Abstract
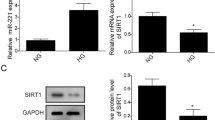
Hyperglycemia or high-glucose (HG)-induced apoptosis in human retinal pigment epithelial (RPE) cells is a characteristic process in diabetic retinopathy. In our study, we examined whether microRNA-29 (miR-29) may regulate HG-induced RPE cell apoptosis. Human RPE cell line, ARPE-19 cells, was treated with various high concentration of glucose in vitro. HG-induced RPE cell apoptosis was examined by terminal deoxynucleotide transferase-mediated dUTP nick end labeling (TUNEL) assay and miR-29 gene expression by quantitative RT-PCR (qRT-PCR). miR-29 was then downregulated in RPE cells, and its effect on HG-induced apoptosis was examined by TUNEL assay and western blot assay on caspase-7 protein. Association of miR-29 on its downstream target, PTEN, in HG-induced RPE cell apoptosis was evaluated by dual-luciferase assay and qRT-PCR. PTEN was silenced in RPE cells. The effects of PTEN downregulation on miR-29-mediated HG-induced RPE cell apoptosis were also examined by TUNEL and western blot assays. HG induced significant apoptosis in RPE cells in a dose-dependent manner. miR-29 was upregulated by HG in RPE cells. miR-29 downregulation protected HG-induced apoptosis and reduced the production of caspase-7 protein in RPE cells. PTEN was shown to be directly downregulated by HG and then upregulated by miR-29 downregulation in RPE cells. Small interfering RNA (siRNA)-mediated PTEN downregulation reversed the protective effect of miR-29 downregulation on HG-induced RPE cell apoptosis. This study demonstrates that miR-29, through inverse association of PTEN, plays an important role in the process of HG-induced apoptosis in RPE cells.




Similar content being viewed by others
References
Ahsan H (2015) Diabetic retinopathy—biomolecules and multiple pathophysiology. Diabetes Metab Syndr 9(1):51–54
Bartel DP (2004) MicroRNAs: genomics, biogenesis, mechanism, and function. Cell 116(2):281–297
Heng LZ, Comyn O, Peto T, Tadros C, Ng E, Sivaprasad S, Hykin PG (2013) Diabetic retinopathy: pathogenesis, clinical grading, management and future developments. Diabet Med 30(6):640–650
Karolina DS, Armugam A, Tavintharan S, Wong MT, Lim SC, Sum CF, Jeyaseelan K (2011) MicroRNA 144 impairs insulin signaling by inhibiting the expression of insulin receptor substrate 1 in type 2 diabetes mellitus. PLoS One 6(8), e22839
Kim DI, Park MJ, Lim SK, Choi JH, Kim JC, Han HJ, Kundu TK, Park JI, Yoon KC, Park SW et al (2014) High-glucose-induced CARM1 expression regulates apoptosis of human retinal pigment epithelial cells via histone 3 arginine 17 dimethylation: role in diabetic retinopathy. Arch Biochem Biophys 560:36–43
Kim JW, Kang KH, Burrola P, Mak TW, Lemke G (2008) Retinal degeneration triggered by inactivation of PTEN in the retinal pigment epithelium. Genes Dev 22(22):3147–3157
Kim VN, Nam JW (2006) Genomics of microRNA. Trends Genet 22(3):165–173
Kong L, Zhu J, Han W, Jiang X, Xu M, Zhao Y, Dong Q, Pang Z, Guan Q, Gao L et al (2011) Significance of serum microRNAs in pre-diabetes and newly diagnosed type 2 diabetes: a clinical study. Acta Diabetol 48(1):61–69
Kovacs B, Lumayag S, Cowan C, Xu S (2011) MicroRNAs in early diabetic retinopathy in streptozotocin-induced diabetic rats. Invest Ophthalmol Vis Sci 52(7):4402–4409
Kutty RK, Samuel W, Jaworski C, Duncan T, Nagineni CN, Raghavachari N, Wiggert B, Redmond TM (2010) MicroRNA expression in human retinal pigment epithelial (ARPE-19) cells: increased expression of microRNA-9 by N-(4-hydroxyphenyl)retinamide. Mol Vis 16:1475–1486
Kwon CH, Zhu X, Zhang J, Knoop LL, Tharp R, Smeyne RJ, Eberhart CG, Burger PC, Baker SJ (2001) Pten regulates neuronal soma size: a mouse model of Lhermitte-Duclos disease. Nat Genet 29(4):404–411
Leslie NR, Bennett D, Gray A, Pass I, Hoang-Xuan K, Downes CP (2001) Targeting mutants of PTEN reveal distinct subsets of tumour suppressor functions. Biochem J 357(Pt 2):427–435
Mastropasqua R, Toto L, Cipollone F, Santovito D, Carpineto P, Mastropasqua L (2014) Role of microRNAs in the modulation of diabetic retinopathy. Prog Retin Eye Res 43:92–107
Sakagami K, Chen B, Nusinowitz S, Wu H, Yang XJ (2012) PTEN regulates retinal interneuron morphogenesis and synaptic layer formation. Mol Cell Neurosci 49(2):171–183
Silva VA, Polesskaya A, Sousa TA, Correa VM, Andre ND, Reis RI, Kettelhut IC, Harel-Bellan A, De Lucca FL (2011) Expression and cellular localization of microRNA-29b and RAX, an activator of the RNA-dependent protein kinase (PKR), in the retina of streptozotocin-induced diabetic rats. Mol Vis 17:2228–2240
Simo R, Villarroel M, Corraliza L, Hernandez C, Garcia-Ramirez M (2010) The retinal pigment epithelium: something more than a constituent of the blood-retinal barrier—implications for the pathogenesis of diabetic retinopathy. J Biomed Biotechnol 2010:190724
Sulis ML, Parsons R (2003) PTEN: from pathology to biology. Trends Cell Biol 13(9):478–483
Vinores SA, Derevjanik NL, Ozaki H, Okamoto N, Campochiaro PA (1999) Cellular mechanisms of blood-retinal barrier dysfunction in macular edema. Doc Ophthalmol 97(3-4):217–228
Winer J, Jung CK, Shackel I, Williams PM (1999) Development and validation of real-time quantitative reverse transcriptase-polymerase chain reaction for monitoring gene expression in cardiac myocytes in vitro. Anal Biochem 270(1):41–49
Wu JH, Gao Y, Ren AJ, Zhao SH, Zhong M, Peng YJ, Shen W, Jing M, Liu L (2012) Altered microRNA expression profiles in retinas with diabetic retinopathy. Ophthalmic Res 47(4):195–201
Zhao C, Yasumura D, Li X, Matthes M, Lloyd M, Nielsen G, Ahern K, Snyder M, Bok D, Dunaief JL et al (2011) mTOR-mediated dedifferentiation of the retinal pigment epithelium initiates photoreceptor degeneration in mice. J Clin Invest 121(1):369–383
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Editor: Tetsuji Okamoto
Rights and permissions
About this article
Cite this article
Lin, X., Zhou, X., Liu, D. et al. MicroRNA-29 regulates high-glucose-induced apoptosis in human retinal pigment epithelial cells through PTEN. In Vitro Cell.Dev.Biol.-Animal 52, 419–426 (2016). https://doi.org/10.1007/s11626-015-9990-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11626-015-9990-z