Abstract
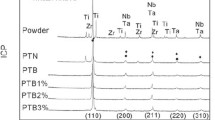
Recently, porous Ti-Nb-based shape memory alloys have been considered as promising implants for biomedical application, because of their non-toxic elements, low elastic modulus, and stable superelasticity. However, the inverse relationship between pore characteristics and superelasticity of porous SMAs will strongly affect their clinical application. Until now, there have been few works specifically focusing on the effect of pore structure on the mechanical properties and superelasticity of porous Ti-Nb-based SMAs. In this study, the pore structure, including porosity and pore size, of porous Ti-22Nb-6Zr alloys was successfully regulated by adjusting the amount and size of space-holder particles. XRD and SEM investigation showed that all these porous alloys had homogeneous composition. Compression tests indicated that porosity played an important role in the mechanical properties and superelasticity of these porous alloys. Those alloys with porosity in the range of 38.5%-49.7% exhibited mechanical properties approaching to cortical bones, with elastic modulus, compressive strength, and recoverable strain in the range of 7.2-11.4 GPa, 188-422 MPa, and 2.4%-2.6%, respectively. Under the same porosity, the alloys with larger pores exhibited lower elastic modulus, while the alloys with smaller pores presented higher compressive strength.







Similar content being viewed by others
References
K. Rezwan, Q.Z. Chen, J.J. Blaker, and A.R. Boccaccini, Biodegradable and Bioactive Porous Polymer/Inorganic Composite Scaffolds for Bone Tissue Engineering, Biomaterials, 2006, 27(18), p 3413–3431
M.H. Elahinia, M. Hashemi, M. Tabesh, and S.B. Bhaduri, Manufacturing and Processing of NiTi Implants: A Review, Prog. Mater. Sci., 2012, 57, p 911–946
A. Biesiekierski, J. Wang, M. Gepreel, and C.E. Wen, A New Look at Biomedical Ti-Based Shape Memory Alloys, Acta Biomater., 2012, 8(5), p 1661–1669
S. Miyazaki, H.Y. Kim, and H. Hosoda, Development and Characterization of Ni-Free Ti-Base Shape Memory and Superelastic Alloys, Mater. Sci. Eng. A., 2006, 438-440, p 18–24
C. Baker, The Shape-Memory Effect in a Titanium-35 wt.% Niobium Alloy, Met. Sci. J., 1971, 5, p 92–100
H.Y. Kim, Y. Ikehara, J.I. Kim, H. Hosoda, and S. Miyazaki, Martensitic Transformation, Shape Memory Effect and Superelasticity of Ti-Nb Binary Alloys, Acta Mater., 2006, 54(9), p 2419–2429
J.I. Kim, H.Y. Kim, H. Hosoda, and S. Miyazaki, Shape Memory Behavior of Ti-22Nb-(0.5-2.0)O(at.%) Biomedical Alloys, Mater. Trans., 2005, 46(4), p 852–857
J.I. Kim, H.Y. Kim, T. Inamura, H. Hosoda, and S. Miyazaki, Shape Memory Characteristics of Ti-22Nb-(2-8)Zr(at.%) Biomedical Alloys, Mater. Sci. Eng. A, 2005, 403(1-2), p 334–339
Y. Fukui, T. Inamura, H. Hosoda, K. Wakashima, and S. Miyazaki, Mechanical Properties of a Ti-Nb-Al Shape Memory Alloy, Mater. Trans., 2004, 45(4), p 1077–1082
J.Y. Rho, T.Y. Tsui, and G.M. Pharr, Elastic Properties of Human Cortical and Trabecular Lamellar Bone Measured by Nanoindentation, Biomaterials, 1997, 18(20), p 1325–1330
G. Ryan, A. Pandit, and D.P. Apatsidis, Fabrication Methods of Porous Metals for Use in Orthopaedic Applications, Biomaterials, 2006, 27(8), p 2651–2670
V. Brailovski, S. Prokoshkin, M. Gauthier, K. Inaekyan, and S. Dubinskiy, Mechanical Properties of Porous Metastable Beta Ti-Nb-Zr Alloys for Biomedical Applications, J. Alloy. Compd., 2013, 577, p S413–S417
V. Brailovski, S. Prokoshkin, M. Gauthier, K. Inaekyan, S. Dubinskiy, M. Petrzhik, and M. Filonov, Bulk and Porous Metastable Beta Ti-Nb-Zr(Ta) Alloys for Biomedical Applications, Mater. Sci. Eng. C., 2011, 31(3), p 643–657
L.W. Ma, C.Y. Chung, Y.X. Tong, and Y.F. Zheng, Properties of Porous TiNbZr Shape Memory Alloy Fabricated by Mechanical Alloying and Hot Isostatic Pressing, J. Mater. Eng. Perform., 2011, 20(4-5), p 783–786
X.J. Wang, Y.C. Li, J.Y. Xiong, P.D. Hodgson, and C.E. Wen, Porous TiNbZr Alloy Scaffolds for Biomedical Applications, Acta Biomater., 2009, 5(9), p 3616–3624
J.P. Li, J.R. Wijn, C.A. Van Blitterswijk, and K. Groot, Porous Ti6Al4V Scaffold Directly Fabricating by Rapid Prototyping: Preparation and In Vitro Experiment, Biomaterials, 2006, 27(8), p 1223–1235
M. Niinomi, Recent Research and Development in Titanium Alloys for Biomedical Applications and healthcare goods, Sci. Technol. Adv. Mater., 2003, 4(5), p 445–454
A. Terayama, N. Fuyama, Y. Yamashita, I. Ishizaki, and H. Kyogoku, Fabrication of Ti-Nb Alloys by Powder Metallurgy Process and Their Shape Memory Characteristics, J. Alloy. Compd., 2013, 577, p S408–S412
S.D. Ryan and J.L. Williams, Tensile Testing of Rodlike Trabeculae Excised from Bovine Femoral Bone, J. Biomech., 1989, 22(4), p 351–355
X.J. Wang, Y.C. Li, P.D. Hodgson, and C.E. Wen, Nano- and Macro-Scale Characterisation of the Mechanical Properties of Bovine Bone, Mater. Forum, 2007, 31, p 156–159
L.J. Gibson and M.F. Ashby, Cellular Solids: Structure and Properties, 2nd ed., Cambridge University Press, Cambridge, 1999, p 183–197
D. Lin, C. Chuang, J. Chern Lin, J. Lee, C. Ju, and H. Yin, Bone Formation at the Surface of Low Modulus Ti-7.5Mo Implants in Rabbit Femur, Biomaterials, 2007, 28(16), p 2582–2589
E.B. Taddei, V.A.R. Henriques, C.R.M. Silva, and C.A.A. Cairo, Production of New Titanium Alloy for Orthopedic Implants, Mater. Sci. Eng. C, 2004, 24(5), p 683–687
T. Aydogmus and S. Bor, Superelasticity and Compression Behavior of Porous TiNi Alloys Produced Using Mg Spacers, J. Mech. Behav. Biomed., 2012, 15, p 59–69
S.A. Shabalovskaya, On the Nature of the Biocompatibility and on Medical Applications of NiTi Shape Memory and Superelastic Alloys, Bio-Med. Mater. Eng., 1996, 6(4), p 267–289
M. Lai, Y. Gao, B. Yuan, and M. Zhu, Indirect Determination of Martensitic Transformation Temperature of Sintered Nickel-Free Ti-22Nb-6Zr Alloy by Low Temperature Compression Test, Mater. Des., 2014, 60, p 193–197
Acknowledgments
The authors acknowledge the supports from Guangdong Provincial Science and Technology Projects (2012B010200020), New Century Excellent Talents in University (NCET-12-0201), and Fundamental Research Funds for the Central Universities (2014ZG0026) and Guangdong Natural Science Funds (S2013010012487).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lai, M., Gao, Y., Yuan, B. et al. Effect of Pore Structure Regulation on the Properties of Porous TiNbZr Shape Memory Alloys for Biomedical Application. J. of Materi Eng and Perform 24, 136–142 (2015). https://doi.org/10.1007/s11665-014-1299-7
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11665-014-1299-7