Abstract
Background
Guidelines recommend neoadjuvant therapy (NT) for clinical stage II–III (locally advanced) rectal adenocarcinoma, but utilization remains suboptimal. The causes of NT omission remain poorly understood.
Methods
The main outcomes in this study of patients with resected clinically non-metastatic rectal adenocarcinoma in the 2010–2015 National Cancer Database were local staging utilization in patients with non-metastatic tumors (i.e., undocumented clinical stage/pathologic stage I–III) and NT utilization for locally advanced tumors. Multivariable regression was used to examine predictors of these outcomes. Facility-specific risk- and reliability-adjusted local staging and NT rates were calculated. Positive margins and overall survival (OS) were examined as secondary outcomes.
Results
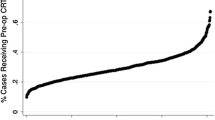
Local staging was omitted in 7737/43,819 (17.7%) patients with clinically non-metastatic tumors and NT was omitted in 5199/31,632 (16.4%) patients with locally advanced tumors. NT was utilized in 24,826 (91.1%) locally advanced patients who had local staging vs. 1607 (36.6%) patients who did not; 2785 (53.6%) locally advanced patients with NT omitted also had local staging omitted. Treatment at facilities with lowest quintile local staging rates was associated with NT omission (relative risk 2.41, 95% confidence interval 2.11, 2.75). Adjusted facility local staging rates varied sixfold (16.1–98.0%), facility NT rates varied twofold (43.9–95.9%), and they were correlated (r = 0.58; P < 0.001). Local staging omission and NT omission were independently associated with positive margins and decreased OS.
Conclusions
Local staging omission is a common care process in over half of cases of omitted NT. These data emphasize the need for quality improvement efforts directed at providing facilities feedback about their local staging rates.


Similar content being viewed by others
References
NCCN Clinical Practice Guidelines in Oncology: Rectal Cancer. Version 1.18. https://www.nccn.org/professionals/physician_gls/pdf/rectal.pdf Accessed June 20, 2018.
Sauer R, Becker H, Hohenberger W, Rodel C, Wittekind C, Fietkau R, Martus P, Tschmelitsch J, Hager E, Hess C, Karstens J, Liersch T, Schmidberger H, Raab R, Group. GRCS. Preoperative versus Postoperative Chemoradiotherapy for Rectal Cancer. NEJM. 2004;351(17):1731–40.
Sauer R, Liersch T, Merkel S, Fietkau R, Hohenberger W, Hess C, Becker H, Raab HR, Villanueva MT, Witzigmann H, Wittekind C, Beissbarth T, Rodel C. Preoperative versus postoperative chemoradiotherapy for locally advanced rectal cancer: results of the German CAO/ARO/AIO-94 randomized phase III trial after a median follow-up of 11 years. J Clin Oncol. 2012;30(16):1926–33. https://doi.org/10.1200/JCO.2011.40.1836.
Monson JR, Probst CP, Wexner SD, Remzi FH, Fleshman JW, Garcia-Aguilar J, Chang GJ, Dietz DW, Consortium for Optimizing the Treatment of Rectal C. Failure of evidence-based cancer care in the United States: the association between rectal cancer treatment, cancer center volume, and geography. Ann Surg. 2014;260(4):625–31; discussion 31-2. https://doi.org/10.1097/SLA.0000000000000928.
Sineshaw HM, Jemal A, Thomas CR, Jr., Mitin T. Changes in treatment patterns for patients with locally advanced rectal cancer in the United States over the past decade: An analysis from the National Cancer Data Base. Cancer. 2016;122(13):1996–2003. https://doi.org/10.1002/cncr.29993.
Midura EF, Jung AD, Daly MC, Hansenman DJ, Shah SA, Paquette IM. Cancer Center Volume and Type Impact Stage-Specific Utilization of Neoadjuvant Therapy in Rectal Cancer. Dig Dis Sci. 2017;62(8):1906–12.
Sun Z, Adam MA, Kim J, Turner MC, Fisher DA, Choudhury KR, Czito BG, Migaly J, Mantyh CR. Association Between Neoadjuvant Chemoradiation and Survival for Patients With Locally Advanced Rectal Cancer. Colorectal Dis. 2017. https://doi.org/10.1111/codi.13754.
Delitto D, George TJ, Jr., Loftus TJ, Qiu P, Chang GJ, Allegra CJ, Hall WA, Hughes SJ, Tan SA, Shaw CM, Iqbal A. Prognostic Value of Clinical vs Pathologic Stage in Rectal Cancer Patients Receiving Neoadjuvant Therapy. J Natl Cancer Inst. 2018;110(5):460–6. https://doi.org/10.1093/jnci/djx228.
NCDB Rectal Measure Specifications. https://www.facs.org/~/media/files/quality%20programs/cancer/ncdb/measure%20specs%20rectal.ashx. Accessed June 15, 2018.
National Accreditation Program for Rectal Cancer. https://www.facs.org/quality-programs/cancer/naprc. Accessed June 22, 2018.
Wexner SD, Berho ME. The Rationale for and Reality of the New National Accreditation Program for Rectal Cancer. Dis Colon rectum. 2017;60(6):595–602.
Lee L, Dietz D, Fleming F, Remzi F, Wexner S, Winchester D, Monson J. Accreditation Readiness in US Multidisciplinary Rectal Cancer Care: A Survey of OSTRICH Member Institutions. JAMA Surg. 2018;153(4):388–90.
Swords D, Skarda D, Sause W, Gawlick U, Cannon G, Lewis M, Scaife C, Gygi JA, Kim H. Surgeon-level Variation in Utilization of Local Staging and Neoadjuvant Therapy for Stage II-III Rectal Adenocarcinoma. J Gastrointest Surg. 2018;Under review.
Boffa DJ, Rosen JE, Mallin K, Loomis A, Gay G, Palis B, Thoburn K, Gress D, McKellar DP, Shulman LN, Facktor MA, Winchester DP. Using the National Cancer Database for Outcomes Research: A Review. JAMA Oncol. 2017;3(12):1722–8. https://doi.org/10.1001/jamaoncol.2016.6905.
American College of Surgeons. National Cancer Database. Available at https://www.facs.org/quality-programs/cancer/ncdb. Accessed May 19, 2018.
Facility Oncology Registry Data Standards (FORDS): Revised for 2016. https://www.facs.org/~/media/files/quality%20programs/cancer/ncdb/fords%202016.ashx. Accessed June 1, 2018.
Bujko K, Nowacki MP, Nasierowska-Guttmejer A, Michalski W, Bebenek M, Kryj M. Long-term results of a randomized trial comparing preoperative short-course radiotherapy with preoperative conventionally fractionated chemoradiation for rectal cancer. Br J Surg. 2006;93(10):1215–23. https://doi.org/10.1002/bjs.5506.
Ngan SY, Burmeister B, Fisher RJ, Solomon M, Goldstein D, Joseph D, Ackland SP, Schache D, McClure B, McLachlan SA, McKendrick J, Leong T, Hartopeanu C, Zalcberg J, Mackay J. Randomized trial of short-course radiotherapy versus long-course chemoradiation comparing rates of local recurrence in patients with T3 rectal cancer: Trans-Tasman Radiation Oncology Group trial 01.04. J Clin Oncol. 2012;30(31):3827–33. https://doi.org/10.1200/JCO.2012.42.9597.
Schrag D, Weiser MR, Goodman KA, Gonen M, Hollywood E, Cercek A, Reidy-Lagunes DL, Gollub MJ, Shia J, Guillem JG, Temple LK, Paty PB, Saltz LB. Neoadjuvant chemotherapy without routine use of radiation therapy for patients with locally advanced rectal cancer: a pilot trial. J Clin Oncol. 2014;32(6):513–8. https://doi.org/10.1200/JCO.2013.51.7904.
Weiser MR, Fichera A, Schrag D, Boughey JC, You YN. Progress in the PROSPECT trial: precision treatment for rectal cancer? Bull Am Coll Surg. 2015;100(4):51–2.
NCCN Clinical Practice Guidelines in Oncology: Rectal Cancer. Version 3.17. https://www.nccn.org/professionals/physician_gls/pdf/rectal.pdf. Accessed June 27, 2017.
Sauer BC, Brookhart MA, Roy J, VanderWeele T. A review of covariate selection for non-experimental comparative effectiveness research. Pharmacoepidemiol Drug Saf. 2013;22(11):1139–45. https://doi.org/10.1002/pds.3506.
Vandenbroucke JP, von Elm E, Altman DG, Gøtzsche PC, Mulrow CD, Pocock SJ, Poole C, Schlesselman JJ, Egger M, initiative. S. Strengthening the Reporting of Observational Studies in Epidemiology (STROBE): explanation and elaboration. Ann Intern Med. 2007;147(8):163–94.
Wallner LP, Griggs JJ. Advancing the Science of Cancer Health Disparities Research. J Clin Oncol. 2018;36(1):1–3. https://doi.org/10.1200/JCO.2017.73.7932.
Knol MJ, Le Cessie S, Algra A, Vandenbroucke JP, Groenwold RH. Overestimation of risk ratios by odds ratios in trials and cohort studies: alternatives to logistic regression. CMAJ. 2012;184(8):895–6.
Norton EC, Miller MM, Kleinman LC. Computing adjusted risk ratios and risk differences in Stata. Stata J. 2013;13(3):492–509.
Sheetz KH, Dimick JB, Ghaferi AA. Impact of Hospital Characteristics on Failure to Rescue Following Major Surgery. Ann Surg. 2016;263(4):692–7. https://doi.org/10.1097/SLA.0000000000001414.
Dimick JB, Welch HG, Birkmeyer JD. Surgical mortality as an indicator of hospital quality: the problem with small sample size. JAMA. 2004;292(7):847–51.
Dimick JB, Ghaferi AA, Osborne NH, Ko CY, Hall BL. Reliability adjustment for reporting hospital outcomes with surgery. Ann Surg. 2012;255(4):703–7. https://doi.org/10.1097/SLA.0b013e31824b46ff.
Dimick JB, Staiger DO, Birkmeyer JD. Ranking hospitals on surgical mortality: the importance of reliability adjustment. Health Serv Res. 2010;45(6):1614–29.
Bland J M, Altman D G. Correlation, regression, and repeated data. BMJ 1994;308:896.
Bland J M, Altman D G. Calculating correlation coefficients with repeated observations: Part 2--Correlation between subjects. BMJ. 1995;310:633.
Chang JS, Lee Y, Lim JS, Kim NK, Baik SH, Min BS, Huh H, Koom WS. The magnetic resonance imaging-based approach for identification of high-risk patients with upper rectal cancer. Ann Surg. 2014;260(2):293–8. https://doi.org/10.1097/SLA.0000000000000503.
Marinello FG, Frasson M, Baguena G, Flor-Lorente B, Cervantes A, Roselló S, Espí A, García-Granero E. Selective approach for upper rectal cancer treatment: total mesorectal excision and preoperative chemoradiation are seldom necessary. Dis Colon Rectum. 2015;58(6):556–65.
Battersby NJ, How P, Moran B, Stelzner S, West NP, Branagan G, Strassburg J, Quirke P, Tekkis P, Pedersen BG, Gudgeon M, Heald B, Brown G, Group MIS. Prospective Validation of a Low Rectal Cancer Magnetic Resonance Imaging Staging System and Development of a Local Recurrence Risk Stratification Model: The MERCURY II Study. Ann Surg. 2016;263(4):751–60. https://doi.org/10.1097/SLA.0000000000001193.
Harris DA, Thorne K, Hutchings H, Islam S, Holland G, Hatcher O, Gwynne S, Jenkins I, Coyne P, Duff M, Feldman M, Winter DC, Gollins S, Quirke P, West N, Brown G, Fitzsimmons D, Brown A, Beynon J. Protocol for a multicentre randomised feasibility trial evaluating early Surgery Alone In LOw Rectal cancer (SAILOR). BMJ Open. 2016;6(11):e012496. https://doi.org/10.1136/bmjopen-2016-012496.
Roh MS, Colangelo LH, O'Connell MJ, Yothers G, Deutsch M, Allegra CJ, Kahlenberg MS, Baez-Diaz L, Ursiny CS, Petrelli NJ, Wolmark N. Preoperative multimodality therapy improves disease-free survival in patients with carcinoma of the rectum: NSABP R-03. J Clin Oncol. 2009;27(31):5124–30. https://doi.org/10.1200/JCO.2009.22.0467.
Author information
Authors and Affiliations
Contributions
All authors have provided substantial contributions to the conception or design of the work or to the acquisition, analysis, or interpretation of the data for the work; drafted the work or revised it critically for important intellectual content; approved the version to be published; and agreed to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Corresponding author
Ethics declarations
NCDB Disclosure
The data used in the study are derived from a de-identified NCDB file. The American College of Surgeons and the Commission on Cancer have not verified and are not responsible for the analytic or statistical methodology employed, or the conclusions drawn from these data by the investigator.
Electronic supplementary material
ESM 1
(DOCX 1049 kb)
Rights and permissions
About this article
Cite this article
Swords, D.S., Brooke, B.S., Skarda, D.E. et al. Facility Variation in Local Staging of Rectal Adenocarcinoma and its Contribution to Underutilization of Neoadjuvant Therapy. J Gastrointest Surg 23, 1206–1217 (2019). https://doi.org/10.1007/s11605-018-4039-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11605-018-4039-8