Abstract
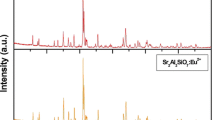
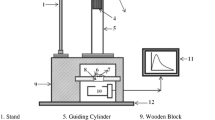
A single-host lattice, white light-emitting SrAl2O4:Dy3+ phosphor was synthesized by a solid-state reaction method. The crystal structure of prepared SrAl2O4:Dy3+ phosphor was in a monoclinic phase with space group P21. The chemical composition of the sintered SrAl2O4:Dy3+ phosphor was confirmed by the energy dispersive x-ray spectroscopy technique. Under ultra-violet excitation, the characteristic emissions of Dy3+ are peaking at 475 nm, 573 nm and 660 nm, originating from the transitions of 4F9/2 → 6H15/2, 4F9/2 →&!nbsp; 6H13/2 and 4F9/2 → 6H11/2 in the 4f9 configuration of Dy3+ ions. Commission International de I’Eclairage color coordinates of SrAl2O4:Dy3+ are suitable for white light-emitting phosphor. In order to investigate the suitability of the samples as white color light sources for industrial uses, correlated color temperature (CCT) and color rendering index (CRI) values were calculated. Values of CCT and CRI were found well within the defined acceptable range. Mechanoluminescence (ML) intensity of SrAl2O4:Dy3+ phosphor increased linearly with increasing impact velocity of the moving piston. Thus, the present investigation indicates piezo-electricity was responsible for producing ML in sintered SrAl2O4:Dy3+ phosphor. Decay rates of the exponential decaying period of the ML curves do not change significantly with impact velocity. The photoluminescence and ML results suggest that the synthesized SrAl2O4:Dy3+ phosphor was useful for the white light-emitting diodes and stress sensor respectively.
Similar content being viewed by others
References
C. Feldmann, T. Justel, C.R. Ronda, and P.J. Schmidt, Adv. Funct. Mater. 13, 511 (2003).
J.S. Kim, P.E. Jeon, J.C. Choi, H. Lpark, S.I. Mho, and G.C. Kim, Appl. Phys. Lett. 84, 2931 (2004).
G.H. Lee, T.H. Kim, C. Yoon, and S. Kang, J. Lumin. 128, 1922 (2008).
Z.P. Yang, X. Li, Y. Yang, and X.M. Li, J. Lumin. 707, 122 (2007).
G.Q. Yao, J.F. Duan, M. Ren, H.D. Yu, and J.H. Lin, J. Lumin. 22, 21 (2001).
G.D. Xia, S.M. Zhou, J.J. Zhang, and J. Xu, J. Cryst. Growth 279, 352 (2005).
Q. Su, Z. Pei, L. Chi, H. Zhang, Z. Zhang, and F. Zou, J. Alloys Compd. 193, 25 (1993).
Q. Su, Z. Pei, J. Lin, and F. Xue, J. Alloys Compd. 225, 103 (1995).
H.B. Liang, Q. Zeng, Y. Tao, S. Wang, and Q. Su, Mater. Sci. Eng., B 98, 213 (2003).
Y. Fang, W.D. Zhuang, Y.S. Hu, X.Y. Ye, and X.W. Huang, J. Alloys Compd. 455, 420 (2008).
F. Zhang, T. Zhang, G. Li, and W. Zhang, J. Alloys Compd. 618, 484 (2015).
M. Shi, D. Zhang, and C. Chang, J. Alloys Compd. 639, 168 (2015).
M.A. Tshabalalaa, F.B. Dejene, S.S. Pitale, S.C. Swart, and O.M. Ntwaeaborwa, Phys. B 439, 126 (2014).
H. Yamamoto and T. Matsuzawa, J. Lumin. 72–74, 287 (1997).
Y. Lin, Z. Zhang, F. Zhang, and Z. Tang, Mater. Chem. Phys. 65, 103 (2000).
M. Murayama, N. Takeuchi, Y. Aoki, and T. Matsuzawa, Phosphorescent Phosphor, US Patent 5424006 (1995).
D.R. Vij, Luminescence of Solids (New York: Plenum, Springer, 1998).
I.P. Sahu, D.P. Bisen, and N. Brahme, J. Biol. Chem. Lumin. 30, 526 (2015).
C.N. Xu, X.G. Zheng, M. Akiyama, K. Nonaka, and T. Watanabe, Appl. Phys. Lett. 76, 179 (2000).
H. Zhang, H. Yamada, N. Terasaki, and C.N. Xu, Appl. Phys. Lett. 91, 081905 (2007).
C. Zhao, D. Chen, Y. Yuan, and M. Wu, Mater. Sci. Eng., B 133, 200 (2006).
JCPDS File Number 34-0379, JCPDS International Center for Diffraction Data.
S.D. Han, K.C. Singh, T.Y. Cho, H.S. Lee, D. Jakhar, J.P. Hulme, C.H. Han, J.D. Kim, I.S. Chun, and J. Gwak, J. Lumin. 128, 301 (2008).
S. Chawla, N. Kumar, and H. Chander, J. Lumin. 129, 114 (2009).
K.E. Foka, F.B. Dejene, and H.C. Swart, Phys. B 439, 177 (2014).
M. Misevicius, O. Scit, I. Grigoraviciute-Puroniene, G. Degutis, I. Bondanovicience, and A. Kareiva, Ceram. Int. 38, 5915 (2012).
I.P. Sahu, D.P. Bisen, and N. Brahme, Displays 35, 279 (2014).
Y. Cheng, Y. Zhao, Y. Zhang, and X. Cao, J. Colloid Interface Sci. 344, 321 (2010).
M.A. Salim, R. Hussain, M.S. Abdullah, S. Abdullah, N.S. Alias, S.A. Ahmadfuzi, M.N. Mdyusuf, and K.M. Mahbor, Solid State Sci Technol 17, 59 (2009).
G.S. Rama Raju, J.Y. Park, H.C. Jung, B.K. Moon, J.H. Jeong, and J.H. Kim, Curr. Appl. Phys. 9, 92 (2009).
Y. Chen, X. Cheng, M. Liu, Z. Qi, and C. Shi, J. Lumin. 129, 531 (2009).
I.P. Sahu, D.P. Bisen, and N. Brahme, Displays 38, 68 (2015).
A. Zukauskas, M.S. Shur, and R. Gaska, Introduction to Solid State Lighting (New York: Wiley, 2002).
CIE (1931) International Commission on Illumination. Publication CIE No. 15 (E-1.3.1).
C.S. McCamy, Color Res. Appl. 17, 142 (1992).
I.P. Sahu, D.P. Bisen, N. Brhame, R.K. Tamrakar, and R. Shrivastava, J. Mater. Sci. Mater. Electron. 26, 8824 (2015).
I.P. Sahu, D.P. Bisen, N. Brhame, R.K. Tamrakar, and R. Shrivastava, J. Mater. Sci. Mater. Electron. 26, 9907 (2015).
R. Shrivastava, J. Kaur, and M. Dash, Superlattices Microstruct. 82, 262 (2015).
T. Matsuzawa, Y. Aoki, N. Takeuchi, and Y. Murayama, J. Electrochem. Soc. 143, 2670 (1996).
B.P. Chandra, J. Lumin. 131, 1203 (2011).
I.P. Sahu, D.P. Bisen, and N. Brahme, Res. Chem. Intermed. 41, 6649 (2015).
I.P. Sahu, D.P. Bisen, N. Brahme, L. Wanjari, and R. Tamrakar, Res. Chem. Intermed. 41, 8797 (2015).
I.P. Sahu, D.P. Bisen, and N. Brahme, Lumin. J. Biol. Chem. Lumin. 30, 1125 (2015).
I.P. Sahu, D.P. Bisen, N. Brahme, and M. Ganjir, Lumin. J. Biol. Chem. Lumin. 30, 1318 (2015)
R. Shrivastava, J. Kaur, and B.P. Chandra, Lumin. J. Biol. Chem Lumin. (2015). doi:10.1002/bio.2882.
I.P. Sahu, D.P. Bisen, N. Brahme, and R.K. Tamrakar, J. Lumin. 167, 278 (2015).
I.P. Sahu, P. Chandrakar, R.N. Baghel, D.P. Bisen, N. Brahme, and R.K. Tamrakar, J. Alloys Compd. 649, 1329 (2015).
H. Zhang, H. Yamada, N. Terasaki, and C.N. Xu, Int. J. Mod. Phys. B 23, 1028 (2009).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sahu, I. ., Bisen, D.P., Brahme, N. et al. Generation of White Light from Dysprosium-Doped Strontium Aluminate Phosphor by a Solid-State Reaction Method. J. Electron. Mater. 45, 2222–2232 (2016). https://doi.org/10.1007/s11664-015-4284-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11664-015-4284-5