Abstract
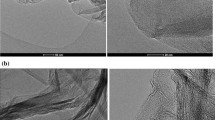
Graphene has shown excellent corrosion protection of copper (Cu). The corrosion protection of Cu is governed by the characteristics of the deposited graphene and Cu surface morphology underneath. In this work, graphene films (1-2, 4-5 layers) were deposited on Cu using a chemical vapor deposition by changing hydrogen (H2) concentration during the annealing stage. The chemical structure and surface microstructural features of graphene/Cu were inspected by Raman spectroscopy, scanning electron microscopy, transmission electron microscopy, and atomic force microscopy. The electrochemical corrosion performance of graphene/Cu was studied in 0.5 M sodium chloride solution using potentiodynamic polarization and electrochemical impedance spectroscopic measurements. The 1-2 layers of graphene and rough Cu surface were obtained for low H2 concentrations (0 and 2.5%), whereas high H2 concentrations, 20 and 50%, resulted in a smooth Cu surface and 4-5 layers of graphene. Our results showed that Cu with a smooth surface and multilayer graphene film exhibited the best corrosion resistance against electrochemical degradation. Tafel analysis revealed that Cu with 4-5 layers of graphene coating corroded nearly three orders of magnitude slower than the annealed Cu, without graphene coating. The results of this study can be useful for several applications where Cu is in close contact with salt species.








Similar content being viewed by others
References
B.P. Singh et al., The Production of a Corrosion Resistant Graphene Reinforced Composite Coating on Copper by Electrophoretic Deposition, Carbon, 2013, 61, p 47–56
V.K. Mittal et al., Formation and Characterization of Bi-layer Oxide Coating on Carbon-Steel for Improving Corrosion Resistance, Thin Solid Films, 2009, 517(5), p 1672–1676
Y. Chen et al., Fabrication and Anti-corrosion Property of Superhydrophobic Hybrid Film on Copper Surface and Its Formation Mechanism, Surf. Interface Anal., 2009, 41(11), p 872–877
J.S. Bunch et al., Impermeable Atomic Membranes from Graphene Sheets, Nano Lett., 2008, 8(8), p 2458–2462
O. Leenaerts, B. Partoens, and F.M. Peeters, Water on Graphene: Hydrophobicity and Dipole Moment Using Density Functional Theory, Phys. Rev. B, 2009, 79(23), p 235440
T. Kim, S. Lee, and H. Park, A Study of Water Transport as a Function of the Micro-Porous Layer Arrangement in PEMFCs, Int. J. Hydrog. Energy, 2010, 35(16), p 8631–8643
N.T. Kirkland et al., Exploring Graphene as a Corrosion Protection Barrier, Corros. Sci., 2012, 56, p 1–4
C. Liang et al., Hydrogen Etching Effect on Single-Crystal Graphene Domains. In: 2013 8th Annual IEEE International Conference on Nano/Micro Engineered and Molecular Systems (IEEE NEMS 2013), 2013: p 697–700
D. Prasai et al., Graphene: Corrosion-Inhibiting Coating, ACS Nano, 2012, 6(2), p 1102–1108
R.K.S. Raman et al., Protecting Copper from Electrochemical Degradation by Graphene Coating, Carbon, 2012, 50(11), p 4040–4045
Y.H. Dong, Q.Q. Liu, and Q. Zhou, Corrosion Behavior of Cu during Graphene Growth by CVD, Corros. Sci., 2014, 89, p 214–219
A. Reina et al., Large Area, Few-Layer Graphene Films on Arbitrary Substrates by Chemical Vapor Deposition, Nano Lett., 2009, 9(1), p 30–35
Y.Q. Liang et al., Large-Scale Synthetic Graphene on Cu as Anti-Corrosion Coating by Chemical Vapor Deposition Approach, Sci. Adv. Mater., 2014, 6(3), p 545–549
A. Ibrahim et al., Effects of Annealing on Copper Substrate Surface Morphology and Graphene Growth by Chemical Vapor Deposition, Carbon, 2015, 94, p 369–377
W. Liu et al., Synthesis of High-Quality Monolayer and Bilayer Graphene on Copper Using Chemical Vapor Deposition, Carbon, 2011, 49(13), p 4122–4130
H. Kim et al., Activation Energy Paths for Graphene Nucleation and Growth on Cu, ACS Nano, 2012, 6(4), p 3614–3623
H. Wang et al., Controllable Synthesis of Submillimeter Single-Crystal Monolayer Graphene Domains on Copper Foils by Suppressing Nucleation (vol 134, pg 3627, 2012), J. Am. Chem. Soc., 2012, 134(44), p 18476–18476
S. Bhaviripudi et al., Role of Kinetic Factors in Chemical Vapor Deposition Synthesis of Uniform Large Area Graphene Using Copper Catalyst, Nano Lett., 2010, 10(10), p 4128–4133
Y.C. Shin and J. Kong, Hydrogen-Excluded Graphene Synthesis via Atmospheric Pressure Chemical Vapor Deposition, Carbon, 2013, 59, p 439–447
L. Gan and Z.T. Luo, Turning Off Hydrogen to Realize Seeded Growth of Subcentimeter Single-Crystal Graphene Grains on Copper, ACS Nano, 2013, 7(10), p 9480–9488
Z.J. Wang et al., Direct Observation of Graphene Growth and Associated Copper Substrate Dynamics by In Situ Scanning Electron Microscopy, ACS Nano, 2015, 9(2), p 1506–1519
X.F. Zhang et al., Hydrogen-Induced Effects on the CVD Growth of High-Quality Graphene Structures, Nanoscale, 2013, 5(18), p 8363–8366
D.H. Jung et al., Effects of Hydrogen Partial Pressure in the Annealing Process on Graphene Growth, J. Phys. Chem. C, 2014, 118(7), p 3574–3580
J. Song, T.Y. Ko, and S. Ryu, Raman Spectroscopy Study of Annealing-Induced Effects on Graphene Prepared by Micromechanical Exfoliation, Bull. Korean Chem. Soc., 2010, 31(9), p 2679–2682
Z.Z. Sun et al., Growth of Graphene from Solid Carbon Sources, Nature, 2010, 468(7323), p 549–552
X.C. Dong et al., Growth of Large-Sized Graphene Thin-Films by Liquid Precursor-Based Chemical Vapor Deposition under Atmospheric Pressure, Carbon, 2011, 49(11), p 3672–3678
J.C. Meyer et al., The Structure of Suspended Graphene Sheets, Nature, 2007, 446(7131), p 60–63
E. Widenkvist et al., Mild Sonochemical Exfoliation of Bromine-Intercalated Graphite: A New Route Towards Graphene, J. Phys. D Appl. Phys., 2009, 42, p 112003
A. Madhankumar et al., EIS Evaluation of Protective Performance and Surface Characterization of Epoxy Coating with Aluminum Nanoparticles After Wet and Dry Corrosion Test, J. Solid State Electrochem., 2012, 16(6), p 2085–2093
S. Akhtar et al., Enhancement of Anticorrosion Property of 304 Stainless Steel Using Silane Coatings, Appl. Surf. Sci., 2018, 440, p 1286–1297
A.V. Benedeti et al., Electrochemical Studies of Copper, Copper-Aluminium and Copper-Aluminium-Silver Alloys: Impedance Results in 0.5 M NaCl, Electrochim. Acta, 1995, 40(16), p 2657–2668
P.G. Pawar et al., Polystyrene Assisted Superhydrophobic Silica Coatings with Surface Protection and Self-Cleaning Approach, Prog. Org. Coat., 2017, 105, p 235–244
A.Y. Adesina, Z.M. Gasem, and A.M. Kumar, Corrosion Resistance Behavior of Single-Layer Cathodic Arc PVD Nitride-Base Coatings in 1 M HCl and 3.5 pct NaCl Solutions, Metall. Mater. Trans. B, 2017, 48(2), p 1321–1332
A.M. Kumar et al., Fabrication of Nitrogen Doped Graphene Oxide Coatings: Experimental and Theoretical Approach for Surface Protection, RSC Adv., 2015, 5(25), p 19264–19272
A.M. Kumar et al., Promising Hard Carbon Coatings on Cu Substrates: Corrosion and Tribological Performance with Theoretical Aspect, J. Mater. Eng. Perform., 2018, 27(5), p 2306–2316
S.D. Costa et al., Resonant Raman Spectroscopy of Graphene Grown on Copper Substrates, Solid State Commun., 2012, 152(15), p 1317–1320
Acknowledgments
The authors acknowledge the support provided by King Fahd University of Petroleum and Minerals in Dhahran, and that of the IMRC administration at Imam Abdulrahman Bin Faisal University in Dammam, Saudi Arabia, as well as that of University of Sharjah, United Arab Emirates.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Akhtar, S., Laoui, T., Ibrahim, A. et al. Few-Layers Graphene Film and Copper Surface Morphology for Improved Corrosion Protection of Copper. J. of Materi Eng and Perform 28, 5541–5550 (2019). https://doi.org/10.1007/s11665-019-04268-9
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11665-019-04268-9