Abstract
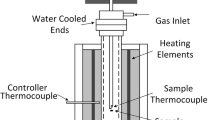
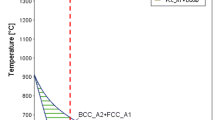
The nucleation potency of iron oxides was verified experimentally through nucleation undercooling of liquid iron using aerodynamic levitation technology for minimized container contaminations. Steady undercooling values were subsequently obtained from multiple melting and freezing thermal cycles, with the average undercooling values of 223 K ± 3 K and 75 K ± 6 K (223 °C ± 3 °C and 75 °C ± 6 °C) for FeO-contained liquid and Fe3O4-contained liquid, respectively. The statistical results showed a negligible difference in the sizes and numbers of particles between FeO and Fe3O4 particles, indicating that the nucleation potency difference is attributed to the nature of nucleants rather than particle size or numbers. Furthermore, high-resolution transmission electron microscopy analysis showed that the potential nucleation interfaces can be assumed as \( \{ 1 1 0\}_{{\delta {\text{{-}Fe}}}} //( 0 0\bar{2})_{\text{FeO}} \) and \( \{ 1 1 2\}_{{\delta {\text{{-}Fe}}}} //(\bar{2} 0 2 )_{{{\text{Fe}}_{3} {\text{O}}_{4} }} \), based on the detected exposed crystal planes of the oxide particles. Both the interfaces have relatively large values of lattice misfit, consistent with the experimentally measured undercooling based on Turnbull’s lattice matching theory.








Similar content being viewed by others
References
B. Pujilaksono, T. Jonsson, M. Halvarsson, J. Svensson and L. Johansson: Corros. Sci., 2010, vol. 52, pp. 1560–69.
J. Yuan, W. Wang, S. Zhu and F. Wang: Corros. Sci., 2013, vol. 75, pp. 309–17.
H. Li, A. Mclean, J.W. Rutter and I.D. Sommerville: Metal. Trans. B (Process Metallurgy), 1988, vol. 19B, pp. 383–95.
T. Suzuki, J. Inoue and T. Koseki: ISIJ Int., 2007, vol. 47, pp. 847–52.
Y. Nuri, T. Ohashi, T. Hiromoto and O. Kitamura: Transaction ISIJ, 1982, vol. 22, pp. 399–407.
K. Nakajima, H. Hasegawa, S. Khumkoa and S. Mizoguchi: Metall. Mater. Trans. B, 2003, vol. 34, pp. 539–47.
K. Zhu and Z. Yang: J. Mater. Sci. Technol., 2011, vol. 27, pp. 252–56.
Ø. Grong, A.O. Kluken, H.K. Nylund, A.L. Dons and J. Hjelen: Metall. Mater. Trans. A, 1995, vol. 26, pp. 525–34.
A. Hunter and M. Ferry: Metall. Mater. Trans. A, 2002, vol. 33, pp. 1499–507.
Y. Kang, S. Jeong, J. Kang and C. Lee: Metall. Mater. Trans. A, 2016, vol. 47A, pp. 2842–54.
T.E. Quested and A.L. Greer: Acta Mater., 2004, vol. 52, pp. 3859–68.
Z. Fan, Y. Wang, M. Xia and S. Arumuganathar: Acta Mater., 2009, vol. 57, pp. 4891–4901.
Y. Wang, Z. Fan, X. Zhou and G.E. Thompson: Phil. Mag. Lett., 2011, vol. 91, pp. 516–29.
J. Du, Y. Shi, M. Zhou and W. Li: J. Mater. Sci. Technol., 2016, vol. 32, pp. 1297–1302.
L. Wang, L. Yang, D. Zhang, M. Xia, Y. Wang and J.G. Li: Metall. Mater. Trans. A, 2016, vol. 47A, pp. 5012–22.
L. Wang, W. Lu, Q. Hu, M. Xia, Y. Wang and J. Li: Acta Mater., 2017, vol. 139, pp. 75–85.
B.L. Bramfitt: Metall. Trans., 1970, vol. 1, pp. 1987–95.
D. Turnbull and B. Vonnegut: Ind. Eng. Chem., 1952, vol. 44, pp. 1292–98.
M.X. Zhang, P.M. Kelly, M. Qian and J.A. Taylor: Acta Mater., 2005, vol. 53, pp. 3261–70.
B.B. He, B. Hu, H.W. Yen, G.J. Cheng, Z.K. Wang, H.W. Luo and M.X. Huang: Science, 2017, vol. p. n177.
G.W. Lee, S. Jeon and D. Kang: Cryst. Growth Des., 2013, vol. 13, pp. 1786–92.
G. Wille, F. Millot and J.C. Rifflet: Int. J. Thermophys., 2002, vol. 23, pp. 1197–1206.
A.L. Greer, A.M. Bunn, A. Tronche, P.V. Evans and D.J. Bristow: Acta Mater., 2000, vol. 48, pp. 2823–35.
A. Karasev and H. Suito: Metall. Mater. Trans. B, 1999, vol. 30, pp. 259–70.
M. Akimitsu, T. Mizoguchi and S. Kimura: J. Phys. Chem. Solids, 1983, vol. 44, pp. 497–505.
S. Mitra, P. Poizot, A. Finke and J.M. Tarascon: Adv. Funct. Mater., 2006, vol. 16, pp. 2281–87.
T. J. Ahrens: Mineral physics & crystallography: a handbook of physical constants, American Geophysical Union, Washington DC, 1995.
T. Mizoguchi and J.H. Perepezko: Mat. Sci. Eng. A-Struct., 1997, vol. 226, pp. 813–17.
B. Cantor: Philos. T. R. Soc. A, 2003, vol. 361, pp. 409–16.
H.T. Li, Y. Wang and Z. Fan: Acta Mater., 2012, vol. 60, pp. 1528–37.
D. Zhang, L. Wang, M. Xia, N. Hari Babu and J.G. Li: Mater. Charact., 2016, vol. 119, pp. 92–98.
Z. Chen, H. Kang, G. Fan, J. Li, Y. Lu, J. Jie, Y. Zhang, T. Li, X. Jian and T. Wang: Acta Mater., 2016, vol. 120, pp. 168–78.
Acknowledgments
The authors are grateful for the financial support of the National Natural Science Foundation of China (Nos. 51474148 and 51727802), the Shanghai International Cooperation Project (Grant No. 14140711000), and the Joint Funds of the National Natural Science Foundation of China (No. U1660203).
Author information
Authors and Affiliations
Corresponding author
Additional information
Manuscript submitted 22 October, 2017.
Rights and permissions
About this article
Cite this article
Xu, M., Wang, L., Lu, W. et al. The Nucleation Potency of In Situ-Formed Oxides in Liquid Iron. Metall Mater Trans A 49, 1762–1769 (2018). https://doi.org/10.1007/s11661-018-4528-0
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11661-018-4528-0