Abstract
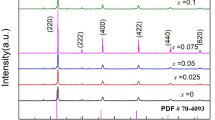
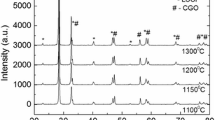
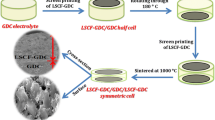
In this study, a potential cathode material, La0.6Sr0.4CoxFe1−xO3 (LSCF), for intermediate-temperature solid oxide fuel cells (SOFCs) is synthesized via a citric acid-assisted combustion method. The electrolyte-supported solid oxide fuel cell is formed in a conventional way to estimate the cell performance. Results show that the as-prepared powders have a pure perovskite structure after calcination at 1050°C, and the initial powders have a fluffy and spongy mass with a porous structure. The cell performance is affected by the LSCF powder morphology and calcination temperature. The peak power density of the Y2O3-stabilized ZrO2 (YSZ) electrolyte-supported single cell using LSCF as cathode is 28 mW cm−2 at 800°C, while the smallest area-specific resistance obtained is 19 Ω cm2. This suggests that La0.6Sr0.4CoxFe1−xO3 is a promising potential cathode material for SOFCs.




Similar content being viewed by others
References
Y. Liu, Z. Shao, M. Toshiyuki, and S. Jiang, Development of Nickel Based Cermet Anode Materials in Solid Oxide Fuel Cells-Now and Future. Mater. Rep.: Energy 1, 100003 (2020).
M. Beigzadeh, F. Pourfayaz, M. Ghazvini, and M.H. Ahmadi, Energy and Exergy Analyses of Solid Oxide Fuel Cell-Gas Turbine Hybrid Systems Fed by Different Renewable Biofuels: A Comparative Study. J. Clean. Prod. 280, 124383 (2021).
O. Sharaf and M. Orhan, An Overview of Fuel Cell Technology: Fundamentals and Applications. Renew. Sust. Energ. Rev. 32, 810 (2014).
K. Al-Hamed and I. Dincer, Development and Optimization of a Novel Solid Oxide Fuel Cell-Engine Powering System for Cleaner Locomotives. Appl. Therm. Eng. 183, 116150 (2021).
M. Shen and P. Zhang, Progress and Challenges of Cathode Contact Layer for Solid Oxide Fuel Cell. Int. J. Hydrogen Energ. 45, 33876 (2020).
Z. Lyu, H. Li, Y. Wang, and M. Han, Performance Degradation of Solid Oxide Fuel Cells Analyzed by Evolution of Electrode Processes Under Polarization. J. Power Sour. 485, 229237 (2021).
B. Maguire, F. Marques, and J. Labrincha, Cathode Materials for Intermediate Temperature SOFCs. Solid State Ion. 127, 329 (2000).
B. Zhu, Advantages of Intermediate Temperature Solid Oxide Fuel Cells for Tractionary Applications. J. Power Sour. 93, 82 (2001).
Y. Bu, S. Joo, Y. Zhang, Y. Wang, D. Meng, X. Ge, and G. Kim, A Highly Efficient Composite Cathode for Proton-Conducting Solid Oxide Fuel Cells. J. Power Sour. 451, 227812 (2020).
S. Rehman, R. Song, T. Lim, J. Hong, and S. Lee, Parametric Study on Electrodeposition of a Nanofibrous LaCoO3 SOFC Cathode. Ceram. Int. 47, 5570 (2021).
K. Song, Z. Yu, X. Luo, S. Zhu, Y. Yang, Q. Yang, D. Tian, X. Lu, Y. Ding, Y. Chen, and B. Lin, A Simple Ce-Doping Strategy to Enhance Stability of Hybrid Symmetrical Electrode for Solid Oxide Fuel Cells. Int. J. Hydrogen Energ. 45, 29259 (2020).
H. Gu, M. Xu, Y. Song, C. Zhou, C. Su, W. Wang, R. Ran, W. Zhou, and Z. Shao, SrCo0.8Ti0.1Ta0.1O3-δ Perovskite: A New Highly Active and Durable Cathode Material for Intermediate-Temperature Solid Oxide Fuel Cells. Compos. Part B-Eng. 213, 108726 (2021).
A. Abdalla, M. Kamel, S. Hossain, J. Irvine, and A. Azad, Synthesis and Electrochemical Characterization of La0.75Sr0.25Mn0.5Cr0.5−xAlxO3 for IT- and HT-SOFCs. Int. J. Appl. Ceram. Tec. 17, 1276 (2019).
K. Pei, Y. Zhou, K. Xu, Z. He, Y. Chen, W. Zhang, S. Yoo, B. Zhao, W. Yuan, M. Liu, and Y. Chen, Enhanced Cr-Tolerance of an SOFC Cathode by an Efficient Electro-Catalyst Coating. Nano Energy 72, 104704 (2020).
N. Droushiotis, A. Torabi, M. Othman, T. Etsell, and G. Kelsall, Effects of Lanthanum Strontium Cobalt Ferrite (LSCF) Cathode Properties on Hollow Fibre Micro-Tubular SOFC Performances. J. Appl. Electrochem. 42, 517 (2012).
G. DiGiuseppe, D. Thompson, C. Gumeci, A. Hussain, and N. Dale, Distribution of Relaxation Times Analysis and Interfacial Effects of LSCF Fired at Different Temperatures. Int. J. Hydrogen Energy. 44, 27067 (2019).
F. Zhou, Y. Liu, X. Zhao, W. Tang, S. Yang, S. Zhong, and M. Wei, Effects of Cerium Doping on the Performance of LSCF Cathodes for Intermediate Temperature Solid Oxide Fuel Cells. Int. J. Hydrogen Energ. 43, 18946 (2018).
S. Biswas, T. Nithyanantham, N. Saraswathi, and S. Bandopadhyay, Evaluation of Elastic Properties of Reduced NiO-8YSZ Anode-Supported Bi-Layer SOFC Structures at Elevated Temperatures in Ambient Air and Reducing Environments. J. Mater. Sci. 44, 778 (2009).
L. da Conceição, A. Silva, N. Ribeiro, and M. Souza, Combustion Synthesis of La0.7Sr0.3Co0.5Fe0.5O3 (LSCF) Porous Materials for Application as Cathode in IT-SOFC. Mater. Res. Bull. 46, 308 (2011).
Y. Boyjoo, M. Wang, V. Pareek, J. Liu, and M. Jaroniec, Synthesis and Applications of Porous Non-Silica Metal Oxide Submicrospheres. Chem. Soc. Rev. 45, 6013 (2016).
Y. Shi, Y. Wen, K. Huang, X. Xiong, J. Wang, M. Liu, D. Ding, Y. Chen, and T. Liu, Surface Enhanced Performance of La0.6Sr0.4Co0.2Fe0.8O3-δ Cathodes by Infiltration Pr-Ni-Mn-O Progress. J. Alloy. Compd. 902, 163337 (2022).
D. Osinkin, S. Beresnev, and N. Bogdanovicha, Influence of Pr6O11 on Oxygen Electroreduction Kinetics and Electrochemical Performance of Sr2Fe1.5Mo0.5O6-δ Based Cathode. J. Power Sour. 392, 41 (2018).
Y. Hu, Y. Su, C. Li, C. Li, and G. Yang, Dense Mn1.5Co1.5O4 Coatings with Excellent Long-Term Stability and Electrical Performance Under the SOFC Cathode Environment. Appl. Surf. Sci. 499, 143726 (2020).
Acknowledgments
This work was supported by the Natural Science Foundation of Anhui Province of China under contact no. 2108085ME152 and the Talent Research Fund Project of Hefei University under contact no. 21-22RC34.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no competing interests to declare that are relevant to the content of this article.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Wang, P., Cheng, J. Preparation and Performance of a La0.6Sr0.4CoxFe1−xO3 Cathode for Solid Oxide Fuel Cells. J. Electron. Mater. 51, 6410–6415 (2022). https://doi.org/10.1007/s11664-022-09876-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11664-022-09876-1