Abstract
Oxidative stress, caused by both endogenous and exogenous factors, affects sperm function by damaging morphology and reducing metabolic activity, leading to reduced fertilization ability. The purpose of this study was to investigate the effects of oxidative stress on bull sperm and to evaluate the efficacy of targeted antioxidants in mitigating these detrimental effects. Fresh bull semen samples were subjected to hydrogen peroxide (H2O2) and antimycin treatments to induce oxidative stress, and the antioxidants PQQ, ergothioneine, and vitamin C were applied to counteract the induced stress. Sperm motility, viability, and reactive oxygen species (ROS) levels in the cytoplasm and mitochondria of sperm were assessed using computer-assisted sperm analysis (CASA) and flow cytometry. The treatment with H2O2 rapidly decreased sperm viability, and antimycin-induced mitochondrial ROS mainly decreased sperm motility; PQQ and vitamin C effectively reduced mitochondrial ROS, while ergothioneine and vitamin C reduced cytosolic ROS. In frozen-thawed sperm, oxidative stress was elevated in both cytoplasm and mitochondria, and all three antioxidants improved sperm motility by inhibiting ROS production. Furthermore, the localization of oxidized lipids (4-hydroxynonenal) in sperm was detected using immunofluorescence, indicating that oxidative stress affects the head and midpiece of sperm. These findings highlight the potential of targeted antioxidants to mitigate the detrimental effects of oxidative stress on bull sperm and provide valuable insights to improve semen quality and optimize the use of antioxidants in artificial insemination.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Artificial insemination in bovine is an important tool for improving reproductive efficiency, and using high-quality semen directly contributes to the productivity and economics of the livestock industry. One of the known factors in the decline of sperm quality is oxidative stress (Simões et al. 2013; Aitken 2020). The oxidative stress not only damages important components of the spermatozoa, such as the axon part of the tail, acrosome, and midpiece, but also negatively affects sperm function by impairing the metabolic activity involved in ATP production in both the mitochondria and cytoplasm (Aitken et al. 1998; Moazamian et al. 2015). Oxidative stress triggers apoptosis in the spermatozoa, leading to DNA fragmentation (Lewis and Aitken 2005; Aitken and Baker 2006; Bansal and Bilaspuri 2011; Agarwal et al. 2014b). Essentially, oxidative stress not only diminishes the sperm's capacity to fertilize oocytes both in vitro and in vivo, but also impedes zygote development caused due to chromosomal abnormalities.
Reactive oxygen species (ROS) generated by both endogenous and exogenous factors oxidize various biomolecules, including lipids, proteins, and nucleic acids. These oxidative processes compromise the functionality and stability of these biomolecules and are collectively termed as oxidative stress (Morielli and O’Flaherty 2015; Panner Selvam et al. 2020). Endogenously, ROS are byproducts of ATP synthesis in the mitochondrial electron transport chain. Normally, ROS are neutralized by the glutathione reduction pathway. However, under conditions of rapid ATP production, ROS generation surpasses the capacity for reduction, thereby exacerbating oxidative stress within the mitochondria and cytoplasm. In addition, mitochondrial damage caused by excessive calcium influx disrupts the electron transport chain resulting in ROS production (Aitken et al. 2016). Since sperm motility depends on ATP synthesis, dysfunction of mitochondria, which are the ATP synthesis organs, significantly impairs sperm motility (Bulkeley et al. 2021). Although addition of substrates, such as glucose and fatty acids, temporarily boosts motility, sustained ATP overproduction exacerbates oxidative stress, ultimately diminishing sperm motility (Zhu et al. 2019; Islam et al. 2021).
Extracellular factors can also induce oxidative stress in sperm. Leukocyte-derived ROS, produced in response to bacterial infections in semen, eradicate bacteria, however, inadvertently damage the sperm (Agarwal and Said 2005; Agarwal et al. 2014a; Fraczek et al. 2016). Cryopreservation-induced fluctuations in osmotic pressure and temperature also generate ROS both inside and outside the cells, altering sperm lipids and protein molecules (John Morris et al. 2012; Pini et al. 2018). Moreover, organelle damage during freezing and thawing releases ROS into the cytoplasm, promoting lipid peroxidation and impairing sperm motility, viability, and function (Chatterjee et al. 2001; Chatterjee and Gagnon 2001).
Various antioxidants naturally present in the seminal plasma mitigate oxidative stress, constituting primary defense against free radical-induced damage (Lewis et al. 1997; Bansal and Bilaspuri 2011; Sharma et al. 2013). However, in frozen-thawed semen, antioxidant activity may be lesser due to semen dilution or the removal of seminal plasma. Consequently, addition of exogenous antioxidants is necessary to mitigate oxidative stress. Small-molecule antioxidants include vitamin C, vitamin E, glutathione, ubiquinol, coenzyme Q10, PQQ, and peroxiredoxins (PRDXs), whereas large-molecule antioxidants include superoxide dismutase (SOD), catalase, glutathione peroxidase (GPx), and albumin. These antioxidants exhibit varying mechanisms of action, necessitating investigations into the factors that effectively counteract specific oxidative stress to ensure sperm normality (O’Flaherty and Scarlata 2022). Therefore, the aim of this study was to elucidate the antioxidant efficacy of fresh bull semen samples treated with hydrogen peroxide and antimycin against endogenous and exogenous oxidative stress. Additionally, the potential applicability of these findings in improving the viability of freeze-thawed bull semen will be explored.
Materials and Methods
Semen collection and incubation of sperm
The Livestock Improvement Association of Japan, INC (Tokyo, Japan) provided fresh semen from Japanese breeding bulls that was collected using an artificial vagina and diluted with TRIS-egg yolk extender. The fresh semen was placed in thermos bottles and transported to the laboratory within 4 h at 4°C. The semen sample was washed twice through centrifugation (400 × g for 3 min) using the modified Human Tubal Fluid (mHTF) medium (Umehara et al. 2020). After centrifugation, the sperm pellet was resuspended in mHTF medium containing pyrroloquinoline quinone (PQQ) disodium salt (BioPQQ®; Mitsubishi Gas Chemical Co., Inc., Tokyo, Japan), ergothioneine (14905, Cayman, Ann Arbor, MI) or vitamin C (A5960, Sigma–Aldrich, St Louis, MO), and was used for all analyses. Some samples were treated with H2O2 (20779-65, Nacalai Tesque, Osaka, Japan) or antimycin (A8674; Sigma) to examine the effects of ROS induction. The samples were incubated at 37°C in a humidified atmosphere containing 5% CO2.
The frozen bull semen was a gift from the Livestock Improvement Association of Japan, Inc. The 0.5 ml straw of frozen semen was thawed in water at 37°C for 30 s and immediately diluted with 6 ml of mHTF medium. In the first thawing medium, 1 µM PQQ, 100 µM ergothioneine or 1000 µM vitamin C was added to the mHTF medium. The first thawing medium containing frozen-thawed sperm was centrifuged at 300 g (5 min at 37°C), and the sperm pellet was washed twice with each medium. After centrifugation, the sperm pellet was resuspended in each medium and used for all analyses. The samples were incubated at 37°C in a humidified atmosphere containing 5% CO2.
Table 1 lists information on the bulls used in the experiment. Sperm motility was assessed before every experiment, and all experiments were performed when a percentage of motile showed motility over 60%.
Assessment of sperm motility
Sperm motility was evaluated using computer-assisted sperm analysis (CASA), as described in our previous study (Islam et al. 2021). Approximately, 3 µL of sample was placed in a pre-warmed counting chamber for CASA after incubating the sperm at different time intervals. Sperm tracks (0.5 s and 45 frames) were captured at 60 Hz according to our previous study using the CASA system (HT CASA-Ceros II; Hamilton Thorne, Beverly, MA). A minimum of 200 spermatozoa were assessed to carry out each CASA. Single sperm motility was calculated by multiplying motility by the total concentration.
Determination of ROS by flow cytometry
Photooxidation-resistant dichloro-dihydro-fluorescein diacetate (DCFH-DA) is a total ROS (H2O2, superoxide anion, and hydroxyl radical) probe (R253; DOJINDO LABOLATORIES Co., Ltd., Kumamoto, Japan). According to the manufacturer’s protocols, sperm was pretreated with a photo-oxidation-resistant DCFH-DA working solution and incubated for 30 min at 37°C in a humidified atmosphere of 5% CO2. The sperm was washed twice with mHTF. After removing the supernatant, the sperm was mixed with mHTF-containing antioxidants (PQQ, ergothioneine, or vitamin C) and/or inducers of oxidative stress (H2O2 or antimycin). After 0, 1, 2, 3 and 4-h of incubation, the samples were analyzed using a flow cytometer (Attune®NxT Acoustic Focusing Cytometer, Thermo Fisher Scientific Inc., Waltham, MA). Green fluorescence (DCF) was evaluated using a 488 nm laser and a 530/30 nm bandwidth filter. Data are expressed as the percentage of fluorescent-positive sperm. Cutoff values were set using an unstained sample. The gating strategy is illustrated in Supplementary Fig. S1. Localization of fluorescence in the sperm was determined using an APX100 Digital Imaging System (EVIDENT Co., Ltd., Tokyo, Japan).
Mitosox deep red assay
Generation of mitochondrial superoxide anion was investigated according to the MitoSOX™ deep Red Assay Kit (MT-14, DOJINDO LABORATORIES). The sperm was pretreated with MitoSOX™ deep Red working solution, incubated at 37°C for 30 min and washed twice with mHTF. After removing the supernatant, the sperm was mixed with mHTF-containing antioxidants (PQQ, ergothioneine, or vitamin C) and/or inducers of oxidative stress (H2O2 or antimycin). The samples were analyzed by flow cytometry after 0, 1, 2, 3 and 4-h of incubation. Fluorescence was evaluated using a 638 nm laser and a 570/14 nm bandwidth filter. Data are expressed as the percentage of fluorescent-positive sperm. Cutoff values were set using an unstained sample. The gating strategy is illustrated in supplementary Fig. S1. Localization of fluorescence in the sperm was determined using a Nikon AX confocal microscope (Nikon Solutions Co., Ltd., Tokyo, Japan).
Mitochondrial activity
Mitochondrial activity of sperm was measured using a MitoPT® JC-1 Assay Kit (911, Immuno Chemistry Technologies, LLC, Bloomington, MN) according to our previous study. Briefly, sperm were incubated with 200 µL of working solution containing 5,5’,6,6’-tetrachloro-1,1’,3,3’-tetraethylbenzimidazolyl carbocyanine iodide (JC-1) dye at 37°C for 30 min in the dark. The sperm suspension was centrifuged and washed twice with mHTF medium. After washing, the sperm pellet was resuspended in mHTF medium and analyzed through flow cytometry using a 488 nm laser and filters with bandwidths of 530/30 and 574/26 nm. Data are expressed as the percentage of fluorescent-positive sperm. The gating strategy is illustrated in supplementary Fig. S1. A total of 50,000 sperm events were analyzed.
Immunofluorescence
Sperm was mounted on glass slides, air-dried, fixed with 4% paraformaldehyde for 30 min, and permeabilized with 0.3% (v/v) Triton X-100 in PBS for 30 min at room temperature. After washing with PBS, samples were incubated with blocking solution from the MOM Kit (MKB-2213, Vector Laboratories, Newark, CA) at 25℃ for 30 min to block nonspecific sites. The samples were then incubated at 4℃ overnight with primary mouse antibody, anti-4 hydroxynonenal antibody (4HNE; 1:100; ab48506; Abcam, Cambridge, UK). After washing with PBS, the antigens were visualized using Cy3-conjugated sheep anti-mouse IgG (1:100; C2181, Sigma). Digital images were captured using an APX100 Digital Imaging System.
Statistical analysis
At least three animals were used for the fresh semen study, and the experiments were replicated at least three times in each group. Five animals were used in the frozen semen study. Quantitative data were presented as mean ± SEM. Percentage data were subjected to arcsine transformation before statistical analysis. Motile single-sperm data were analyzed using a paired t-test with p-value correction using the Bonferroni method for multiple tests. Differences between groups were assessed using one-way analysis of variance (ANOVA). When the ANOVA was significant, differences among values were analyzed using Tukey's honest significant difference test for multiple comparisons. Dunnett's test was used to analyze the ROS inducer/antioxidant combination (Fig. 4) and frozen sperm experiments (Fig. 5). The comparison controls are listed in the figure captions below. R (version 4.3.1) was used for statistical analysis. Statistical value of p<0.05 was defined as a significant difference.
Results
Changes in motility and intracytoplasmic and mitochondrial ROS levels of fresh sperm with incubation time
The number of motile single sperm and ALH (µm) significantly decreased from 0 to 4 h of incubation (Fig. 1A and B). The VCL (µm/sec) did not differ from 0 to 4 h of incubation (Fig. 1B). However, in VSL (µm/sec) highly significant differences were found from 0 to 4 h of incubation (Fig. 1B). The ROS levels in the cytoplasm and mitochondria of fresh samples were measured using two probes (DCFH-DA and mtSOX deep red). The total ROS levels significantly increased in a time-dependent manner in fresh sperm from 0 to 4 h of incubation (Fig. 1C). However, mitochondrial ROS levels increased sharply for up to 1 h of incubation and remained significantly higher for up to 4 h. Thus, both total ROS levels and mitochondrial ROS levels increased in different patterns with incubation, and cell viability greatly reduced on incubation. However, the relationship between them remains unclear.
Significant decrease in viability and an increase in ROS levels of fresh bull sperm during the incubation period. (A) Tracks of sperm incubated for 0 and 4 h were determined using the CASA system. Red arrows indicate the aggregated sperm, which is the cause of overestimating CASA parameters because it lowers the number of dead sperm. (B) Changes in fresh sperm motility single counts implying sperm viability and in kinetics patterns with incubation time. (C and D) Time-dependent changes in total ROS in the cytoplasm or mitochondrial superoxide level of fresh bull sperm detected using DCFH-DA or mtSOX deep Red, respectively. Flow cytometric patterns and percentage of positive sperm were shown. Different letters represent significantly different groups (p < 0.05).
Induced oxidative stress in fresh semen using H2O2 and antimycin
To elucidate the relationship between the three factors that were unknown in the above experiments, fresh bull sperm was treated with two different oxidative stress inducers, hydrogen peroxide or antimycin (Fig. 2A). After one hour of incubation, sperm motility and ROS levels were examined using DCFH-DA and mtSOX deep probes. Percent of positive sperm stained using DCFH-DA (total ROS positive) differed significantly from the control group on treatment with more than 50 µM of H2O2 (Fig. 2B-D). However, the motile single sperm percentage significantly reduced by treatment with more than 10 μM H2O2 (Fig. 2E). On the contrary, the motion speeds (VSL and VCL) significantly reduced at concentrations of 50 μM or more (Fig. 2E). The percentage of mtSOX deep red-positive sperm significantly increased on treatment with antimycin in a dose-dependent manner (Fig. 2F and G). Although the motile single sperm ratio slightly decreased due to the treatment, the VCL (µm/sec) and VSL (µm/sec) significantly reduced in a dose-dependent manner (Fig. 2H). Thus, the viability of sperm was sensitively reduced by treatment with hydrogen peroxide, whereas the motility rate of the sperm was reduced by an increase in endogenous ROS in the mitochondria of fresh bull sperm.
The impact of oxidative stress inducers, H2O2 and antimycin, on fresh bull sperm motility and ROS levels. (A) Experimental design to investigate the effect of the oxidative stress inducers on fresh bull sperm. (B) Representative images of the sperm fluorescence signal with DCFH-DA and mtSOX deep Red. Left: DCFH-DA fluorescence image of sperm treated with H2O2. Right: mtSOX deep Red fluorescence image of sperm treated with antimycin. (C) Flow cytometric patterns of DCFH-DA. (D) Percentages of DCFH-DA positive sperm. (E) Sperm kinetics changed by H2O2. (F) Flow cytometric patterns of mtSOX deep Red. (G) Percentages of mtSOX deep Red positive sperm. (H) Sperm kinetics changed by antimycin. Different letters represent significantly different groups (p < 0.05).
Effect of different antioxidants on total ROS and mitochondrial ROS levels under the presence of different oxidative stress inducers
The effects of the antioxidant factors, PQQ, ergothioneine, and vitamin C, on extracellular oxidative stress stimulation (H2O2) and mitochondrial oxidative stress were investigated in fresh bull sperm (Fig. 3A). The percentage of sperm showing an increase in total ROS after H2O2 treatment did not change significantly with PQQ treatment, however a significant dose-dependent decrease was observed in the presence of ergothioneine and vitamin C (Fig 3B and C). PQQ and vitamin C reduced the percentage of mtSOX deep red-positive sperms (ROS generated in mitochondria by antimycin treatment) in a dose-dependent manner (Fig. 3D and E). In contrast, there was no dose-dependent change in the ergothioneine levels (Fig. 3D and E).
Impact of antioxidants on total ROS and mitochondrial ROS levels in fresh bull sperm under oxidative stress inducers. (A) Experimental design to investigate the effect of three antioxidants on induced reactive oxygen species in fresh bull sperm. (B) Flow cytometric patterns of H2O2 and antioxidant-treated sperm stained with DCFH-DA. (C) Percentages of DCFH-DA positive sperm. (D) Flow cytometric patterns of antimycin and antioxidant-treated sperm stained with mtSOX deep Red. (E) Percentages of mtSOX deep Red positive sperm. Different letters represent significantly different groups (p < 0.05).
Effect of different antioxidants on sperm motility and ROS level under the presence of different oxidative stress inducers
The negative effect of H2O2 on sperm motility was not improved by addition of PQQ or ergothioneine, however a significant improvement was observed only with vitamin C (Fig. 4B). Ergothioneines, like vitamin C, significantly increased the motility rate of sperm, which was suppressed by H2O2 (Fig. 4B). In contrast, the negative effect of antimycin on sperm motility was ameliorated by antioxidant factors (Fig. 4B). However, none of the antioxidant factors affected the effect of antimycin on the sperm exercise speed (Fig. 4C). H2O2 treatment not only increased the percentage of total ROS-positive sperm, but also increased the percentage of mitochondrial ROS-positive sperm (Fig. 4D and E). Ergothioneine and vitamin C significantly reduced the percentage of total ROS-positive cells, whereas the percentage of mitochondrial ROS-positive sperm significantly reduced in the presence of all antioxidant factors (Fig. 4D and E). The effect of antimycin significantly increased not only the percentage of ROS-positive sperm in the mitochondria but also the percentage of total ROS-positive sperm, and the total ROS-positive sperm significantly reduced in the presence of all antioxidant factors (Fig. 4F and G). In measuring mitochondrial membrane potential using JC-1, H2O2 did not affect the high mitochondrial membrane potential (hMMP; Fig. 4H and I). On the other hand, antimycin significantly reduced the percentage of spermatozoa with hMMPs, but they were rescued by all antioxidant factors (Fig. 4J).
Impact of various antioxidants on fresh bull sperm motility and reactive oxygen species (ROS) levels when exposed to different oxidative stress triggers. (A) Experimental design to examine the impact of various antioxidants on sperm motility and levels of reactive oxygen species (ROS) under the two oxidative stress inducers. (B and C) Changes in sperm viability and motility under H2O2 (B) or antimycin (C). (D-G) Effect of antioxidants on the percentages of DCFH-DA and mtSOX deep Red positive sperm under H2O2 or antimycin. (H-J) Measurement of mitochondrial activity using the JC-1 kit. (H) Representative flow cytometric pattern of sperm that was not treated with any drug (control) stained with JC-1. Effect of antioxidants on the percentages of high mitochondrial membrane potential (hMMP) under the treatment of H2O2 (I) and antimycin (J). H2O2 or antimycin groups were used as controls for statistical analysis (p < 0.05).
Effect of antioxidants on frozen-thawed bull sperm motility and ROS levels
The effects of PQQ, which reduced ROS generation in mitochondria; vitamin C, which reduced both total ROS and ROS in mitochondria; and ergothioneine, which reduced the total level of ROS generated by H2O2 in frozen-thawed bull sperm were examined (Fig. 5A). The percentage of total ROS-positive sperm significantly reduced in the presence of vitamin C and ergothioneine, however not in the presence of PQQ (Fig. 5B). However, mitochondrial ROS-positive sperm significantly reduced in the presence of antioxidant factors (Fig. 5C). Thus, when frozen-thawed sperm was incubated, oxidative stress elevated in both the cytoplasm and mitochondria. When induction was suppressed by PQQ, vitamin C, or ergothioneine, sperm motility significantly increased in the presence of all antioxidants (Fig. 5D). In addition, a significant increase in total mobility speed (VCL) was observed, suggesting that the increase in reactive oxygen species due to both mitochondrial abnormalities and the thawing process in frozen-thawed sperm caused a decrease in sperm motility and mobility speed (Fig. 5D).
The effect of various antioxidants on frozen-thawed bull sperm motility and ROS levels. (A) Experimental design to examine the impact of PQQ, vitamin C, and ergothioneine on frozen-thawed bull sperm motility and ROS levels. Various antioxidants were included in all thawing, washing, and incubation processes using HTF medium as the basic medium. (B and C) Effect of antioxidants on the percentages of DCFH-DA (B) and mtSOX deep Red (C) positive sperm. (D) Changes in frozen-thawed sperm viability and kinetics patterns with antioxidants. Asterisks represent significant differences from the control (Ctrl; *: p < 0.05, **: p < 0.01, ***: p < 0.001).
Localization of oxidized lipids and the effects of antioxidant factors in fresh or frozen-thawed bull sperm
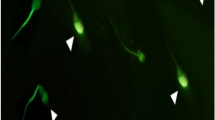
Localization of oxidized lipids was detected by immunostaining with anti-4-HNE antibodies. Positive 4-HNE signals were strongly detected not only in the middle piece after antimycin treatment but also in the sperm head. These positive signals were attenuated by antioxidant factors. Similarly, positive 4-HNE signals were detected in the sperm head and midpiece following H2O2 treatment. The 4-HNE positive signals were not abated by PQQ, however were suppressed by ergothioneine and vitamin C (Fig. 6A). These results show the same trend as the ROS-positive sperm detection results. Furthermore, from the results of the experiments with frozen-thawed sperm, strong signals were detected in the midpiece, which reduced in the presence of PQQ and vitamin C, and these were the same as in the detection experiment of ROS-positive sperm (Fig. 6B).
Localization of oxidized lipids in fresh or frozen-thawed bull sperm. (A, B) Representative images of the sperm fluorescence signal with 4-HNE. (A) The ROS inducer/antioxidant combination experiment in fresh sperm. (B) The effect of various antioxidants on frozen-thawed sperm. The scale bar represents 20 μm. (C) The graphical image shows the ROS production cascade in sperm beginning at the mitochondria. It also shows the site of action of the antioxidants used in this experiment.
Discussion
The results obtained from this study suggest that sperm function was inhibited due to oxidative stress in both the cytoplasm and mitochondria (Fig. 6C). In particular, it was thought that viability declined sharply due to oxidative stress caused by H2O2 from outside the cell, rather than from the mitochondria. In an experiment investigating lipid oxidation, strong signals were detected in the sperm head region after H2O2 treatment, consistent with a previous report showing that the normality of the acrosome cap is linked to sperm survival (Nagy et al. 2004; Fayyaz et al. 2017; Guidobaldi et al. 2017; Islam et al. 2023). In contrast, elevated ROS levels in the mitochondria reduced sperm viability. Mitochondrial dysfunction may lead to the release of large amounts of cytochrome c and calcium within the mitochondria, which may adversely affect sperm survival. In addition, oxidative stress in the mitochondria reduces sperm motility, even at levels that do not affect sperm viability. It has been reported that decreased sperm mobility was due to the oxidation of mitochondrial DNA, mitochondrial transcription factors, and mitochondrial RNA polymerase (Amaral et al. 2007; Zhu et al. 2019). In this study, the decrease in sperm motility rate was induced at concentrations where antimycin treatment did not significantly reduce sperm viability. This was due to the decrease in ATP synthesis in the mitochondria by the above mechanism. Thus, oxidative stress from outside the mitochondria rapidly reduces the viability of spermatozoa, whereas oxidative stress inside the mitochondria reduces mobility of the sperm and further reduces sperm survival due to severe stress.
In particular, strong signals of oxidized lipids, which were attenuated by PQQ and vitamin C, were detected in the midpiece region of frozen–thawed sperm. This suggests that excessive oxidative stress, mainly due to mitochondrial dysfunction, significantly reduces the viability of frozen-thawed sperm. It has been reported that abnormal mitochondrial membrane potentials are detected in frozen-thawed spermatozoa and morphological abnormalities in the mitochondria are observed using electron microscopy (Khalil et al. 2017). Functional and structural disruption of mitochondria by freezing and thawing increases oxidative stress. On the contrary, ergothioneine improved the viability of frozen-thawed sperm, as well as its positive effect on the decrease in sperm viability due to oxidative stress in the cytoplasm and cell membrane induced by H2O2 treatment. This indicates that increase in ROS caused by the rapidly increasing temperature and osmotic pressure during the thawing process is a factor that reduces the viability of frozen-thawed sperm. There are many reports of abnormalities in the sperm cell membrane of frozen-thawed spermatozoa in the normality test of the cell membrane using fluorescent staining reagents, which cannot normally pass through the cell membrane (Peña et al. 2003; Partyka et al. 2010). In other words, in frozen-thawed sperm, oxidative stress on the cell membrane during the thawing process and excessive ROS production due to mitochondrial destruction during incubation can significantly reduce sperm viability.
PQQ has been reported to act directly on the mitochondria by passing through the cell membrane to maintain mitochondrial function (Zhu et al. 2006; Rucker et al. 2009; Hoque et al. 2021). In fact, it has been reported that PQQ not only improves the stability of mitochondrial DNA in boar sperm, but also induces sustained ATP synthesis by improving gene expression from mitochondrial DNA and maintains increased motility for a long time (Zhu et al. 2019). Vitamin C is a water-soluble antioxidant that penetrates the cell membrane and exerts ROS-scavenging abilities in both the cytoplasm and mitochondria (Sönmez et al. 2005; Hu et al. 2010). Similarly, MitoQ and CoQ10 are antioxidant factors that are expected to act on the mitochondria. Although their detailed effects have not been analyzed, their positive effects on sperm have been reported (Zhu et al. 2019; Arjun et al. 2022). However, ergothioneine, although a water-soluble factor, can exert antioxidant effects not only outside the cell but also inside the cell because it crosses the blood-brain barrier and is taken into the cell via the transporter (OCTN1). OCTN1 is expressed in human spermatozoa, however, its localization has not yet been analyzed (Xuan et al. 2003). In this study, ergothioneine did not completely reverse the effect of ROS on the mitochondrial membrane potential induced by antimycin but improved sperm survivability. This suggests that ergothioneine was taken up into the sperm cytoplasm in bull sperm and could not efficiently penetrate the mitochondria, however exerted antioxidant functions in the cytoplasm.
Antioxidants maintain sperm motility, do not degrade sperm quality during handling, and increase sperm motility after freezing and thawing. However, when antioxidant factors are added, it is necessary to consider whether they are taken up by sperm. For example, membrane-impermeable antioxidants such as glutathione have been reported to improve sperm motility, however they are unlikely to have a direct effect (Sławeta and Laskowska 1987; Chatterjee et al. 2001; Ansari et al. 2012). Another important factor is the stability of the antioxidants. CoQ10 and vitamin C is easily oxidized in solution, decreasing its antioxidant function. Simplicity is also important in reproductive techniques used in animal production, such as artificial insemination, however, antioxidant factors must be mixed immediately before use. Such stability must be verified in the same manner as cell membrane permeability.
Oxidative stress in the testis has a direct negative impact on the spermatogenesis process (Aitken and Roman 2008; Aitken 2020). In addition, it has been reported that oxidative stress in semen increases with age (Vince et al. 2018; Baharun et al. 2021). In our study using boar sperm, with increasing time of sperm incubation, endogenous ROS was generated in the mitochondria, which reduced sperm motility (Zhu et al. 2019). These results indicate not only an artificial increase in exogenous and endogenous oxidative stress, including freezing and thawing, but also a decrease in sperm quality due to increased oxidative stress in vivo. The difference in the action of antioxidant factors revealed in this study is considered to be an important finding for treatment methods that improve spermatogenesis and semen quality deterioration in vivo. In fact, it has been reported that antioxidants improve spermatogenesis and sperm motility in aging males (Agarwal and Sekhon 2010; Leisegang et al. 2017; Ioannidou et al. 2022).
Thus, to prevent qualitative deterioration of sperm in bulls, it is essential to combine appropriate antioxidant factors according to the oxidative stress that occurs in each sperm compartment. The results obtained from this study provide important insights into the use of antioxidants in semen preparations for artificial insemination.
Data availability
Additional information needed to reanalyze the data reported in this paper is available from the primary contact upon request.
References
Agarwal A, Mulgund A, Alshahrani S et al (2014a) Reactive oxygen species and sperm DNA damage in infertile men presenting with low level leukocytospermia. Reprod Biol Endocrinol 12:126. https://doi.org/10.1186/1477-7827-12-126
Agarwal A, Said TM (2005) Oxidative stress, DNA damage and apoptosis in male infertility: a clinical approach. BJU Int 95:503–507. https://doi.org/10.1111/j.1464-410X.2005.05328.x
Agarwal A, Sekhon LH (2010) The role of antioxidant therapy in the treatment of male infertility. Hum Fertil Camb Engl 13:217–225. https://doi.org/10.3109/14647273.2010.532279
Agarwal A, Virk G, Ong C et al (2014b) Effect of Oxidative Stress on Male Reproduction. World J Mens Health 32:1–17. https://doi.org/10.5534/wjmh.2014.32.1.1
Aitken RJ (2020) Impact of oxidative stress on male and female germ cells: implications for fertility. Reprod Camb Engl 159:R189–R201. https://doi.org/10.1530/REP-19-0452
Aitken RJ, Baker MA (2006) Oxidative stress, sperm survival and fertility control. Mol Cell Endocrinol 250:66–69. https://doi.org/10.1016/j.mce.2005.12.026
Aitken RJ, Gibb Z, Baker MA et al (2016) Causes and consequences of oxidative stress in spermatozoa. Reprod Fertil Dev 28:1–10. https://doi.org/10.1071/RD15325
Aitken RJ, Gordon E, Harkiss D et al (1998) Relative Impact of Oxidative Stress on the Functional Competence and Genomic Integrity of Human Spermatozoa1. Biol Reprod 59:1037–1046. https://doi.org/10.1095/biolreprod59.5.1037
Aitken RJ, Roman SD (2008) Antioxidant systems and oxidative stress in the testes. Oxid Med Cell Longev 1:15–24. https://doi.org/10.4161/oxim.1.1.6843
Amaral A, Ramalho-Santos J, St John JC (2007) The expression of polymerase gamma and mitochondrial transcription factor A and the regulation of mitochondrial DNA content in mature human sperm. Hum Reprod 22:1585–1596. https://doi.org/10.1093/humrep/dem030
Ansari MS, Rakha BA, Andrabi SMH et al (2012) Glutathione-supplemented tris-citric acid extender improves the post-thaw quality and in vivo fertility of buffalo (Bubalus bubalis) bull spermatozoa. Reprod Biol 12:271–276. https://doi.org/10.1016/j.repbio.2012.10.001
Arjun V, Kumar P, Dutt R et al (2022) Effect of mitochondria-targeted antioxidant on the regulation of the mitochondrial function of sperm during cryopreservation. Andrologia 54:e14431. https://doi.org/10.1111/and.14431
Baharun A, Said S, Arifiantini RI et al (2021) Correlation between age, testosterone and adiponectin concentrations, and sperm abnormalities in Simmental bulls. Vet World 14:2124–2130. https://doi.org/10.14202/vetworld.2021.2124-2130
Bansal AK, Bilaspuri GS (2011) Impacts of Oxidative Stress and Antioxidants on Semen Functions. Vet Med Int 2011:1–7. https://doi.org/10.4061/2011/686137
Bulkeley EA, Foutouhi A, Wigney K et al (2021) Effects from disruption of mitochondrial electron transport chain function on bull sperm motility. Theriogenology 176:63–72. https://doi.org/10.1016/j.theriogenology.2021.09.015
Chatterjee S, de Lamirande E, Gagnon C (2001) Cryopreservation alters membrane sulfhydryl status of bull spermatozoa: Protection by oxidized glutathione. Mol Reprod Dev 60:498–506. https://doi.org/10.1002/mrd.1115
Chatterjee S, Gagnon C (2001) Production of reactive oxygen species by spermatozoa undergoing cooling, freezing, and thawing. Mol Reprod Dev 59:451–458. https://doi.org/10.1002/mrd.1052
Fayyaz MH, Ahmad M, Ahmad N (2017) Survival of buffalo bull spermatozoa: effect on structure and function due to alpha-lipoic acid and cholesterol-loaded cyclodextrin. Andrologia 49:e12652. https://doi.org/10.1111/and.12652
Fraczek M, Hryhorowicz M, Gill K et al (2016) The effect of bacteriospermia and leukocytospermia on conventional and nonconventional semen parameters in healthy young normozoospermic males. J Reprod Immunol 118:18–27. https://doi.org/10.1016/j.jri.2016.08.006
Guidobaldi H, Hirohashi N, Cubilla M et al (2017) An intact acrosome is required for the chemotactic response to progesterone in mouse spermatozoa. Mol Reprod Dev 84:310–315. https://doi.org/10.1002/mrd.22782
Hoque SAM, Umehara T, Kawai T et al (2021) Adverse effect of superoxide-induced mitochondrial damage in granulosa cells on follicular development in mouse ovaries. Free Radic Biol Med 163:344–355. https://doi.org/10.1016/j.freeradbiomed.2020.12.434
Hu J-H, Tian W-Q, Zhao X-L et al (2010) The cryoprotective effects of ascorbic acid supplementation on bovine semen quality. Anim Reprod Sci 121:72–77. https://doi.org/10.1016/j.anireprosci.2010.04.180
Ioannidou PG, Papanikolaou DA, Bosdou JK et al (2022) Improvement in sperm quality by oral antioxidant supplementation in infertile men with varicocele who have not undergone surgical repair: Systematic review and meta-analysis. Andrologia 54:e14533. https://doi.org/10.1111/and.14533
Islam MM, Umehara T, Tsujita N et al (2023) Treatment with cholesterol just after thawing maintains the fertility of bull sperm. Mol Hum Reprod 29: gaad031. https://doi.org/10.1093/molehr/gaad031
Islam MdM, Umehara T, Tsujita N et al (2021) Saturated fatty acids accelerate linear motility through mitochondrial ATP production in bull sperm. Reprod Med Biol 20:289–298. https://doi.org/10.1002/rmb2.12381
John Morris G, Acton E, Murray BJ et al (2012) Freezing injury: The special case of the sperm cell. Cryobiology 64:71–80. https://doi.org/10.1016/j.cryobiol.2011.12.002
Khalil WA, El-Harairy MA, Zeidan AEB et al (2017) Evaluation of bull spermatozoa during and after cryopreservation: Structural and ultrastructural insights. Int J Vet Sci Med 6:S49–S56. https://doi.org/10.1016/j.ijvsm.2017.11.001
Leisegang K, Henkel R, Agarwal A (2017) Redox Regulation of Fertility in Aging Male and the Role of Antioxidants: A Savior or Stressor. Curr Pharm Des 23:4438–4450. https://doi.org/10.2174/1381612822666161019150241
Lewis SEM, Aitken RJ (2005) DNA damage to spermatozoa has impacts on fertilization and pregnancy. Cell Tissue Res 322:33–41. https://doi.org/10.1007/s00441-005-1097-5
Lewis SEM, Sterling ESL, Young IS et al (1997) Comparison of individual antioxidants of sperm and seminal plasma in fertile and infertile men. Fertil Steril 67:142–147. https://doi.org/10.1016/S0015-0282(97)81871-7
Moazamian R, Polhemus A, Connaughton H et al (2015) Oxidative stress and human spermatozoa: diagnostic and functional significance of aldehydes generated as a result of lipid peroxidation. Mol Hum Reprod 21:502–515. https://doi.org/10.1093/molehr/gav014
Morielli T, O’Flaherty C (2015) Oxidative stress impairs function and increases redox protein modifications in human spermatozoa. Reprod Camb Engl 149:113–123. https://doi.org/10.1530/REP-14-0240
Nagy S, Hallap T, Johannisson A et al (2004) Changes in plasma membrane and acrosome integrity of frozen-thawed bovine spermatozoa during a 4 h incubation as measured by multicolor flow cytometry. Anim Reprod Sci 80:225–235. https://doi.org/10.1016/j.anireprosci.2003.08.003
O’Flaherty C, Scarlata E (2022) Oxidative stress and reproductive function: The protection of mammalian spermatozoa against oxidative stress. Reprod Camb Engl 164:F67–F78. https://doi.org/10.1530/REP-22-0200
Panner Selvam MK, Agarwal A, Henkel R et al (2020) The effect of oxidative and reductive stress on semen parameters and functions of physiologically normal human spermatozoa. Free Radic Biol Med 152:375–385. https://doi.org/10.1016/j.freeradbiomed.2020.03.008
Partyka A, Niżański W, Łukaszewicz E (2010) Evaluation of fresh and frozen-thawed fowl semen by flow cytometry. Theriogenology 74:1019–1027. https://doi.org/10.1016/j.theriogenology.2010.04.032
Peña FJ, Johannisson A, Wallgren M et al (2003) Assessment of fresh and frozen–thawed boar semen using an Annexin-V assay: a new method of evaluating sperm membrane integrity. Theriogenology 60:677–689. https://doi.org/10.1016/S0093-691X(03)00081-5
Pini T, Leahy T, de Graaf SP (2018) Sublethal sperm freezing damage: Manifestations and solutions. Theriogenology 118:172–181. https://doi.org/10.1016/j.theriogenology.2018.06.006
Rucker R, Chowanadisai W, Nakano M (2009) Potential physiological importance of PQQ. Altern Med Rev J Clin Ther 14:268–77
Sharma R, Agarwal A, Mohanty G et al (2013) Proteomic analysis of seminal fluid from men exhibiting oxidative stress. Reprod Biol Endocrinol RBE 11:85. https://doi.org/10.1186/1477-7827-11-85
Simões R, Feitosa WB, Siqueira AFP et al (2013) Influence of bovine sperm DNA fragmentation and oxidative stress on early embryo in vitro development outcome. Reprod Camb Engl 146:433–441. https://doi.org/10.1530/REP-13-0123
Sławeta R, Laskowska T (1987) The effect of glutathione on the motility and fertility of frozen bull sperm. Anim Reprod Sci 13:249–253. https://doi.org/10.1016/0378-4320(87)90061-3
Sönmez M, Türk G, Yüce A (2005) The effect of ascorbic acid supplementation on sperm quality, lipid peroxidation and testosterone levels of male Wistar rats. Theriogenology 63:2063–2072. https://doi.org/10.1016/j.theriogenology.2004.10.003
Umehara T, Tsujita N, Zhu Z et al (2020) A simple sperm-sexing method that activates TLR7/8 on X sperm for the efficient production of sexed mouse or cattle embryos. Nat Protoc 15:2645–2667. https://doi.org/10.1038/s41596-020-0348-y
Vince S, Žura Žaja I, Samardžija M et al (2018) Age-related differences of semen quality, seminal plasma, and spermatozoa antioxidative and oxidative stress variables in bulls during cold and warm periods of the year. Anim Int J Anim Biosci 12:559–568. https://doi.org/10.1017/S1751731117001811
Xuan W, Lamhonwah A-M, Librach C et al (2003) Characterization of organic cation/carnitine transporter family in human sperm. Biochem Biophys Res Commun 306:121–128. https://doi.org/10.1016/S0006-291X(03)00930-6
Zhu B, Simonis U, Cecchini G et al (2006) Comparison of Pyrroloquinoline Quinone and/or Metoprolol on Myocardial Infarct Size and Mitochondrial Damage in a Rat Model of Ischemia/Reperfusion Injury. J Cardiovasc Pharmacol Ther 11:119–128. https://doi.org/10.1177/1074248406288757
Zhu Z, Kawai T, Umehara T et al (2019) Negative effects of ROS generated during linear sperm motility on gene expression and ATP generation in boar sperm mitochondria. Free Radic Biol Med 141:159–171. https://doi.org/10.1016/j.freeradbiomed.2019.06.018
Acknowledgements
This work was supported in part by Bill and Melinda Gates Foundation (to MS). AMI and TY performed the experiments and analyzed the data. AMI and TY wrote the manuscripts. MS and TY designed this study, supervised all aspects of this study, and revised the manuscript.
Funding
Open Access funding provided by Hiroshima University.
Author information
Authors and Affiliations
Corresponding author
Supplementary Information
Below is the link to the electronic supplementary material.
11626_2024_944_MOESM1_ESM.pptx
Supplementary file1 Supplemental Figure 1: Gating strategy of flow cytometry. (A) Gating strategy for the selection of single sperm. Using forward scatter (FSC)-A and side scatter (SSC)-A dot plots, cells of similar size and complexity were first selected (R1). In FSC-A and FSC-H dot plots and FSC-A and FSC-W dot plots, similar-size cells were accumulated near the area; thus, using these plots, again similar-size cells were selected (R2, R3). The cells in R3 were used for the below analysis. (B) Histograms of DCFH-DA staining. (C) Histograms of mtSOX deep Red staining. (D) The dot plots of 5,5’,6,6’-tetrachloro-1,1’,3,3’-tetraethylbenzimidazolyl carbocyanine iodide (JC-1) green (x-axis) and red (y-axis). The percentage of JC-1 red-positive sperm (R5) was used for the analysis. (PPTX 349 KB)
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Younus, A.M., Yamanaka, T. & Shimada, M. The protective effects of antioxidants against endogenous and exogenous oxidative stress on bull sperm. In Vitro Cell.Dev.Biol.-Animal (2024). https://doi.org/10.1007/s11626-024-00944-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11626-024-00944-w