Abstract
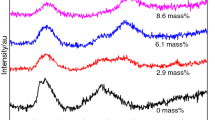
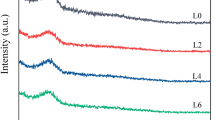
The effect of the substitution of CaF2 with Li2O on the viscosity and structure of low-fluoride CaF2-CaO-Al2O3-MgO slag was studied with an aim to develop low-fluoride slag for electroslag remelting. Increasing Li2O addition up to 4.5 mass pct was observed to significantly reduce the slag viscosity monotonically. Increasing temperature significantly lowered the viscosity of slag, whereas this influence is less effective with increasing Li2O content especially above 3.5 mass pct. The activation energy for viscous flow decreases with increasing Li2O content. The polymerization degree of aluminate networks decreased with increasing Li2O content, as demonstrated by Raman analysis. The dominant structural unit in [AlO4]5−-tetrahedral network is \( {\text{Q}}_{\text{Al}}^{4} \). The amount of symmetric Al-O0 stretching vibrations significantly decreased with increasing Li2O content. The relative fraction of \( {\text{Q}}_{\text{Al}}^{4} \) in the [AlO4]5−-tetrahedral units shows a decreasing trend, whereas \( {\text{Q}}_{\text{Al}}^{2} \) increases with the increase in Li2O content accordingly. The change in slag viscosity with chemistry variation agrees well with the changes in slag structural units.








Similar content being viewed by others
References
H. Nakada and K. Nagata: ISIJ Int., 2006, vol. 46, pp. 441–49.
[2] N. Takahira, M. Hanao, and Y. Tsukaguchi: ISIJ Int., 2013, vol. 53, pp. 818–822.
J.L. Klug, R. Hagemann, N.C. Heck, A.C.F. Vilela, H.P. Heller, and P.R. Scheller: Steel Res. Int., 2012, vol. 83, pp. 1186-1193.
[4] M. Persson, S. Seetharaman, and S. Seetharaman: ISIJ Int., 2007, vol. 47, pp. 1711-1717.
[5] T. Omoto, Y. Iwamoto, and H. Yamaji: Shinagawa Tech. Rep., 2002, vol. 45, pp. 85.
D.L. Xiang: Heavy Casting Forging, 2011, pp. 26–35.
K. Narita, T. Onoye, T. Ishii, and T. Kusamichi: Tetsu–to–Hagané, 1978, vol. 64, pp. 1568–77.
[8] H. Mao and Z. Li: Central Iron and Steel Research Institute Technical Bulletin. 1983, vol. 3, pp. 597-611.
[9] Z.B. Li: Electroslag Metallurgy Theory and Practice, Metallurgical Industry Press, Beijing, China, 2010.
[10] H. Kim and I. Sohn: ISIJ Int., 2011, vol. 51, pp. 1–8.
[11] H.S. Park, H. Kim, and I. Sohn: Metall. Mater. Trans. B, 2011, vol. 42B, pp. 324-330.
[12] G.H. Kim and I. Sohn: ISIJ Int., 2012, vol. 52, pp. 68–73.
[13] W. Wang, X. Yan, L. Zhou. S. Xie, and D. Huang: Metall. Mater. Trans. B, 2016, vol. 47B, pp. 963–973.
[14] C.B. Shi, J. Li, J.W. Cho, F. Jiang, and I.H. Jung: Metall. Mater. Trans. B, 2015, vol. 46B, pp. 2110-2120.
[15] S.H. Shin, D.W. Yoon, J.W. Cho, and S.H. Kim: J. Non-Cryst. Solids, 2015, vol. 425, pp. 83–90.
[16] S.H. Shin, J.W. Cho, and S.H. Kim: J. Am. Ceram. Soc., 2014, vol. 97, pp. 3263–3269.
[17] G.H. Kim, C.S. Kim, and I. Sohn: ISIJ Int., 2013, vol. 53, pp. 170–176.
[18] T.A. Litovitz: J. Chem. Phys., 1952, vol. 20, pp. 1088–1089.
[19] G.H. Kim and I. Sohn: J. Non-Cryst. Solids, 2012, vol. 358, pp. 1530–1537.
[20] P.W. Atkins: Physical Chemistry, 4th ed., Oxford University Press, Oxford, UK, 1990, p. 671.
[21] K.C. Mills: ISIJ Int., 1993, vol. 33, pp. 148-155.
[22] K.C. Mills: ISIJ Int., 2016, vol. 1, pp. 1-13.
[23] B.O. Mysen, L.W. Finger, D.Virgo, and F.A. Seifert: Am. Mineral., 1982, vol. 67, pp. 686-695.
[24] J.D. Frantz and B.O. Mysen: Chem. Geol., 1995, vol. 121, pp. 155–176.
[25] B.O. Mysen, D. Virgo, and I. Kushiro: Am. Mineralogist, 1981, vol. 66, pp. 678-701.
[26] T.S. Kim and J.H. Park: ISIJ Int., 2014, vol. 54, pp. 2031–2038.
[27] V.N. Bykov, A.A. Osipov, and V.N. Anfilogov: Glass Phy. Chem., 2003, vol. 29, pp. 105–07.
[28] C. Huang and E.C. Behrman: J. Non-Cryst. Solids, 1991, vol. 128, pp. 310-328.
[29] P. McMillan and B. Piriou: J. Non-Cryst. Solids, 1983, vol. 55, pp. 221-242.
[30] M. Licheron, V. Montouillout, F. Millot, and D.R. Neuville: J. Non-Cryst. Solids. 2011, vol. 357, pp. 2796–2801.
Acknowledgments
This work was financially supported by the Fundamental Research Funds for the Central Universities (Grant No. FRF-TP-15-010A2), and China Postdoctoral Science Foundation (Grant No. 2016T90035).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Manuscript submitted April 21, 2016.
Rights and permissions
About this article
Cite this article
Shi, CB., Shin, SH., Zheng, DL. et al. Development of Low-Fluoride Slag for Electroslag Remelting: Role of Li2O on the Viscosity and Structure of the Slag. Metall Mater Trans B 47, 3343–3349 (2016). https://doi.org/10.1007/s11663-016-0826-3
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11663-016-0826-3