Abstract
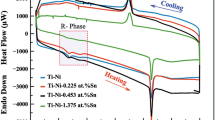
Porous Ti-Nb-xSn shape memory alloys (SMAs) are fabricated by microwave sintering technology. The microstructures exhibit needle-like morphologies, β(N) (normal straight and crossed needles along with needle-like morphology that resembles spaghetti or irregular lines with α-phases in between) as well as plate-like morphologies [normal straight plate-like morphology, α′′ and dendritic plate-like morphology, β(D)]. Increases in Sn addition significantly induce an increase in the density of the α-phase. XRD patterns exhibited three phases, namely the β-main phase with smaller intensities of α′′ and α. Further, the addition of 0.25% Sn led to more effective improvement in the intensity of the α′′-phase compared with 0.5% and 1.5% Sn addition. Additions of Sn also enhanced the fracture strength and its corresponding strain along with the shape memory effect (SME), where the best enhancement was achieved at 0.25% Sn. The corrosion rate (Ri) was reduced by rising Sn content, while both corrosion resistance and antibacterial zones were increased. The lower elastic modulus, as well as the robust mechanical properties and bioactivity, made these SMAs rather suitable for biomedical application purposes, where the low elastic modulus had value in terms of avoiding the problem of “stress shielding.”









Similar content being viewed by others
References
X. Wang, Y. Li, J. Xiong, P.D. Hodgson, and C.E. Wen, Porous TiNbZr Alloy Scaffolds for Biomedical Applications, Acta Biomater., 2009, 5(9), p 3616–3624
A. Choubey, R. Balasubramaniam, and B. Basu, Effect of Replacement of V by Nb and Fe on the Electrochemical and Corrosion Behavior of Ti-6Al-4V in Simulated Physiological Environment, J. Alloy. Compd., 2004, 381(1), p 288–294
Y. Tong, B. Guo, Y. Zheng, C.Y. Chung, and L.W. Ma, Effects of Sn and Zr on the Microstructure and Mechanical Properties of Ti-Ta-Based Shape Memory Alloys, J. Mater. Eng. Perform., 2011, 20(4–5), p 762–766
S. Lu, F. Ma, P. Liu, W. Li, X. Liu, X. Chen, K. Zhang, Q. Han, and L.-C. Zhang, Recrystallization Behavior and Super-Elasticity of a Metastable β-Type Ti-21Nb-7Mo-4Sn Alloy During Cold Rolling and Annealing, J. Mater. Eng. Perform., 2018, 27(8), p 4100–4106
T. Ogawa, H. Takada, and K. Maruoka, Corrosion and Mechanical Degradation of Ni-Ti Superelastic Alloy in Neutral Fluoride Solution, J. Mater. Eng. Perform., 2018, 27, p 1–6
Y. Xiao, H. Liu, D. Yi, J. Le, H. Zhou, Y. Jiang, X. Zhao, Z. Chen, J. Wang, and Q. Gao, High-Temperature Deformation Behavior of Ti-6Al-2Sn-4Zr-2Mo Alloy with Lamellar Microstructure Under Plane-Strain Compression, J. Mater. Eng. Perform., 2018, 27, p 1–14
M. Long and H. Rack, Titanium Alloys in Total Joint Replacement—A Materials Science Perspective, Biomaterials, 1998, 19(18), p 1621–1639
D.M. Cullinane and T.A. Einhorn, Biomechanics of Bone, Princ. Bone Biol., 2002, 1, p 16–32
M. Geetha, A. Singh, R. Asokamani, and A. Gogia, Ti Based Biomaterials, the Ultimate Choice for Orthopaedic Implants—A Review, Prog. Mater Sci., 2009, 54(3), p 397–425
H. Kröger, P. Venesmaa, J. Jurvelin, H. Miettinen, O. Suomalainen, and E. Alhava, Bone Density at the Proximal Femur After Total Hip Arthroplasty, Clin. Orthop. Relat. Res., 1998, 352, p 66–74
T. Ozaki, H. Matsumoto, S. Watanabe, and S. Hanada, Beta Ti Alloys with Low Young’s Modulus, Mater. Trans., 2004, 45(8), p 2776–2779
D. Kuroda, M. Niinomi, M. Morinaga, Y. Kato, and T. Yashiro, Design and Mechanical Properties of New β Type Titanium Alloys for Implant Materials, Mater. Sci. Eng., A, 1998, 243(1), p 244–249
T. Ahmed, A New Low Modulus, Biocompatible Titanium Alloy, in Titanium’95: Science and Technology (1996), pp. 1760–1767
A.K. Mishra, J.A. Davidson, R.A. Poggie, P. Kovacs, and T.J. FitzGerald, Mechanical and Tribological Properties and Biocompatibility of Diffusion Hardened Ti-13Nb-13Zr—A New Titanium Alloy for Surgical Implants, Medical Applications of Titanium and its Alloys: The Material and Biological Issuesed, ASTM International, West Conshohocken, 1996
J. Xu, L. Bao, A. Liu, X. Jin, Y. Tong, J. Luo, Z. Zhong, and Y. Zheng, Microstructure, Mechanical Properties and Superelasticity of Biomedical Porous NiTi Alloy Prepared by Microwave Sintering, Mater. Sci. Eng., C, 2015, 46, p 387–393
D. Yang, Z. Guo, H. Shao, X. Liu, and Y. Ji, Mechanical Properties of Porous Ti-Mo and Ti-Nb Alloys for Biomedical Application by Gelcasting, Proc. Eng., 2012, 36, p 160–167
M. Mour, D. Das, T. Winkler, E. Hoenig, G. Mielke, M.M. Morlock, and A.F. Schilling, Advances in Porous Biomaterials for Dental and Orthopaedic Applications, Materials, 2010, 3(5), p 2947–2974
A. Bansiddhi, T. Sargeant, S.I. Stupp, and D. Dunand, Porous NiTi for Bone Implants: A Review, Acta Biomater., 2008, 4(4), p 773–782
G. Ryan, A. Pandit, and D.P. Apatsidis, Fabrication Methods of Porous Metals for Use in Orthopaedic Applications, Biomaterials, 2006, 27(13), p 2651–2670
H. Matsumoto, S. Watanabe, and S. Hanada, Beta TiNbSn Alloys with Low Young’s Modulus and High Strength, Mater. Trans., 2005, 46(5), p 1070–1078
O. Khalifa, E. Wahab, and A. Tilp, The Effect of Sn and TiO2 Nano Particles Added in Electroless Ni-P Plating Solution on the Properties of Composite Coatings, Aust. J. Basic Appl. Sci., 2011, 5(6), p 136–144
M. Ghoranneviss and S. Shahidi, Effect of Various Metallic Salts on Antibacterial Activity and Physical Properties of Cotton Fabrics, J. Ind. Text., 2013, 42(3), p 193–203
M. Wen, C. Wen, P. Hodgson, and Y. Li, Fabrication of Ti-Nb-Ag Alloy Via Powder Metallurgy for Biomedical Applications, Mater. Des., 2014, 56, p 629–634
J. Xiong, Y. Li, X. Wang, P. Hodgson, and C.E. Wen, Mechanical Properties and Bioactive Surface Modification via Alkali-Heat Treatment of a Porous Ti-18Nb-4Sn Alloy for Biomedical Applications, Acta Biomater., 2008, 4(6), p 1963–1968
D. Zhao, K. Chang, T. Ebel, H. Nie, R. Willumeit, and F. Pyczak, Sintering Behavior and Mechanical Properties of a Metal Injection Molded Ti-Nb Binary Alloy as Biomaterial, J. Alloy. Compd., 2015, 640, p 393–400
D. Zhao, K. Chang, T. Ebel, M. Qian, R. Willumeit, M. Yan, and F. Pyczak, Microstructure and Mechanical Behavior of Metal Injection Molded Ti-Nb Binary Alloys as Biomedical Material, J. Mech. Behav. Biomed. Mater., 2013, 28, p 171–182
F. Kafkas and T. Ebel, Metallurgical and Mechanical Properties of Ti-24Nb-4Zr-8Sn Alloy Fabricated by Metal Injection Molding, J. Alloy. Compd., 2014, 617, p 359–366
A. Aleksanyan, S. Dolukhanyan, V.S. Shekhtman, S. Khasanov, O. Ter-Galstyan, and M. Martirosyan, Formation of Alloys in the Ti-Nb System by Hydride Cycle Method and Synthesis of Their Hydrides in Self-Propagating High-Temperature Synthesis, Int. J. Hydrogen Energy, 2012, 37(19), p 14234–14239
L.W. Ma, C.Y. Chung, Y. Tong, and Y. Zheng, Properties of Porous TiNbZr Shape Memory Alloy Fabricated by Mechanical Alloying and Hot Isostatic Pressing, J. Mater. Eng. Perform., 2011, 20(4–5), p 783–786
A. Terayama, N. Fuyama, Y. Yamashita, I. Ishizaki, and H. Kyogoku, Fabrication of Ti-Nb Alloys by Powder Metallurgy Process and their Shape Memory Characteristics, J. Alloy. Compd., 2013, 577, p S408–S412
X. Wang, Y. Chen, L. Xu, Z. Liu, and K.-D. Woo, Effects of Sn Content on the Microstructure, Mechanical Properties and Biocompatibility of Ti-Nb-Sn/Hydroxyapatite Biocomposites Synthesized by Powder Metallurgy, Mater. Des., 2013, 49, p 511–519
M. Oghbaei and O. Mirzaee, Microwave Versus Conventional Sintering: A Review of Fundamentals, Advantages and Applications, J. Alloy. Compd., 2010, 494(1), p 175–189
S. Das, A. Mukhopadhyay, S. Datta, and D. Basu, Prospects of Microwave Processing: An Overview, Bull. Mater. Sci., 2009, 32(1), p 1–13
R. Roy, D. Agrawal, J. Cheng, and S. Gedevanishvili, Full Sintering of Powdered-Metal Bodies in a Microwave Field, Nature, 1999, 399(6737), p 668–670
H. Bakhsheshi-Rad, M. Idris, M. Abdul-Kadir, A. Ourdjini, M. Medraj, M. Daroonparvar, and E. Hamzah, Mechanical and Bio-corrosion Properties of Quaternary Mg-Ca-Mn-Zn Alloys Compared with Binary Mg-Ca Alloys, Mater. Des., 2014, 53, p 283–292
G. Argade, K. Kandasamy, S. Panigrahi, and R. Mishra, Corrosion Behavior of a Friction Stir Processed Rare-Earth Added Magnesium Alloy, Corros. Sci., 2012, 58, p 321–326
N. Iqbal, M.R.A. Kadir, N.H.B. Mahmood, S. Iqbal, D. Almasi, F. Naghizadeh, H. Balaji, and T. Kamarul, Characterization and Biological Evaluation of Silver Containing Fluoroapatite Nanoparticles Prepared Through Microwave Synthesis, Ceram. Int., 2015, 41(5), p 6470–6477
P.J.S. Buenconsejo, H.Y. Kim, and S. Miyazaki, Effect of Ternary Alloying Elements on the Shape Memory Behavior of Ti-Ta Alloys, Acta Mater., 2009, 57(8), p 2509–2515
P.J.S. Buenconsejo, H.Y. Kim, and S. Miyazaki, Novel β-TiTaAl Alloys with Excellent Cold Workability and a Stable High-Temperature Shape Memory Effect, Scripta Mater., 2011, 64(12), p 1114–1117
H.Y. Kim, T. Fukushima, P.J.S. Buenconsejo, T.-H. Nam, and S. Miyazaki, Martensitic Transformation and Shape Memory Properties of Ti-Ta-Sn High Temperature Shape Memory Alloys, Mater. Sci. Eng., A, 2011, 528(24), p 7238–7246
H. Kim, Y. Ikehara, J. Kim, H. Hosoda, and S. Miyazaki, Martensitic Transformation, Shape Memory Effect and Superelasticity of Ti-Nb Binary Alloys, Acta Mater., 2006, 54(9), p 2419–2429
Y. Chai, H. Kim, H. Hosoda, and S. Miyazaki, Self-Accommodation in Ti-Nb Shape Memory Alloys, Acta Mater., 2009, 57(14), p 4054–4064
J.L. Murray, The Nb-Ti (Niobium-Titanium) System, Bull. Alloy Phase Diagr., 1981, 2(1), p 55–61
H.Y. Kim and S. Miyazaki, Martensitic Transformation and Superelastic Properties of Ti-Nb Base Alloys, Mater. Trans., 2015, 56(5), p 625–634
Y. Guo, beta-bcc and Amorphous Ti-Based Biocompatible Alloys for Human Body Implants, Université Grenoble Alpes, 2014
B. Sharma, S.K. Vajpai, and K. Ameyama, Microstructure and Properties of Beta Ti-Nb Alloy Prepared by Powder Metallurgy Route Using Titanium Hydride Powder, J. Alloy. Compd., 2016, 656, p 978–986
A. Nouri, J. Lin, Y. Li, Y. Yamada, P. Hodgson, C. Wen, Microstructure Evolution of Ti-Sn-Nb Alloy Prepared by Mechanical Alloying, in Materials Forum (CD-ROM), 2007, Institute of Materials Engineering Australasia, pp. 64–70
Q.-M. Hu, S.-J. Li, Y.-L. Hao, R. Yang, B. Johansson, and L. Vitos, Phase Stability and Elastic Modulus of Ti Alloys Containing Nb, Zr, and/or Sn from First-Principles Calculations, Appl. Phys. Lett., 2008, 93(12), p 121902
Y. Guo, K. Georgarakis, Y. Yokoyama, and A. Yavari, On the Mechanical Properties of TiNb Based Alloys, J. Alloy. Compd., 2013, 571, p 25–30
C. Lee, C.-P. Ju, and J. Chern Lin, Structure-Property Relationship of Cast Ti-Nb Alloys, J. Oral Rehabil., 2002, 29(4), p 314–322
R.P. Kolli, W.J. Joost, and S. Ankem, Phase Stability and Stress-Induced Transformations in Beta Titanium Alloys, JOM, 2015, 67(6), p 1273–1280
S. Ehtemam-Haghighi, Y. Liu, G. Cao, and L.-C. Zhang, Influence of Nb on the β → α Martensitic Phase Transformation and Properties of the Newly Designed Ti-Fe-Nb Alloys, Mater. Sci. Eng., C, 2016, 60, p 503–510
N. Vellios and P. Tsakiropoulos, The Role of Sn and Ti Additions in the Microstructure of Nb-18Si Base Alloys, Intermetallics, 2007, 15(12), p 1518–1528
I. Gorna, M. Bulanova, K. Valuiska, M. Bega, O.Y. Koval, A. Kotko, Y.I. Evich, and S. Firstov, Alloys of the Ti-Si-Sn System (Titanium Corner): Phase Equilibria, Structure, and Mechanical Properties, Powder Metall. Met. Ceram., 2011, 50(7–8), p 452–461
M. Kato and H.R. Pak, Thermodynamics of Stress-Induced First-Order Phase Transformations in Solids, Phys. Status Solidi B, 1984, 123(2), p 415–424
T.T. Sasaki, B.C. Hornbuckle, R.D. Noebe, G.S. Bigelow, M.L. Weaver, and G.B. Thompson, Effect of Aging on Microstructure and Shape Memory Properties of a Ni-48Ti-25Pd (At. Pct) Alloy, Metall. Mater. Trans. A, 2013, 44(3), p 1388–1400
J. Gutiérrez-Moreno, Y. Guo, K. Georgarakis, A. Yavari, G. Evangelakis, and C.E. Lekka, The Role of Sn Doping in the β-Type Ti-25 at.% Nb Alloys: Experiment and Ab Initio Calculations, J. Alloy. Compd., 2014, 615, p S676–S679
J. Nagels, M. Stokdijk, and P.M. Rozing, Stress Shielding and Bone Resorption in Shoulder Arthroplasty, J. Shoulder Elbow Surg., 2003, 12(1), p 35–39
M. Niinomi, Metallic Biomaterials, J. Artif. Organs, 2008, 11(3), p 105–110
X. Wu, Q. Peng, J. Zhao, and J. Lin, Effect of Sn Content on the Corrosion Behavior of Ti-Based Biomedical Amorphous Alloys, Int. J. Electrochem. Sci., 2015, 10, p 2045–2054
S.M. Amininezhad, A. Rezvani, M. Amouheidari, S.M. Amininejad, and S. Rakhshani, The Antibacterial Activity of SnO2 Nanoparticles Against Escherichia coli and Staphylococcus aureus, Zahedan J. Res. Med. Sci., 2015, 17(9), p e1053
P. Kamaraj, R. Vennila, M. Arthanareeswari, and S. Devikala, Biological Activities of Tin Oxide Nanoparticles Synthesized Using Plant Extract, Pharm. Pharm. Sci., 2014, 3, p 338–382
L.H. Yun Lu, Y. Hirakawa, and H. Sato, Antibacterial Activity of TiO2/Ti Composite Photocatalyst Films Treated by Ultrasonic Cleaning, Adv. Mater. Phys. Chem., 2012, 2, p 9–12
Y.S. Kim, E.S. Park, S. Chin, G.-N. Bae, and J. Jurng, Antibacterial Performance of TiO2 Ultrafine Nanopowder Synthesized by a Chemical Vapor Condensation Method: Effect of Synthesis Temperature and Precursor Vapor Concentration, Powder Technol., 2012, 215, p 195–199
Y.L. Zhou, M. Niinomi, T. Akahori, H. Fukui, and H. Toda, Corrosion Resistance and Biocompatibility of Ti-Ta Alloys for Biomedical Applications, Mater. Sci. Eng., A, 2005, 398(1), p 28–36
R. Ahmad and M. Sardar, TiO2 Nanoparticles as an Antibacterial Agents Against E. coli, Int. J. Innov. Res. Sci. Eng. Technol., 2013, 2(8), p 3569–3574
G. Ramírez, S. Rodil, H. Arzate, S. Muhl, and J. Olaya, Niobium Based Coatings for Dental Implants, Appl. Surf. Sci., 2011, 257(7), p 2555–2559
Acknowledgments
The authors would like to thank the Ministry of Higher Education of Malaysia and Universiti Teknologi Malaysia for providing the financial support under the University Research Grant No. Q.J130000.2524.12H60 and research facilities.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ibrahim, M.K., Hamzah, E. & Saud, S.N. Microstructure, Phase Transformation, Mechanical Behavior, Bio-corrosion and Antibacterial Properties of Ti-Nb-xSn (x = 0, 0.25, 0.5 and 1.5) SMAs. J. of Materi Eng and Perform 28, 382–393 (2019). https://doi.org/10.1007/s11665-018-3776-x
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11665-018-3776-x