Abstract
In recent years, various methods to decrease carbon dioxide emissions from iron and steel making industries have been developed. The latest blast furnace operation design is intended to induce the low reducing agent operation, highly reactive material is considered a promising way to improve reaction efficiency. Another method utilizes hydrogen in the blast furnace process for highly efficient reduction. Mathematical modeling may help to predict complex in-furnace phenomena, including momentum, heat, and mass transport. However, the current macroscopic continuum model gives no information on the individual particles. In this work, a new approach based on the discrete element method was introduced to consider the interaction between particles under fluid flow in accordance with the arrangement and properties of individual particles. We used an Euler–Lagrange method to precisely understand the influence of the reaction conditions on the behavior of coke and ore particles in three dimensions. The heterogeneity of the reaction rate and temperature distribution was observed to be influenced by the particle arrangement. The endothermic and exothermic reactions influenced each other in the packed bed. Temperature distributions nearly correlated with the gas velocity distribution because convection processes greatly affected the reaction rate. Although convection heat transfer was not a dominant issue in the packed bed, promotion of the reaction by a gas flow was effective.














Similar content being viewed by others
Abbreviations
- A :
-
Surface area (m 2)
- a :
-
Rate coefficient of multi-interface unreacted core model (m2 Pa mol/kJ)
- B :
-
Fluid drag coefficient (–)
- C p :
-
Specific heat capacity kJ/(kg K)
- c :
-
Mol concentration (mol/m3)
- D :
-
Mass transfer constant in a particle (m/s)
- d :
-
Particle diameter (m)
- \( \varvec{F} \) :
-
Force (N)
- f :
-
Catalytic factor (–)
- \( \varvec{g} \) :
-
Gravity (m/s2)
- H :
-
Enthalpy (kJ/mol)
- h :
-
Heat transfer coefficient
- I :
-
Moment of inertia (kg m2)
- K :
-
Equilibrium constant (–)
- k :
-
Rate coefficient (m/s)
- l :
-
Relative displacement increment of particle (m)
- M :
-
Molecular weight (g/mol)
- m :
-
Mass (kg)
- N :
-
Particle number (–)
- p :
-
Pressure (Pa)
- Q :
-
Quantity of heat (kJ/s)
- R :
-
Reaction rate (mol/s)
- r :
-
Radius (m)
- S :
-
Generation rate (kg/s)
- T :
-
Temperature (K)
- t :
-
Time (s)
- V :
-
Volume (m3)
- \( \varvec{v} \) :
-
Velocity (m/s)
- w :
-
Overall reduction degree of ore (–)
- x :
-
Mole fraction (–)
- α :
-
Particle–fluid momentum exchange coefficient (kg/(m3 s))
- β :
-
Heat transfer conductance (kJ/(K s))
- Γ :
-
Diffusion coefficient (m2/s)
- ɛ :
-
Void fraction (–)
- η :
-
Dashpot coefficient (–)
- θ :
-
Stefan–Boltzmann constant (5.67 × 10−8 W m−2 K−4)
- λ :
-
Heat conductivity (W/(m K))
- μ :
-
Frictional coefficient (–)
- ν:
-
Stoichiometric coefficient (–)
- ρ :
-
Density (kg/m3)
- σ :
-
Collision diameter (10−10 m)
- τ :
-
Spring coefficient (N/s)
- ϕ :
-
Emissivity (–)
- ψ :
-
Mass fraction (–)
- \( \varvec{\omega} \) :
-
Angular velocity (/s)
- Ω:
-
Collision integral (–)
- b:
-
Bed
- c:
-
Chemical reaction
- ct:
-
Contact
- cd:
-
Conduction
- D:
-
Mass transfer
- e:
-
Effective
- f:
-
Fluid
- I:
-
Chemical species
- i :
-
Index of particle
- j :
-
Vicinity particle index of particle i
- k:
-
Number of interface
- l:
-
Index of chemical reaction equation
- n:
-
Normal direction
- p:
-
Particle
- R:
-
Reaction
- r:
-
Rotational
- s:
-
Shear direction
- NU :
-
Nusselt number
- Pr :
-
Prandtl number
- Re :
-
Reynolds number
- Sc :
-
Schmidt number
- Sh :
-
Sherwood number
- Φ :
-
Thiele number
References
H. Nogami, Y. Ueki, T. Murakami and S. Ueda: Tetsu-to-Hagané, 2014, vol. 100, pp. 227–45.
M. Chu, H. Nogami and J. Yagi: ISIJ Int., 2004, vol. 44, pp. 801–8.
H. Nogami, S. Kitamura, J. Yagi and P.R. Austin: ISIJ Int., 2006, vol. 46, pp. 1759–66.
H. Nogami, Y. Kashiwaya and D. Yamada: ISIJ Int., 2012, vol. 52, pp. 1523–27.
A. Rist and N. Meysson: J. Met., 1967, vol. 19, pp. 50–9.
J. Yagi : ISIJ Int., 1991, vol. 31, 387–94.
X.F. Dong, A.B. Yu, S.J. Chew and P. Zulli: Metall. Mater. Trans. B, 2010, vol. 41B, pp. 330–49.
J.A. Castro, H. Nogami, and J. Yagi: ISIJ Int., 2002, vol. 42, pp. 44–52.
P.R. Austin, H. Nogami and J. Yagi: ISIJ Int., 1997, vol. 37, pp. 458–67.
P.R. Austin, H. Nogami and J. Yagi: ISIJ Int., 1997, vol. 37, pp. 748–55.
B. Wright, P. Zulli, Z.Y. Zhou, A.B. Yu: Powder Technol., 2011, vol. 208, pp. 86–97.
P.D. Cundall and O.D.L. Strack: Geotechnique, 1979, vol. 29, pp. 47–65.
Y. Yu and H. Saxén: ISIJ Int., 2012, vol. 52, pp. 788–96.
A.T. Adema, Y. Yang and R. Boom: ISIJ Int., 2010, vol. 50, pp. 954–61.
S. Natsui, H. Nogami, S. Ueda, J. Kano, R. Inoue and T. Ariyama: ISIJ Int., 2011a, vol. 51, pp. 41–50.
S. Natsui, H. Nogami, S. Ueda, J. Kano, R. Inoue and T. Ariyama: ISIJ Int., 2011b, vol. 51, pp. 51–8.
Z.Y. Zhou, H.P. Zhu, B. Wrighta, A.B. Yu and P. Zulli: Powder Technol., 2011, vol. 208, pp. 72–85.
Z.Y. Zhou, H.P. Zhu, A.B. Yu and P. Zulli: ISIJ Int., 2011, vol. 50, pp. 515–23.
S. Natsui, T. Kon, S. Ueda, J. Kano, R. Inoue, T. Ariyama and H. Nogami: Tetsu-to-Hagané, 2012, vol. 98, pp. 341–50.
S. Natsui, R. Shibasaki, T. Kon, S. Ueda, R. Inoue and T. Ariyama: ISIJ Int., 2013, vol. 53, pp. 1770–78.
H. Zhou, G. Flamant and D. Gauthier: Chem. Eng. Sci., 2004, vol. 59, pp. 4193–203.
H. Zhou, G. Flamant and D. Gauthier: Chem. Eng. Sci., 2004, vol. 59, pp. 4205–15.
Y. Kaneko, T. Shiojima and M. Horio: Chem. Eng. Sci., 1999, vol. 54, pp. 5809–21.
T. Sakurai, T. Minami, T. Kawaguchi, T. Tsuji, T. Tanaka and Y. Tsuji: Trans. Jpn Soc. Mech. Eng. Ser. B, 2009, vol. 75, pp. 1041–48.
W.L. Vargas and J.J. McCarthy: Int. J. Heat Mass Transfer, 2002, vol. 45, pp. 4847–56.
N. Wakao, S. Kaguei and T. Funazkri: Chem. Eng. Sci., 1979, vol. 34, pp. 325–36.
W.E. Ranz and W.R. Marshall, Jr.: Chem. Eng. Prog., 1952, vol. 48, pp. 141–6, 173–80.
H. Nogami, T. Yamamoto and K. Miyagawa: Tetsu-to-Hagané, 2010, vol. 96, pp. 319–27.
N. Wakao and T. Funazkri: Chem. Eng. Sci., 1978, vol. 33, pp. 1375–384.
T. Usui, M. Naito, T. Murayama and Z. Morita: Tetsu-to-Hagané, 1994, vol. 80, pp. 431–9.
O. Levenspiel: Chemical Reaction Engineering, 3rd ed., Wiley, New York, 1999, pp. 385–8.
S. Kobayashi and Y. Omori: Tetsu-to-Hagané, 1977, vol. 63, pp. 1081–9.
N. Miyasaka and S. Kondo: Tetsu-to-Hagané, 1968, vol. 54, pp. 1427–31.
R.H. Spitzer, F.S. Manning and W.O. Philbrook: Trans. Metall. Soc. AIME, 1966, vol. 236, pp. 1715–24.
Y. Hara, M. Sakawa and S. Kondo: Tetsu-to-Hagané, 1976, vol. 62, pp. 315–23.
R. Takahashi, Y. Takahashi, J. Yagi and Y. Omori: Ironmaking Conf. Proc., 1983, vol. 43, pp. 485–500.
O. Kubaschewski and C.B. Alcock: Metallurgical Thermochemistry, 5th ed., Pergamon, London, 1979.
R.B. Bird, W.E. Stewart, and E.N. Lightfoot: Transport Phenomena, 2nd ed., Wiley, New York, 1976, pp. 23, 274, 525, 866.
B.E. Poling, J.M. Prausnitz, and J.P. O’Connell: The Properties of Gases and Liquids, 5th ed., McGraw-Hill, New York, 2000, chap. 11.6, B.1.
C.R. Wilke: J. Chem. Phys., 1950, vol. 18, pp. 517–9.
S. Natsui, S. Ueda, H. Nogami, J. Kano, R. Inoue and T. Ariyama: Chem. Eng. Sci., 2012, vol. 71, pp. 274–82.
S. Ergun: Chem. Eng. Prog., 1952, vol. 48, pp. 89–94.
C.Y. Wen and Y.H. Yu (1966): Chem. Eng. Prog. Symp. Series, vol.62, 100–111.
Y. Tsuji, T. Kawaguchi and T. Tanaka: Powder Technol., 1993, vol.77, pp.79–87.
Acknowledgments
A part of this research was supported by Steel Foundation for Environmental Protection Technology (SEPT) of Japan.
Author information
Authors and Affiliations
Corresponding author
Additional information
Manuscript submitted March 8, 2014.
Appendix
Appendix
DEM–CFD Combining Model for Particle–Fluid Motion
The DEM–CFD model is briefly described here for completeness. The same procedure was used in previous research.[15,16,44] The governing equations for the translational and rotational motion of particle i can be described as
where \( m_{\text{p}} \) is the particle mass, \( \varvec{v}_{\text{p}} \) is the particle velocity, \( t \) is time, \( \varvec{F}_{\text{c}} \) is the particle–particle contact force, \( N_{\text{c}} \) is the contact particle number, \( \varvec{F}_{{f}} \) is the particle–fluid interaction force, \( \varvec{F}_{\text{g}} \) is the gravity force, \( I_{\text{p}} \) is the moment of inertia, \( \varvec{\omega}_{\text{p}} \) is the angular velocity, \( r_{\text{p}} \) is the particle radius, \( \varvec{F}_{\text{cn}} \) is the normal component of the contact force, \( \varvec{F}_{\text{cs}} \) is the tangential component of contact force, and \( \varvec{F}_{\text{r}} \) is the rolling friction force. The interparticle contact force was calculated by the “soft sphere” assumption based on Hertz’s contact theory:
where \( \tau \) is the spring coefficient, \( \eta \) is the dashpot coefficient, \( l \) is the relative displacement increment at a discrete time \( \Delta t \), and \( \mu \) is the frictional coefficient. Refer to previous article for further information to calculate \( \tau \), \( \eta \), and \( \varvec{F}_{r} \) [15].
The motion of the continuum fluid is calculated from the equation of continuity and Navier–Stokes equation based on local mean variables over a calculational cell.
where \( \varepsilon \) is the void fraction, ρ g is the fluid density, v g is the fluid velocity, \( p_{\text{g}} \) is the fluid pressure, and \( \mu_{\text{g}} \) is the fluid viscosity. The void fraction \( \varepsilon \) was calculated from the proportion of the solid existing within a local cell volume (Natsui et al.[15]). To determine \( \varvec{F}_{\text{p}} \), Ergun’s equation (1952)[42] was applied in the low-void fraction region \( \left( {\varepsilon < 0.8} \right) \), and Wen-Yu’s equation (1966)[43] was applied in the high-void fraction region \( \left( {\varepsilon > 0.8} \right) \):
where \( \alpha \) is the particle–fluid momentum exchange coefficient. The drag force coefficient was derived from the Schiller–Naumann equation expressed as a function of the particle Reynolds number[44]:
F f is distributed to particles as a reaction force of F p:
Rate Coefficient of the Multi-interface Unreacted Core Model
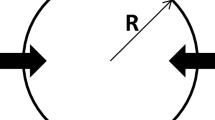
A gas reduction reaction occurs at the interface of a particle, and thus iron ore can be applied to the unreacted core model.[34,35] In this research, the temperature range was assumed to be T > 848 K (575 °C), and the composition of iron oxide changes with the degree of oxidation; i.e., Fe2O3 (hematite), Fe3O4 (magnetite), FeO (wüstite), Fe (iron). Since different reduction reactions occur on the inside and outside of a particle, it was necessary to set up a multi-reaction interface (see Figure 2). Table II shows the rate coefficients of the multi-interface unreacted core model in each interface. Here, in this section, A is the chemical reaction resistance, k c is the chemical reaction rate coefficient, B is the inner particle diffusion resistance, D e is the inner particle effective diffusion coefficient, F is the surface mass transfer resistance, k F is the surface mass transfer coefficient, R is the reduction degree, W is the ore flux, Y is the mol fraction of gaseous component, and K is the equilibrium constant.
Rights and permissions
About this article
Cite this article
Natsui, S., Kikuchi, T. & Suzuki, R.O. Numerical Analysis of Carbon Monoxide–Hydrogen Gas Reduction of Iron Ore in a Packed Bed by an Euler–Lagrange Approach. Metall Mater Trans B 45, 2395–2413 (2014). https://doi.org/10.1007/s11663-014-0132-x
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11663-014-0132-x