Abstract
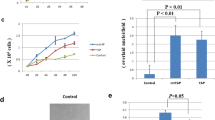
Template activating factor-I (TAF-I) is a multifunctional protein involved in various biological processes including the inhibition of histone acetylation, DNA replication, cell cycle regulation, and oncogenesis. Two main TAF-I isoforms with different N-termini, TAF-Iα and TAF-Iβ (SET), are expressed in cells. There are numerous data about functional properties of TAF-Iβ, whereas the effects of TAF-Iα remain largely unexplored. Here, we employed focus formation and cell proliferation assays, TUNEL staining, cytological analysis, and RT-qPCR to compare the effects of human TAF-Iα and TAF-Iβ genes, transiently expressed in Rat2 cells and in Misgurnus fossilis loaches. We found that both TAF-I isoforms possessed equal oncogenic potential in these systems. Furthermore, an overexpression of human TAF-Iα and TAF-Iβ in Rat2 cells promoted their proliferation. Accordingly, the mitotic index was increased in the transgenic loaches expressing human TAF-Iα or TAF-Iβ. TUNEL assay as well as downregulation of p53 gene and upregulation of bcl-2 gene in these transgenic loaches demonstrated that both isoforms suppressed apoptosis. Thus, TAF-Iα isoform exerts the same oncogenic potential as TAF-Iβ, likely by suppressing the apoptosis and promoting cell proliferation.




Similar content being viewed by others
References
Andreeva LE, Sleptsova LA, Grigorenko AP, Gavriushkin AV, Kuznetsov AV (2003) Loach spermatozoa transfer foreign DNA, which expression is discovered in the early development stages. Genetika 39:758–761
Asaka MN, Murano K, Nagata K (2008) Sp1-mediated transcription regulation of TAF-Ialpha gene encoding a histone chaperone. Biochem Biophys Res Commun 376:665–670. https://doi.org/10.1016/j.bbrc.2008.09.053
Baron EM, Bobrysheva IV, Varshaver NB (1992) The plasmid pEJ6.6 carrying the activated c-Ha-ras-1 oncogene increases the mutation frequency in Chinese hamster cells. Genetika 28:35–40
Brander DM, Friedman DR, Volkheimer AD, Christensen DJ, Rassenti LZ, Kipps TJ, Guadalupe E, Chen Y, Zhang D, Wang X, Davis C, Owzar K, Weinberg JB (2019) SET alpha and SET beta mRNA isoforms in chronic lymphocytic leukaemia. Br J Haematol 184:605–615. https://doi.org/10.1111/bjh.15677
Bui PL, Nishimura K, Seminario Mondejar G, Kumar A, Aizawa S, Murano K, Nagata K, Hayashi Y, Fukuda A, Onuma Y, Ito Y, Nakanishi M, Hisatake K (2019) Template activating factor-I α regulates retroviral silencing during reprogramming. Cell Rep 29:1909–1922.e5. https://doi.org/10.1016/j.celrep.2019.10.010
Cristóbal I, Garcia-Orti L, Cirauqui C, Cortes-Lavaud X, García-Sánchez MA, Calasanz MJ, Odero MD (2012) Overexpression of SET is a recurrent event associated with poor outcome and contributes to protein phosphatase 2A inhibition in acute myeloid leukemia. Haematologica 97:543–550. https://doi.org/10.3324/haematol.2011.050542
Deng X, Gao F, May WS (2009) Protein phosphatase 2A inactivates Bcl2’s antiapoptotic function by dephosphorylation and up-regulation of Bcl2-p53 binding. Blood 113:422–428. https://doi.org/10.1182/blood-2008-06-165134
Edupuganti RR, Harikumar A, Aaronson Y, Biran A, Sailaja BS, Nissim-Rafinia M, Azad GK, Cohen MA, Park JE, Shivalila CS, Markoulaki S, Sze SK, Jaenisch R, Meshorer E (2017) Alternative SET/TAFI promoters regulate embryonic stem cell differentiation. Stem Cell Rep 9:1291–1303. https://doi.org/10.1016/j.stemcr.2017.08.021
Jariwalla RJ, Tanczos B, Jones C, Ortiz J, Salimi-Lopez S (1986) DNA amplification and neoplastic transformation mediated by a herpes simplex DNA fragment containing cell-related sequences. Proc Natl Acad Sci U S A 83:1738–1742. https://doi.org/10.1073/pnas.83.6.1738
Jiang SW, Xu S, Chen H, Liu J, Duan P (2017) Oncogenic role of SET/I2PP2A for gynecologic cancers. Curr Drug Targets 18:1152–1157. https://doi.org/10.2174/1389450118666170328114506
Kadota S, Nagata K (2014) Silencing of IFN-stimulated gene transcription is regulated by histone H1 and its chaperone TAF-I. Nucleic Acids Res 42:7642–7653. https://doi.org/10.1093/nar/gku485
Kajitani K, Kato K, Nagata K (2017) Histone H1 chaperone activity of TAF-I is regulated by its subtype-dependent intramolecular interaction. Genes Cells 22:334–347. https://doi.org/10.1111/gtc.12478
Kim JY, Lee KS, Seol JE, Yu K, Chakravarti D, Seo SB (2012) Inhibition of p53 acetylation by INHAT subunit SET/TAF-Iβ represses p53 activity. Nucleic Acids Res 40:75–87. https://doi.org/10.1093/nar/gkr614
Liu Y, Jia Y, Fu X, He P (2019) TAF-Iβ deficiency inhibits proliferation and promotes apoptosis by rescuing PP2A and inhibiting the AKT/GSK-3β pathway in leukemic cells. Exp Ther Med 18:3801–3808. https://doi.org/10.3892/etm.2019.8012
Liu W, Akhand A, Takeda K, Kawamoto Y, Itoigawa M, Kato M, Suzuki H, Ishikawa N, Nakashima I (2003) Protein phosphatase 2A-linked and -unlinked caspase-dependent pathways for downregulation of Akt kinase triggered by 4-hydroxynonenal. Cell Death Differ 10:772–781. https://doi.org/10.1038/sj.cdd.4401238
Makarova IV, Kazakov AA, Makarova AV, Khaidarova NV, Kozikova LV, Nenasheva VV, Gening LV, Tarantul VZ, Andreeva LE (2012) Transient expression and activity of human DNA polymerase iota in loach embryos. Biotechnol Lett 34:205–212. https://doi.org/10.1007/s10529-011-0764-8
Matsumoto K, Nagata K, Miyaji-Yamaguchi M, Kikuchi A, Tsujimoto M (1999) Sperm Chromatin decondensation by template activating factor I through direct interaction with basic proteins. Mol Cell Biol 19:6940–6952. https://doi.org/10.1128/mcb.19.10.6940
Mazhar S, Taylor SE, Sangodkar J, Narla G (2019) Targeting PP2A in cancer: combination therapies. Biochim Biophys Acta, Mol Cell Res 1866:51–63. https://doi.org/10.1016/j.bbamcr.2018.08.020
Nenasheva VV, Stepanenko EA, Makarova IV, Khaidarova NV, Antonov SA, Kozikova LV, Ayed Z, Vovk AD, Shcherbatova NA, Andreeva LE, Tarantul VZ (2018) Expression of the human TRIM14 and its mutant form (P207L) promotes apoptosis in transgenic loaches. Mol Biol Rep 45:2087–2093. https://doi.org/10.1007/s11033-018-4365-7
Neviani P, Santhanam R, Trotta R, Notari M, Blaser BW, Liu S, Mao H, Chang JS, Galietta A, Uttam A, Roy DC, Valtieri M, Bruner-Klisovic R, Caligiuri MA, Bloomfield CD, Marcucci G, Perrotti D (2005) The tumor suppressor PP2A is functionally inactivated in blast crisis CML through the inhibitory activity of the BCR/ABL-regulated SET protein. Cancer Cell 8:355–368. https://doi.org/10.1016/j.ccr.2005.10.015
Okuwaki M, Nagata K (1998) Template activating factor-I remodels the chromatin structure and stimulates transcription from the chromatin template. J Biol Chem 273:34511–34518. https://doi.org/10.1074/jbc.273.51.34511
Ouchida AT, Uyemura VT, Queiroz AL, Brauer VS, Cavalcanti-Neto MP, Sousa LO, Uyemura SA, Curti C, Leopoldino AM (2019) SET protein accumulation prevents cell death in head and neck squamous cell carcinoma through regulation of redox state and autophagy. Biochim Biophys Acta, Mol Cell Res 1866:623–637. https://doi.org/10.1016/j.bbamcr.2019.01.005
Rosati R, La Starza R, Barba G, Gorello P, Pierini V, Matteucci C, Roti G, Crescenzi B, Aloisi T, Aversa F, Martelli MF, Mecucci C (2007) TAF-Ialpha/CAN and TAF-Ibeta/CAN fusion transcripts are generated by chromosome 9q34 deletion in acute myeloid leukemia. Haematologica 92:232–235. https://doi.org/10.3324/haematol.10538
Saavedra F, Rivera C, Rivas E, Merino P, Garrido D, Hernández S, Forné I, Vassias I, Gurard-Levin ZA, Alfaro IE, Imhof A, Almouzni G, Loyola A (2017) PP32 and SET/TAF-Iβ proteins regulate the acetylation of newly synthesized histone H4. Nucleic Acids Res 45:11700–11710. https://doi.org/10.1093/nar/gkx775
Saito S, Miyaji-Yamaguchi M, Shimoyama T, Nagata K (1999) Functional domains of template-activating factor-I as a protein phosphatase 2A inhibitor. Biochem Biophys Res Commun 259:471–475. https://doi.org/10.1006/bbrc.1999.0790
Serifi I, Tzima E, Soupsana K, Karetsou Z, Beis D, Papamarcaki T (2016) The zebrafish homologs of SET/I2PP2A oncoprotein: expression patterns and insights into their physiological roles during development. Biochem J 473:4609–4627. https://doi.org/10.1042/BCJ20160523
Tarantul VZ, Nikolaev AI, Martynenko A, Hannig H, Hunsmann G, Bodemer W (2000) Differential gene expression in B-cell non-Hodgkin’s lymphoma of SIV-infected monkey. AIDS Res Hum Retrovir 16:173–179. https://doi.org/10.1089/088922200309511
Tarantul V, Nikolaev A, Hannig H, Kalmyrzaev B, Muchoyan I, Maximov V, Nenasheva V, Dubovaya V, Hunsmann G, Bodemer W (2001) Detection of abundantly transcribed genes and gene translocation in human immunodeficiency virus-associated non-Hodgkin’s lymphoma. Neoplasia 3:132–142. https://doi.org/10.1038/sj.neo.7900137
von Lindern M, van Baal S, Wiegant J, Raap A, Hagemeijer A, Grosveld G (1992) Can, a putative oncogene associated with myeloid leukemogenesis, may be activated by fusion of its 3′ half to different genes: characterization of the set gene. Mol Cell Biol 12:3346–3355. https://doi.org/10.1128/mcb.12.8.3346
Wang D, Kon N, Lasso G, Jiang L, Leng W, Zhu WG, Qin J, Honig B, Gu W (2016b) Acetylation-regulated interaction between p53 and SET reveals a widespread regulatory mode. Nature 538:118–122. https://doi.org/10.1038/nature19759
Wang Y, He PC, Liu YF, Qi J, Zhang M (2016a) Construction of SET overexpression vector and its effects on the proliferation and apoptosis of 293T cells. Mol Med Rep 13:4329–4334. https://doi.org/10.3892/mmr.2016.5049
Wei Y, Maximov V, Morrissy SA, Taylor MD, Pallas DC, Kenney AM (2019) p53 function Is compromised by inhibitor 2 of phosphatase 2A in sonic hedgehog medulloblastoma. Mol Cancer Res 17:186–198. https://doi.org/10.1158/1541-7786.MCR-18-0485
Westerfield M (2000) The Zebrafish Book. A guide for the laboratory use of zebrafish (Danio rerio), 4th edn. University of Oregon Press, Eugene
Wu M, Yu G, Yan T, Ke D, Wang Q, Liu R, Wang JZ, Zhang B, Chen D, Wang X (2018) Phosphorylation of SET mediates apoptosis via P53 hyperactivation and NM23-H1 nuclear import. Neurobiol Aging 69:38–47. https://doi.org/10.1016/j.neurobiolaging.2018.04.022
Yuan X, Zhang T, Zheng X, Zhang Y, Feng T, Liu P, Sun Z, Qin S, Liu X, Zhang L, Song J, Liu Y (2017) Overexpression of SET oncoprotein is associated with tumor progression and poor prognosis in human gastric cancer. Oncol Rep 38:1733–1741. https://doi.org/10.3892/or.2017.5788
Zhang DL, Yang N (2020) TNFAIP8 regulates cisplatin resistance through TAF-Iα and promotes malignant progression of esophageal cancer. Eur Rev Med Pharmacol Sci 24:4775–4784. https://doi.org/10.26355/eurrev_202005_21166
Zhong J, Ren X, Chen Z, Zhang H, Zhou L, Yuan J, Li P, Chen X, Liu W, Wu D, Yang X, Liu J (2019) miR-21-5p promotes lung adenocarcinoma progression partially through targeting SET/TAF-Iα. Life Sci 231:116539. https://doi.org/10.1016/j.lfs.2019.06.014
Acknowledgements
This study was performed using equipment of the Center of Common Scientific Equipment of the Institute of Molecular Geneticsof National Research Centre “Kurchatov Institute”.
Funding
This work was supported by the Program for Molecular and Cellular Biology from Presidium of Russian Academy of Sciences (01201356275) and Russian Fundamental Scientific Research Programs (АААА-А19-119022290059-8; АААA-А18-118021590132-9. Theme number: 0445-2019-0030).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Editor: Tetsuji Okamoto
Supplementary Information
ESM 1
(PDF 5756 kb)
Rights and permissions
About this article
Cite this article
Nenasheva, V.V., Makarova, I.V., Stepanenko, E.A. et al. Human TAF-Iα promotes oncogenic transformation via enhancement of cell proliferation and suppression of apoptosis. In Vitro Cell.Dev.Biol.-Animal 57, 531–538 (2021). https://doi.org/10.1007/s11626-021-00572-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11626-021-00572-8