in the human body as a bioelement. Mg alloys have found their way into biomedical materials due to their low modulus (43 GPa), low density (1.74 g/cm3), excellent biocompatibility, and low cost (Ref 10, 30). Moreover, Mg are easily biodegraded in body fluids due to the presence of chloride ions (Ref 30), so they can be used to manufacture temporary implants and components, such as bolts, screws or stents (Ref 10, 31). However, there are several problems related to the connection of magnesium with titanium. First of all, different physical properties of Mg and Ti do not allow to combine these two elements conventionally. The melting point of Mg is 650 °C, which is over 1000 °C lower than Ti. Moreover, the Mg boiling point is as low as 1090 °C, causing the element to evaporate while alloying in a liquid state. Second, the solubility of Mg in Ti-α phase is very low, slightly better in Ti-β phase, but using non-equilibrium processes, such as mechanical alloying, it is possible to extend the limit (Ref 32,33,34,35,36). Ward-Close et al. (Ref 33) report the possible solubility of Mg in Ti up to 20% by using vapor quenching. However, Mg does not create any stable compound either with Ti or Ta (Ref 37). Thus, a potential liquid-phase sintering process may occur, allowing rapid densification (Ref 38). Third, even though these alloys have great biocompatibility, it is hard to control their degradation rate, which can cause unexpected loosening of the implant. Despite that magnesium can be used as an space holder for the dealloying process to manufacture foams or porous structures (Ref 39).
Zinc is not a commonly used additive in titanium-based alloys. Pure zinc is denser than titanium (7.1 g/cm3), but its melting point is lower than magnesium (420 °C), and its elastic modulus is close to pure titanium (105 GPa). The boiling point is even lower than magnesium (906 °C), which causes the same manufacturing problems as with magnesium. Zinc is not soluble in Ti-α, but it is soluble up to 31 at.% in Ti-β (Ref 40). It may form some phases with Ti, but due to the low melting point of the element, a liquid phase during the sintering process is also expected to happen, as with magnesium (Ref 38). There are not many reports of the Ti-Zn alloys (Ref 41), as well as Ta-Zn probably due to zinc properties. Zinc and its alloys are used as temporary implants, such as stents or bone fixation tools (Ref 9, 42,43,44,45,46). They are biodegradable in the same way as magnesium alloys but at a slower rate. It usually takes three months for a magnesium stent to biodegrade completely (Ref 47), whereas zinc ones degrade partially after 20 months (Ref 42). Reports claim that zinc implants are well-tolerated in the human body (Ref 42, 43). Formation of thick corrosion oxide layer after long-term implantation suppresses inflammation, contributing to bio-integration but is not preventing progressive corrosion (Ref 42). As a trace metal, zinc also has multiple biological functions, from enzymatic catalysis to taking an essential role in cellular neuronal systems (Ref 48). Zinc can improve cardiac functions (Ref 8) and play an important role in immune functions (Ref 49).
The main goal of this study is to investigate the effect of different content (30, 40%) of Ta with small amounts (3, 5%) of Mg and Zn in titanium on the microstructure, mechanical properties, corrosion resistance wetting properties as well as metabolic activity and determination of the effect of individual additives on the final properties of alloys. The combination of alloying elements and the formation of the optimal structure of alloys requires the use of an appropriate technological processes. Non-equilibrium processes, such as sequential mechanical alloying and hot-pressing, seem to be promising, making it possible to obtain a homogeneous, ultrafine or nanocrystalline structure with better properties than conventional microcrystalline alloys (Ref 50, 51).
2 Materials and Methods
The ternary Ti-Ta-Mg and Ti-Ta-Zn alloys were prepared by mechanical alloying (MA) under argon atmosphere using a SPEX 8000 Mixer Mill with hardened steel vial and balls. Based on our preliminary studies, we selected the following chemical compositions: TixTayMg and TixTayZn, where x = 30 and 40 wt.% and y = 3 and 5 wt.%. The ball to powder weight ratio was 10:1 (total ball weight 50 g, 5 items, powder weight 5 g). Initial powders were: Ti (purity 99.5%), Ta (99.98%) and Mg (99.8%), all 325 mesh from Alfa Aesar and Zn (99%) 30-100 mesh from Sigma-Aldrich (Fig. 1). The Ti, Ta and Mg powders were irregular in shape, wherein Ta had sponge-like particles. Zn presented a granular and elongated shape. The weight of the starting powder was measured by precision balance (0.001 g repeatability; Radwag, Poland). The milling process was stopped every 2 h to investigate the structural and powder morphology changes, which was performed by XRD and SEM. The milling time was designed to achieve a fine Ti-β structure, and it was 14, 20, and 24 h, depending on the chemical composition of the alloys. All the powders handling was done in Unilab glove box (MBraun, Germany) providing high-purity inert atmosphere (argon) of controlled oxygen and moisture content (< 2 ppm). The milling container is filled with argon from the glove box.
As-prepared powder alloys were consolidated by hot pressing combined with high-frequency induction heating (Elbit FI-W, Poland). Hot pressing was carried out at 750 °C with an acting pressure of 50 MPa in a graphite die in a vacuum condition (< 50 Pa). The heating rate was set to 75 °C/s for 10 s, holding time 10 s, then the hot-pressed alloys were slowly cooled down to the room temperature with the furnace (graphite die). The high heating rate and low final consolidation temperature were selected on the basis of our preliminary study in order to avoid excessive grain growth and ensuring an acceptable level of material strength. The compacts were 8 mm in diameter and about 5 mm high. The samples for all tests were mechanically ground on the SiC papers up to 1200 grit and polished in diamond suspension, to obtain a mirror surface finish.
The structure and phase analysis were investigated using Empyrean XRD (Panalytical, Netherlands) with CuKα radiation, λ = 1.54178 Å. Measurements were conducted at set parameters: voltage 45 kV, anode current 40 mA, and scanning range 30°-80° 2θ. Crystallographic data were analyzed using Rietveld refinement by HighScore Plus software. Pseudo-Voigt profile function for peaks profile analysis was used. The phase composition of the examined samples was calculated by a standardless x-ray diffraction method using ICDD- PDF-4 + database. The microstructure was characterized by Mira 3 FEG SEM (Tescan, Czech Rep.) equipped with In-Beam SE, BSE and EDS UltimMax 65 (Oxford Instruments, England) detector. The porosity level was examined by taking several pictures of random areas on the samples with GX51 (Olympus, Japan) optical microscope on polished samples. The taken pictures were analyzed using built in Stream Start software phase analyzing module.
The corrosion tests of the materials were performed in Ringer’s solution (BTL Zaklad Enzymow i Peptonow, Poland): NaCl 9 g/L, KCl 0.42 g/L, CaCl2 0.48 g/L, NaHCO3 0.2 g/L, using Solartron 1285 (Solartron Analytical, England) potentiostat working in a potentiodynamic mode with a scan rate of 0.5 mV/s, in the established potential range of from − 0.7 to + 2.5 V versus open-circuit potential (OCP). Before starting the potentiodynamic tests, the OCP was stabilized for 3600 s. The electrolyte temperature was maintained at 37 °C, which correspond to human body temperature. The counter and reference electrodes were made of graphite and Pt, respectively.
The wetting and surface free energy measurements were carried out on the Drop Shape Analyzer DSA25 (KRÜSS-Scientific, Germany). A distilled water and glycerol drop of 2.0 µL volume was used for analysis. The surface free energy was determined by measuring the contact angle (CA) according to the Owens, Wendt, Rabel, and Kaelble (OWRK) model by ADVANCE software. Other parameters used were as follows: dosing speed 0.2 mL/min, measuring time 5 s, probing frequency for multiple measurements 10 fps, Young Laplace CA fitting method, and ambient temperature 22.0 °C.
Mechanical measurements were made with a Picodentor HM500 (Helmut Fisher, Germany) nanoindenter. According to ISO 14577-1 (Ref 52) standard, the following parameters were measured: HM-Martens Hardness, HV-Vickers Hardness, and EIT-indentation modulus. For each sample, five indentations were made at the force of 300 mN for 20 s.
Normal human osteoblast (CC-2538) (NHost) and human periodontal ligament fibroblasts (CC-7049) (HPLF) (Lonza Group Ltd., Basel, Switzerland) were used for the in vitro cytocompatibility tests. The tests were obtained under static conditions. The cells were cultured at a concentration of 5000 cells/well in 1 mL of culture medium on each sample at 37 °C in a 5% CO2 incubator for 24, 72, and 96 h. The proliferation of the cells in the conditioned mediums was expressed as a percentage of the value of relative viability of the cells (RVC) of the reference medium. The reference medium was prepared using pure bulk microcrystalline titanium samples, and it was represented by 100%. All samples were sterilized by autoclaving at 120 °C for 15 min and separately located in 24-well microplates. The statistical significance was analyzed using Kruskal–Wallis one-way analysis of variance on ranks with multiply repetition option SigmaStat 3.5 (Systat Software Inc., Erkrath, Germany) with Mann–Whitney U test. The significance level was p value < 0.05.
3 Results and Discussion
Mechanical alloying and hot pressing were used to obtain ternary Ti-Ta-(Mg or Zn) alloys. The effect of tantalum, and magnesium, or zinc on the properties of titanium alloys and the possibility of acquiring nano-grained, non-porous, bulk Ti alloys were studied.
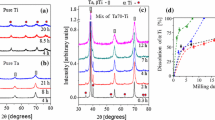
In this paper, the preparation of Ti-Ta-Mg and Ti-Ta-Zn alloys is closely examined. Eight compositions were prepared in total. Four of them were Ti-alloys containing 30 and 40 wt.% of Ta and 3 and 5 wt.% of Mg: Ti30Ta3Mg, Ti30Ta5Mg, Ti40Ta3Mg and Ti40Ta5Mg (Fig. 2), and four Ti-alloys with the same amount of Ta, but with Zn instead: Ti30Ta3Zn, Ti30Ta5Zn, Ti40Ta3Zn and Ti40Ta5Zn (Fig. 3). Figure 2 and 3 present phase changes (Fig. 2a, 3a) and powder morphology for all compositions at the second hour (Fig. 2b, 3b) and the end of MA (Fig. 2c, 3c). After two hours of milling, there are still visible peaks from Ti and Ta for all the alloys. Images from the BSE detector show that the powders after MA are simply mixtures of the powders (Fig. 2b, 3b). BSE contrast that indicates the presence of heavy (bright) and light (dark) elements in certain areas is not visible for final powder agglomerates. With increasing milling time, peaks from individual components were decreasing in intensity. As a result, new peaks corresponding to Ti-β phase have started to appear. Synthesis lasted as long as the elements were finely distributed in the titanium, and Ti-β peaks were only visible. Depending on the composition, MA required different milling times. With an increasing amount of Ta, the MA process of tested alloys required less time. The Ti30Ta(3 and 5)Mg group needed 20 h of synthesis, with 24 h for Ti30Ta(3 and 5)Zn alloys. In contrast, for all alloys consisting of 40 wt.% of Ta, 14 h of synthesis was sufficient. Alloys with 30 wt.% of Ta and the addition of Mg required 4 h less synthesis time than alloys with Zn instead. This effect is not visible in alloys with 40 wt.% of Ta content. At the end of the MA process, BSE images show homogenously distributed powders mixture, which is strongly deformed and in lamellar form, typical for MA process (Fig. 2c, 3c). Use of SPEX mill allowed to achieve much greater distribution of elements in titanium than in planetary ball mill, for example (Ref 12). Moreover in the same work, after 20 h of mechanical alloying of Ti50Ta, both Ti-β and Ti-α were evident in XRD spectra with calculated grain size of accordingly 58 and 43 nm.
Powder crystallite size and strain were calculated using the Williamson–Hall (W–H) formula and visualized in Fig. 4. Table 1 shows summarized results for W–H analysis of the powders after mechanical alloying. The estimated crystallite size varies from 16 up to 28 nm for almost every analyzed powders. These powders have a negative strain value, which can correspond to the presence of vacancies inside the crystal structure (Ref 53,54,55). Only Ti40Ta5Zn posed twice as big crystallites (43 nm), with a positive strain value that can reassemble to expanded grain with substitutional atoms. Possibly with elongating the MA process for this specific alloy, the grain size could be further decreased. The results can be compared to those measured with transmission electron microscopy (TEM), as presented in studies (Ref 56, 57), but only for calculated crystallites below 100 nm.
Figure 5 shows the XRD spectra of hot-pressed alloys. Every alloy exhibited a dual-phase structure composed of Ti-α and Ti-β. With increasing concentrations of Ta and Mg/Zn in the titanium alloy, more dominant peaks of Ti-β are visible, while peaks intensity of Ti-α decreases. A difference of only 2 wt.% of Mg/Zn significantly impacts the phase composition of alloys, favorably stabilizing Ti-β, i.e., phase composition of Ti40Ta3Mg (72.3Ti-β%/27.7%Ti-α) and Ti40Ta5Mg (81.3Ti-β%/18.7%Ti-α) as well for alloys with Zn as the additive, Ti40Ta3Zn (68.3Ti-β%/31.7%Ti-α) and Ti40Ta5Zn (73.6%Ti-β/26.4%Ti-α). Although Mg and Zn are relatively neutral stabilizers (Ref 34, 37, 40), a more substantial stabilizing effect is visible in alloys with Mg instead of Zn.
The XRD crystallographic data of bulk samples have been calculated using Rietveld refinement and are also presented in Table 1. In almost every alloy examined, Ti-β was the leading phase. Only in Ti30Ta3Zn Ti-α was dominating over Ti-β phase (49.0%Ti-β/51.0%Ti-α). The highest concentration of Ti-β in each Mg and Zn group has been found in Ti40Ta5Mg alloy (81.3%Ti-β/18.7%Ti-α), and Ti40Ta5Zn alloy (73.6%Ti-β/26.4%Ti-α), respectively. In every case, magnesium additive gives a better stabilization effect of Ti-β over zinc comparing the same wt.% compositional groups. Calculated lattice parameters of Ti-α and Ti-β are higher or close to reference values for all alloys (Ref 58, 59). On the one hand, the presence of bigger Ta and Mg atoms in Ti solid solution may cause the expansion of the crystal lattice and volume (Ref 16, 60, 61). On the other hand, Zn atomic radius is smaller in comparison with Ti, and it can lead to contraction of the crystal lattice and a decrease in lattice parameters (Ref 60).
With chosen hot-pressing parameters, it was possible to produce bulk samples with minimal porosity up to about 1% (Fig. 6). Compositions containing magnesium had higher porosity than specimens with Zn. For comparison, the highest porosity for Mg-containing alloys was 1.09 and 0.52% for Ti40Ta3Mg and Ti30Ta3Mg, respectively, whereas for Zn-containing alloys, it was 0.14 and 0.12% for Ti30Ta3Zn and Ti30Ta5Zn, respectively. Since atomically, there are more magnesium atoms than zinc atoms in the same mass value, there is a higher chance of porosity in the hot-pressed alloys. However, low melting elements are good candidates for introducing porous structures into a material by thermal dealloying (Ref 39). The diffusional mass transport occurs in the potential coexisting liquid phase throughout the sintering process. This effect allows for rapid densification at relatively low temperatures (Ref 38). In addition, it is possible to acquire a bulk sample made of binary Ti-Mo alloy at a relatively low temperature of 800 °C, without the additional low melting element (Ref 55). Nonetheless, it is worth mentioning that longer sintering times have been used.
SEM images allowed to analyze grain size and elements (powders) distribution in the samples. In BSE scanning mode, bright and dim areas were observed (Fig. 7). Bright areas are larger in enriched Ta content than shady ones. Due to a difference in diffusion coefficients between titanium and tantalum (Ref 14) in the selected consolidated temperature, all studied alloys have a gradient structure. It could be said that the bright area represents cubic (Ti-β) when the dark region hexagonal (Ti-α) structure.
Comparing BSE pictures of the samples (Fig. 7), it is possible to notice an effect of magnesium on general microstructure. Elements in those alloys (Fig. 8a, b, c, d) are more evenly distributed than in alloys containing zinc (Fig. 8e, f, g, h). Thus, magnesium seems to compensate for diffusion coefficient differences. Possibly, it causes grain refinement to be better in alloys with magnesium.
Even though BSE pictures (Fig. 8) show differences in elemental distribution, EDS mapping does not demonstrate it. Examples of two alloys with the highest amount of additives are presented in Fig. 8. The (a–d) are related to Ti40Ta5Mg and (e–h) to Ti40Ta5Zn. EDS maps indicate a fair distribution of elements through entire samples. Because of the EDS nature, information about the composition is also gathered from the area beneath the current image.
The hot-pressing processes were performed at temperatures above the melting point of magnesium and zinc. Therefore, there was a chance that it may affect the final composition and cause porosity in the structure. Theoretical compositions, as well as those calculated from EDS analysis, are presented in Table 2. For most compositions, the values measured are similar to those assumed at the beginning. Alloys consisting of the most additives were not far from the theoretical values, i.e., Ti40Ta5Mg has a true composition Ti42.7Ta4.1Mg and Ti40Ta5Zn has a true composition Ti41.3Ta5.4Zn. However, as magnesium belongs to the light group of elements, it may slightly lower the precision of the actual measurements. Therefore, for higher accuracy, a different measurement technique is suggested.
Nanoindentation (load–displacement) curves are shown in Fig. 9, and the results are collected in Table 3. With the increasing amount of additives in titanium, the elastic modulus and the hardness of alloys decreases. The lowest Young modulus exhibit Ti40Ta5Mg (102.1 GPa), while the highest modulus values are for alloys with the least amount of additional elements. The difference in Ti30Ta3Mg (131.6 GPa) and Ti30Ta5Mg (142.3 GPa) might come from the presence of porosity in the sample. Thus, for porous structure, the deviation in Young modulus is more noticeable (Ref 39). Comparing alloys with the highest amount of additives Ti40Ta5Mg/Zn (accordingly 102.1 and 130.2 GPa), the zinc-containing alloy shows an observable rise in the modulus. It might result from a bigger grain size after mechanical alloying, which is about twice as big as the others (Table 1). Additionally, the measurements might come from the regions rich in Ta content (Fig. 7), as pointed out earlier, due to the low diffusion coefficient between Ti and Ta, causing a gain in elastic modulus. Moreover, other factors can cause a shift in E value, i.e., solid solution strengthening with increasing Ta content (Ref 55). Similar strengthening effect was visible in hot-pressed bulk Ti-Mo alloys (Ref 55), where molybdenum content was in between 18 and 52 wt.%. The measured Young modulus was for extremes contents 124 and 158 GPa, respectively. Moreover, a Young modulus could be potentially affected by a phenomenon of complete or partial wetting grain boundaries (GBs) by second solid phase(Ref 62,63,64,65). In this case, solid phase of Ti-β, is being wetted by the second solid phase of Ti-α. The wetting effect appears to be greater in alloys containing Mg, where grain boundaries are rounded and well-refined (Fig. 7a, b, c, d). For alloys with Zn as the additive, GBs are angular and prominent; thus, the wetting effect is not as evident as for alloys with Mg (Fig. 7e, f, g, h). It can be assumed that the more Ti-α grain boundaries are refined, the higher decrease in the module is (Ref 62). Nevertheless, confirming this effect would require a more in-depth investigation to be performed.
Polarization curves of the alloys tested in the Ringer solution are presented in Fig. 10. Interestingly, in both cases, Ti30Ta5Mg/Zn exhibited the lowest corrosion potential (Ecorr = − 0.36/− 0.44 V for Mg/Zn), as well as corrosion current (Icorr = 0.054251/0.030321 µA/cm2) among each analyzed group of materials with magnesium and zinc as the additive to Ti-Ta alloys (Table 4). Higher concentration of additives, noticeably changed the corrosion behavior of the alloy in the solution. Introducing more tantalum to the alloy caused the rise both of current density and corrosion potential. Recent studies suggest that alloys with ≥ 30 wt.% of Ta have greater resistance than pure titanium (Ref 66). Considering all aspects, such as corrosion resistance, density and cost, the authors propose Ti alloys with 40 wt.% of Ta as a favorable choice. It is worth mentioning that both Mg and Zn are more vulnerable to corrosion than Ti and Ta. Therefore, as the additives, they can compromise the general corrosion resistance (Ref 9, 45, 47, 61, 67). The existence of different phases in the alloy can also lead to the formation of local unfavorable galvanic coupling (Ref 9, 45, 67).
Wetting properties of the alloys, such as estimated surface free energy, disperse and polar components, together with contact angle (CA) of water and glycerol drops (Fig. 11, 12), are given in Table 5. Surface free energy (SFE) decreases with the increasing amount of additives for magnesium samples (51.00, 44.47, 43.23, 38.45 mN/m, respectively). This relationship does not apply for alloys with zinc, and the value remains steady for every case except for Ti40Ta5Zn. For the first three alloys, it stays almost steady (42.21, 42.75, 41.55 mN/m); the richest in content alloy exhibit SFE of 46.34 mN/m. As it was the case for Young modulus, the grain size might also play a role here. In addition, regions rich in Ta might impact general wettability, because Ta is more hydrophilic than Ti (Ref 68). Influencing wettability it is possible to induce faster bone growth and improve the healing process. For hard tissue growth, it is more desirable that implant poses a more hydrophilic surface (Ref 69). Although, there can still be found studies suggesting that intermediate hydrophilic/hydrophobic properties are better for cell adhesion (Ref 70, 71). From the other point of view, a higher contact angle prevents biofilm formation and may act as an antibacterial agent (Ref 72). All the hot-pressed alloys with polished surface investigated show moderately hydrophilic properties, with contact angle significantly below 90°. For future work, it would be interesting to improve CA, for example, by applying surface treatment, such as anodic oxidation (Ref 73).
Cytotoxic activity was analyzed using the MTT assay. The RVC results obtained for normal human osteoblast and human periodontal ligament fibroblasts are shown in Fig. 13(a) and (b), respectively. The results revealed a significant effect of time as well as material composition on cell viability. After 24 h, the RVC for all the materials tested was lower (oscillating in the range 59-69%) than that of the reference sample, which is often caused by longer adaptation time of cells to the richer chemical composition of new materials medium. Cytotoxicity decreased over time. Such results allow forming a conclusion that all tested materials show non-toxic effects on the cells. However, there are significant differences between Zn- and Mg-doped alloys in all evaluation points. After 96 h, all materials showed the highest viability. Ti-Ta alloys with Zn shows better cell viability compared to Ti-Ta-Mg alloys. Alloys containing zinc showed 1.5-1.6 and alloys with magnesium 1.0-1.3 times better RVC (NHost). The results for fibroblasts showed the same trend, however, lower RVC. It was 1.35-1.4 and 0.9-1.05 times higher than the reference condition for Zn- and Mg-doped alloys, respectively. The alloy with the highest cell viability was Ti30Ta3Zn (NHost), Ti30Ta5Zn, Ti40Ta5Zn (HPLF) and Ti30Ta5Mg (NHost) Ti40Ta5Mg (HPLF) showed the lowest.
Many different methods are available to produce nanocrystalline and/or fine-grained structures (Ref 74, 75). Powder metallurgy is one of the most favorable choices due to the possibility of producing a significant amount of material. Results show that it provides homogenous, nanocrystalline Ti-β alloy powder as a precursor (Fig. 2, 3). The studied alloys crystal structure strongly depended on the composition. Not only the addition of Ta, but also Mg and Zn affected the phase content, favoring stabilization of Ti-β phase with the more substantial effect of Mg over Zn. Hot-pressing as a consolidation procedure in such a short time, under relatively low temperature, made it possible to produce hot-pressed alloys with minimal porosity and unchanged composition. Numerous studies prove the positive effect of Ti-β in comparison with Ti-α in the alloys structure, which improves mechanical properties, i.e., by lowering the Young modulus of titanium and its alloys (Ref 17, 18, 28, 55, 76,77,78). The existence of two or more phases can compromise corrosion resistance and the addition of elements with low standard electrode potential (Ref 9, 45, 47, 61, 67). However, potential of tantalum oxides forming on the surface could introduce antibacterial properties into the sample (Ref 5, 29). Ti alloys enriched in additives of Ta and Mg become slightly more hydrophobic than Ti-Ta-Zn alloys. Grain refinement and well-distribution of elements may affect wettability, as seen with Ti alloys consisting of Ta and Zn, where wetting properties are much closer to bare Ta than Ti (Ref 68). The results obtained in this study suggest how important role plays phase composition, grain refinement, and chemical composition in designing new alloys, i.e., for biomedical applications. Everything stated in this work is susceptible to composition, consolidation temperature, and manufacturing technique.
4 Conclusions
The main goal of this study was to achieve the nanocrystalline biomedical alloy using non-toxic elements such as titanium, tantalum, magnesium and zinc. The influence of Ta, Mg, and Zn content as well as utilizing hot-pressing technique on Ti alloys were studied. It was determined that the addition of Mg and Zn positively influenced the consolidation process, allowing to achieve bulk alloys with minimal porosity. However, Mg brought some more beneficial aspects, such as shortened mechanical alloying time, microstructure refinement, but also improved mechanical properties. Both alloys with Mg and Zn consisting of 30 wt.% Ta and 5% wt.% Mg/Zn exhibited the lowest corrosion potential and corrosion current density among each tested group. The addition of Mg caused a shift toward more hydrophobic surface properties than the Zn additive. All prepared materials are nontoxic; however, the addition of Zn showed significantly better viability of cells. In summary, Mg and Zn have a positive impact on Ta-Ta medical alloy properties and are interesting elements to be introduced in future commercial alloys.
References
M. Abdel-Hady Gepreel and M. Niinomi, Biocompatibility of Ti-Alloys for Long-Term Implantation, J. Mech. Behav. Biomed. Mater., 2013, 20, p 407–415. https://doi.org/10.1016/j.jmbbm.2012.11.014
A. Sidambe, Biocompatibility of Advanced Manufactured Titanium Implants—A Review, Materials, 2014, 7(12), p 8168–8188. https://doi.org/10.3390/ma7128168
P.R. Walker, J. LeBlanc, and M. Sikorska, Effects of Aluminum and Other Cations on the Structure of Brain and Liver Chromatin, Biochemistry, 1989, 28(9), p 3911–3915. https://doi.org/10.1021/bi00435a043
S. Rao, T. Ushida, T. Tateishi, Y. Okazaki, and S. Asao, Effect of Ti, Al, and V Ions on the Relative Growth Rate of Fibroblasts (L929) and Osteoblasts (MC3T3-E1) Cells, Biomed. Mater. Eng., 1996, 6(2), p 79–86. https://doi.org/10.3233/BME-1996-6202
Y.-Y. Chang, H.-L. Huang, H.-J. Chen, C.-H. Lai, and C.-Y. Wen, Antibacterial Properties and Cytocompatibility of Tantalum Oxide Coatings, Surf. Coat. Technol., 2014, 259(PB), p 193–198. https://doi.org/10.1016/j.surfcoat.2014.03.061
C. Yang, J. Li, C. Zhu, Q. Zhang, J. Yu, J. Wang, Q. Wang, J. Tang, H. Zhou, and H. Shen, Advanced Antibacterial Activity of Biocompatible Tantalum Nanofilm via Enhanced Local Innate Immunity, Acta Biomater., 2019, 89, p 403–418. https://doi.org/10.1016/j.actbio.2019.03.027
Y.-L. Zhou, M. Niinomi, T. Akahori, M. Nakai, and H. Fukui, Comparison of Various Properties between Titanium-Tantalum Alloy and Pure Titanium for Biomedical Applications, Mater. Trans., 2007, 48(3), p 380–384. https://doi.org/10.2320/matertrans.48.380
P.J. Little, R. Bhattacharya, A.E. Moreyra, and I.L. Korichneva, Zinc and Cardiovascular Disease, Nutrition, 2010, 26(11–12), p 1050–1057. https://doi.org/10.1016/j.nut.2010.03.007
E. Mostaed, M. Sikora-Jasinska, J.W. Drelich, and M. Vedani, Zinc-Based Alloys for Degradable Vascular Stent Applications, Acta Biomater., 2018, 71, p 1–23. https://doi.org/10.1016/j.actbio.2018.03.005
Y. Chen, Z. Xu, C. Smith, and J. Sankar, Recent Advances on the Development of Magnesium Alloys for Biodegradable Implants, Acta Biomater., 2014, 10(11), p 4561–4573. https://doi.org/10.1016/j.actbio.2014.07.005
M. Balog, A.M.H. Ibrahim, P. Krizik, O. Bajana, A. Klimova, A. Catic, and Z. Schauperl, Bioactive Ti + Mg Composites Fabricated by Powder Metallurgy: The Relation Between the Microstructure and Mechanical Properties, J. Mech. Behav. Biomed. Mater., 2019, 90(October 2018), p 45–53. https://doi.org/10.1016/j.jmbbm.2018.10.008
I. Matuła, G. Dercz, M. Zubko, K. Prusik, and L. Pająk, Influence of High Energy Milling Time on the Ti-50Ta Biomedical Alloy Structure, Acta Phys. Pol. A, 2016, 130(4), p 1033–1036. https://doi.org/10.12693/APhysPolA.130.1033
G. Chen, J. Yin, S. Zhao, H. Tang, and X. Qu, Microstructure and Tensile Properties of a Ti-28Ta Alloy Studied by Transmission Electron Microscopy and Digital Image Correlation, Int. J. Refract. Met. Hard Mater., 2019, 81(February), p 71–77. https://doi.org/10.1016/j.ijrmhm.2019.02.020
Y. Liu, K. Li, H. Wu, M. Song, W. Wang, N. Li, and H. Tang, Synthesis of Ti–Ta Alloys with Dual Structure by Incomplete Diffusion Between Elemental Powders, J. Mech. Behav. Biomed. Mater., 2015, 51, p 302–312. https://doi.org/10.1016/j.jmbbm.2015.07.004
S. Xu, Y. Liu, C. Yang, H. Zhao, B. Liu, J. Li, and M. Song, Compositionally Gradient Ti-Ta Metal-Metal Composite with Ultra-High Strength, Mater. Sci. Eng. A, 2017, 2018(712), p 386–393. https://doi.org/10.1016/j.msea.2017.11.089
Y.L. Zhou, M. Niinomi, and T. Akahori, Effects of Ta Content on Young’s Modulus and Tensile Properties of Binary Ti–Ta Alloys for Biomedical Applications, Mater. Sci. Eng. A, 2004, 371(1–2), p 283–290. https://doi.org/10.1016/j.msea.2003.12.011
S.L. Sing, W.Y. Yeong, and F.E. Wiria, Selective Laser Melting of Titanium Alloy with 50 Wt% Tantalum: Microstructure and Mechanical Properties, J. Alloys Compd., 2016, 660, p 461–470. https://doi.org/10.1016/j.jallcom.2015.11.141
A. Bahador, S. Kariya, J. Umeda, E. Hamzah, and K. Kondoh, Tailoring Microstructure and Properties of a Superelastic Ti-Ta Alloy by Incorporating Spark Plasma Sintering with Thermomechanical Processing, J. Mater. Eng. Perform., 2019, 28(5), p 3012–3020. https://doi.org/10.1007/s11665-019-04061-8
Y.-L. Zhou and M. Niinomi, Ti-25Ta Alloy with the Best Mechanical Compatibility in Ti-Ta Alloys for Biomedical Applications, Mater. Sci. Eng. C, 2009, 29(3), p 1061–1065. https://doi.org/10.1016/j.msec.2008.09.012
G. Dercz, I. Matuła, M. Zubko, A. Kazek-Kęsik, J. Maszybrocka, W. Simka, J. Dercz, P. Świec, and I. Jendrzejewska, Synthesis of Porous Ti-50Ta Alloy by Powder Metallurgy, Mater. Charact., 2018, 142(October 2017), p 124–136. https://doi.org/10.1016/j.matchar.2018.05.033
J. Kesteven, M.B. Kannan, R. Walter, H. Khakbaz, and H.-C. Choe, Low Elastic Modulus Ti–Ta Alloys for Load-Bearing Permanent Implants: Enhancing the Biodegradation Resistance by Electrochemical Surface Engineering, Mater. Sci. Eng. C, 2015, 46, p 226–231. https://doi.org/10.1016/j.msec.2014.10.038
W. Wei, S. Berger, N. Shrestha, and P. Schmuki, Ideal Hexagonal Order: Formation of Self-Organized Anodic Oxide Nanotubes and Nanopores on a Ti-35Ta Alloy, J. Electrochem. Soc., 2010, 157(12), p C409. https://doi.org/10.1149/1.3490424
W.G. Kim, H.C. Choe, and Y.M. Ko, Electrochemical Characteristics of Osteoblast Cultured Ti-Ta Alloy for Dental Implant, J. Korean Inst. Surf. Eng., 2008, 41(2), p 69–75. https://doi.org/10.5695/JKISE.2008.41.2.069
Y.L. Zhou, M. Niinomi, T. Akahori, H. Fukui, and H. Toda, Corrosion Resistance and Biocompatibility of Ti-Ta Alloys for Biomedical Applications, Mater. Sci. Eng. A, 2005, 398(1–2), p 28–36. https://doi.org/10.1016/j.msea.2005.03.032
D. Mareci, R. Chelariu, D.-M. Gordin, G. Ungureanu, and T. Gloriant, Comparative Corrosion Study of Ti-Ta Alloys for Dental Applications, Acta Biomater., 2009, 5(9), p 3625–3639. https://doi.org/10.1016/j.actbio.2009.05.037
N. Soro, H. Attar, E. Brodie, M. Veidt, A. Molotnikov, and M.S. Dargusch, Evaluation of the Mechanical Compatibility of Additively Manufactured Porous Ti–25Ta Alloy for Load-Bearing Implant Applications, J. Mech. Behav. Biomed. Mater., 2019, 97(May), p 149–158. https://doi.org/10.1016/j.jmbbm.2019.05.019
N. Taniguchi, S. Fujibayashi, M. Takemoto, K. Sasaki, B. Otsuki, T. Nakamura, T. Matsushita, T. Kokubo, and S. Matsuda, Effect of Pore Size on Bone Ingrowth into Porous Titanium Implants Fabricated by Additive Manufacturing: An in Vivo Experiment, Mater. Sci. Eng. C, 2016, 59, p 690–701. https://doi.org/10.1016/j.msec.2015.10.069
Y.Q. Ma, W.J. Jin, S.Y. Yang, J. Bin Zhang, Y.X. Huang, and X.J. Liu, Microstructure and Shape Memory Properties of Biomedical Ti-(40–65) Ta (Wt.%) Alloys, Mater. Sci. Forum, 2009, 610–613, p 1382–1386. https://doi.org/10.4028/www.scientific.net/MSF.610-613.1382
M. Sopata, T.M. Karpiński, J. Jakubowicz, and M. Sopata, Development of Tantalum with Highly Hydrophilic Surface and Antimicrobial Properties Obtained by Micro-arc Oxidation Process, J. Biomed. Mater. Res. B Appl. Biomater., 2021, 109(6), p 829–840. https://doi.org/10.1002/jbm.b.34748
M.P. Staiger, A.M. Pietak, J. Huadmai, and G. Dias, Magnesium and Its Alloys as Orthopedic Biomaterials: A Review, Biomaterials, 2006, 27(9), p 1728–1734. https://doi.org/10.1016/j.biomaterials.2005.10.003
J. Wang, L. Liu, Y. Wu, M.F. Maitz, Z. Wang, Y. Koo, A. Zhao, J. Sankar, D. Kong, N. Huang, and Y. Yun, Ex Vivo Blood Vessel Bioreactor for Analysis of the Biodegradation of Magnesium Stent Models With and Without Vessel Wall Integration, Acta Biomater., 2017, 50, p 546–555. https://doi.org/10.1016/j.actbio.2016.12.039
D.M.J. Wilkes, P.S. Goodwin, C.M. Ward-Close, K. Bagnall, and J. Steeds, Solid Solution of Mg in Ti by Mechanical Alloying, Mater. Lett., 1996, 27(1–2), p 47–52. https://doi.org/10.1016/0167-577X(95)00265-0
C.M. Ward-Close and P.G. Partridge, The Production of Titanium-Magnesium Alloys by Vapour Quenching, Mater. Lett., 1991, 11(8–9), p 295–300. https://doi.org/10.1016/0167-577X(91)90206-L
J.L. Murray, The Mg-Ti (Magnesium-Titanium) System, Bull. Alloy Phase Diagr., 1986, 7(3), p 245–248. https://doi.org/10.1007/BF02868999
G. Liang and R. Schulz, Synthesis of Mg-Ti Alloy by Mechanical Alloying, J. Mater. Sci., 2003, 38, p 1179–1184. https://doi.org/10.1023/A:1022889100360
C. Suryanarayana and F.H. Froes, Nanocrystalline Titanium-Magnesium Alloys Through Mechanical Alloying, J. Mater. Res., 1990, 5(9), p 1880–1886. https://doi.org/10.1557/JMR.1990.1880
R.H. Taylor, S. Curtarolo, and G.L.W. Hart, Guiding the Experimental Discovery of Magnesium Alloys, Phys. Rev. B, 2011, 84(8), p 084101. https://doi.org/10.1103/PhysRevB.84.084101
R.M. German, P. Suri, and S.J. Park, Review: Liquid Phase Sintering, J. Mater. Sci., 2009, 44(1), p 1–39. https://doi.org/10.1007/s10853-008-3008-0
G. Adamek, M. Kozlowski, M. Jurczyk, P. Wirstlein, J. Zurawski, and J. Jakubowicz, Formation and Properties of Biomedical Ti-Ta Foams Prepared from Nanoprecursors by Thermal Dealloying Process, Materials, 2019, 12(17), p 2668. https://doi.org/10.3390/ma12172668
H. Okamoto, Ti-Zn (Titanium-Zinc), J. Phase Equilibria Diffus., 2008, 29(2), p 211–212. https://doi.org/10.1007/s11669-008-9271-6
M.H. Qi, J.L. Xu, T. Lai, J. Huang, Y.C. Ma, J.M. Luo, and Y.F. Zheng, Novel Bioactive Ti-Zn Alloys with High Strength and Low Modulus for Biomedical Applications, J. Alloys Compd., 2023, 931, p 167555. https://doi.org/10.1016/j.jallcom.2022.167555
A.J. Drelich, S. Zhao, R.J. Guillory, J.W. Drelich, and J. Goldman, Long-Term Surveillance of Zinc Implant in Murine Artery: Surprisingly Steady Biocorrosion Rate, Acta Biomater., 2017, 58, p 539–549. https://doi.org/10.1016/j.actbio.2017.05.045
H. Yang, C. Wang, C. Liu, H. Chen, Y. Wu, J. Han, Z. Jia, W. Lin, D. Zhang, W. Li, W. Yuan, H. Guo, H. Li, G. Yang, D. Kong, D. Zhu, K. Takashima, L. Ruan, J. Nie, X. Li, and Y. Zheng, Evolution of the Degradation Mechanism of Pure Zinc Stent in the One-Year Study of Rabbit Abdominal Aorta Model, Biomaterials, 2017, 145, p 92–105. https://doi.org/10.1016/j.biomaterials.2017.08.022
J. Venezuela and M.S. Dargusch, The Influence of Alloying and Fabrication Techniques on the Mechanical Properties, Biodegradability and Biocompatibility of Zinc: A Comprehensive Review, Acta Biomater., 2019, 87, p 1–40. https://doi.org/10.1016/j.actbio.2019.01.035
D. Vojtěch, J. Kubásek, J. Šerák, and P. Novák, Mechanical and Corrosion Properties of Newly Developed Biodegradable Zn-Based Alloys for Bone Fixation, Acta Biomater., 2011, 7(9), p 3515–3522. https://doi.org/10.1016/j.actbio.2011.05.008
G. Li, H. Yang, Y. Zheng, X.-H. Chen, J.-A. Yang, D. Zhu, L. Ruan, and K. Takashima, Challenges in the Use of Zinc and Its Alloys as Biodegradable Metals: Perspective from Biomechanical Compatibility, Acta Biomater., 2019, 97(5), p 23–45. https://doi.org/10.1016/j.actbio.2019.07.038
F. Witte, H. Ulrich, M. Rudert, and E. Willbold, Biodegradable Magnesium Scaffolds: Part 1: Appropriate Inflammatory Response, J. Biomed. Mater. Res. A, 2007, 81A(3), p 748–756. https://doi.org/10.1002/jbm.a.31170
K. Kaur, R. Gupta, S.A. Saraf, and S.K. Saraf, Zinc: The Metal of Life, Compr. Rev. Food Sci. Food Saf., 2014, 13(4), p 358–376. https://doi.org/10.1111/1541-4337.12067
A.S. Prasad, B. Bao, F.W.J. Beck, and F.H. Sarkar, Zinc Enhances the Expression of Interleukin-2 and Interleukin-2 Receptors in HUT-78 Cells by Way of NF-ΚB Activation, J. Lab. Clin. Med., 2002, 140(4), p 272–289. https://doi.org/10.1067/mlc.2002.127908
J. Jakubowcz, G. Adamek, M. Sopata, J.K. Kope, and P. Siwak, Hot Pressing of Nanocrystalline Tantalum Using High Frequency Induction Heating and Pulse Plasma Sintering, IOP Conf. Ser. Mater. Sci. Eng., 2017, 283(1), p 012001. https://doi.org/10.1088/1757-899X/283/1/012001
H. Yu, J. Zhou, R. Wang, Q. Su, S. Zhang, J. Wu, X. Wang, and L. Hu, Effects of Hot Pressing Temperature and Annealing Temperature on Microstructure and Compressive Properties of a Bulk Nanocrystalline AZ61 Magnesium Alloy Contain Ti, J. Alloys Compd., 2021, 888, p 161533. https://doi.org/10.1016/j.jallcom.2021.161533
Standard ISO 14577-1:2015. Metallic Materials-Instrumented Indentation Test for Hardness and Materials Parameters—Part 1: Test Method, International Organization for Standardization, (Geneva, Switzerland), 2015.
P. Salwa and T. Goryczka, Influence of Milling Time on Formation of NiTi Alloy Produced by High-Energy Ball Milling, Arch. Metall. Mater., 2019 https://doi.org/10.24425/amm.2019.129489
A. Şelte and B. Özkal, Crystallite Size and Strain Calculations of Hard Particle Reinforced Composite Powders (Cu/Ni/Fe–WC) Synthesized via Mechanical Alloying, Proc. Est. Acad. Sci., 2019, 68(1), p 66–78. https://doi.org/10.3176/proc.2019.1.02
P. Sochacka, A. Miklaszewski, and M. Jurczyk, Development of β-Type Ti-x at.% Mo Alloys by Mechanical Alloying and Powder Metallurgy: Phase Evolution and Mechanical Properties (10 ≤ x ≤ 35), J. Alloys Compd., 2019, 776, p 370–378. https://doi.org/10.1016/j.jallcom.2018.10.217
V. Mote, Y. Purushotham, and B. Dole, Williamson–Hall Analysis in Estimation of Lattice Strain in Nanometer-Sized ZnO Particles, J. Theor. Appl. Phys., 2012, 6(1), p 6. https://doi.org/10.1186/2251-7235-6-6
M. Rabiei, A. Palevicius, A. Monshi, S. Nasiri, A. Vilkauskas, and G. Janusas, Comparing Methods for Calculating Nano Crystal Size of Natural Hydroxyapatite Using X-Ray Diffraction, Nanomaterials, 2020, 10(9), p 1627. https://doi.org/10.3390/nano10091627
B.W. Levinger, Lattice Parameter of Beta Titanium at Room Temperature, JOM, 1953, 5(2), p 195–195. https://doi.org/10.1007/BF03397474
J. Zemann, Crystal Structures, Acta Crystallogr., 1965, 18(1), p 139–139. https://doi.org/10.1107/s0365110x65000361
J.C. Slater, Atomic Radii in Crystals, J. Chem. Phys., 1964, 41(10), p 3199–3204. https://doi.org/10.1063/1.1725697
J. Hieda, M. Niinomi, M. Nakai, and K. Cho, In Vitro Biocompatibility of Ti-Mg Alloys Fabricated by Direct Current Magnetron Sputtering, Mater. Sci. Eng. C, 2015, 54, p 1–7. https://doi.org/10.1016/j.msec.2015.04.029
A.S. Gornakova, A.B. Straumal, I.I. Khodos, I.B. Gnesin, A.A. Mazilkin, N.S. Afonikova, and B.B. Straumal, Effect of Composition, Annealing Temperature, and High Pressure Torsion on Structure and Hardness of Ti-V and Ti-V-Al Alloys, J. Appl. Phys., 2019, 125(8), p 082522. https://doi.org/10.1063/1.5053937
A.S. Gornakova, S.I. Prokofiev, B.B. Straumal, and K.I. Kolesnikova, Growth of (ΑTi) Grain-Boundary Layers in Ti-Co Alloys, Russ. J. Non-Ferr. Met., 2016, 57(7), p 703–709. https://doi.org/10.3103/S1067821216070099
A.S. Gornakova, S.I. Prokofiev, K.I. Kolesnikova, and B.B. Straumal, Formation Regularities of Grain-Boundary Interlayers of the α-Ti Phase in Binary Titanium Alloys, Russ. J. Non-Ferr. Met., 2016, 57(3), p 229–235. https://doi.org/10.3103/S106782121603007X
B.B. Straumal, A.R. Kilmametov, Yu. Ivanisenko, A.S. Gornakova, A.A. Mazilkin, M.J. Kriegel, O.B. Fabrichnaya, B. Baretzky, and H. Hahn, Phase Transformations in Ti-Fe Alloys Induced by High-Pressure Torsion, Adv. Eng. Mater., 2015, 17(12), p 1835–1841. https://doi.org/10.1002/adem.201500143
S. Mendis, W. Xu, H.P. Tang, L.A. Jones, D. Liang, R. Thompson, P. Choong, M. Brandt, and M. Qian, Characteristics of Oxide Films on Ti-(10-75)Ta Alloys and Their Corrosion Performance in an Aerated Hank’s Balanced Salt Solution, Appl Surf Sci, 2020, 506(November 2019), p 145013. https://doi.org/10.1016/j.apsusc.2019.145013
Y. Liu, K. Li, T. Luo, M. Song, H. Wu, J. Xiao, Y. Tan, M. Cheng, B. Chen, X. Niu, R. Hu, X. Li, and H. Tang, Powder Metallurgical Low-Modulus Ti-Mg Alloys for Biomedical Applications, Mater. Sci. Eng. C, 2015, 56, p 241–250. https://doi.org/10.1016/j.msec.2015.06.010
S. Minagar, C. Berndt, and C. Wen, Fabrication and Characterization of Nanoporous Niobia, and Nanotubular Tantala, Titania and Zirconia via Anodization, J. Funct. Biomater., 2015, 6(2), p 153–170. https://doi.org/10.3390/jfb6020153
C.P. Hao, N.J. Cao, Y.H. Zhu, and W. Wang, The Osseointegration and Stability of Dental Implants with Different Surface Treatments in Animal Models: A Network Meta-Analysis, Sci. Rep., 2021, 11(1), p 1–12. https://doi.org/10.1038/s41598-021-93307-4
M. Ferrari, F. Cirisano, and M.C. Morán, Mammalian Cell Behavior on Hydrophobic Substrates: Influence of Surface Properties, Colloids Interfaces, 2019, 3(2), p 48. https://doi.org/10.3390/colloids3020048
H. Garbacz, P. Wieciński, D. Kuczyńska, D. Kubacka, and K.J. Kurzydłowski, The Effect of Grain Size on the Surface Properties of Titanium Grade 2 after Different Treatments, Surf. Coat. Technol., 2018, 335(December 2017), p 13–24. https://doi.org/10.1016/j.surfcoat.2017.12.005
X.H. Wu, Y.K. Liew, C.-W. Mai, and Y.Y. Then, Potential of Superhydrophobic Surface for Blood-Contacting Medical Devices, Int. J. Mol. Sci., 2021, 22(7), p 3341. https://doi.org/10.3390/ijms22073341
J. Jakubowicz, G. Adamek, and L. Smardz, Porous Surface State Analysis of Anodized Titanium for Biomedical Applications, Metall. Mater. Trans. A, 2022, 53(1), p 86–94. https://doi.org/10.1007/s11661-021-06492-2
N. Baig, I. Kammakakam, and W. Falath, Nanomaterials: A Review of Synthesis Methods, Properties, Recent Progress, and Challenges, Mater. Adv., 2021, 2(6), p 1821–1871. https://doi.org/10.1039/D0MA00807A
C.A. Charitidis, P. Georgiou, M.A. Koklioti, A.-F. Trompeta, and V. Markakis, Manufacturing Nanomaterials: From Research to Industry, Manuf. Rev. (Les Ulis), 2014, 1, p 11. https://doi.org/10.1051/mfreview/2014009
M. Long and H.J. Rack, Titanium Alloys in Total Joint Replacement—A Materials Science Perspective, Biomaterials, 1998, 19(18), p 1621–1639. https://doi.org/10.1016/S0142-9612(97)00146-4
M. Marczewski, A. Miklaszewski, and M. Jurczyk, Structure Evolution Analysis in Ultrafine-Grained Zr and Nb-Based Beta Titanium Alloys, J. Alloys Compd., 2018, 765, p 459–469. https://doi.org/10.1016/j.jallcom.2018.06.224
M. Marczewski, A. Miklaszewski, X. Maeder, and M. Jurczyk, Crystal Structure Evolution, Microstructure Formation, and Properties of Mechanically Alloyed Ultrafine-Grained Ti-Zr-Nb Alloys at 36 ≤ Ti ≤ 70 (at.%), Materials, 2020, 13(3), p 87. https://doi.org/10.3390/ma13030587
Acknowledgments
This work was financed by the Ministry of Education and Science in Poland project (0513/SBAD/4739).
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Kozłowski, M., Adamek, G., Siwak, P. et al. The Effect of Ta, Mg, and Zn Content on the Properties of Ti-Ta-Mg and Ti-Ta-Zn Alloys Prepared by Mechanical Alloying and Hot Pressing. J. of Materi Eng and Perform 32, 9825–9837 (2023). https://doi.org/10.1007/s11665-023-08245-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11665-023-08245-1