Abstract
Background
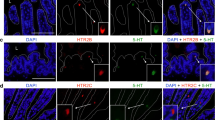
Enteric serotonin may function as a mucosal growth factor. Previous work demonstrated increased crypt cell proliferation and intestinal mucosal surface area with potentiation of serotonin. While an indirect mechanism was postulated to explain these effects, the presence of 5-HT4 receptors on enterocytes raises the possibility of a direct action of serotonin. We hypothesized that a 5-HT4 specific agonist, prucalopride, would stimulate intestinal mucosal growth and enhance absorptive function in the murine small intestine.
Methods
Adult wild-type mice were treated parenterally with prucalopride for 14 days via surgically implanted osmotic pumps. In vivo d-xylose absorption was assessed by oral gavage and serum d-xylose measurements. On day 14, glucose absorption was assessed by instilling a glucose solution into isolated segments of small intestine. The bowel was harvested and examined for morphologic parameters and crypt cell proliferation.
Results
Villus height, crypt depth, and crypt proliferation were significantly increased in the distal small bowel of prucalopride-treated mice compared with control animals. Crypt depth was also increased in the proximal and middle small intestine in treated mice. There was no difference in d-xylose absorption throughout the study period; however, glucose absorption was significantly increased in the distal small intestine of prucalopride-treated mice.
Conclusion
Parenteral administration of the 5-HT4 receptor specific agonist, prucalopride, results in morphologic and functional changes in the murine small intestine that are most prominent in the distal small bowel. While further studies are necessary to delineate the mechanism, it is plausible that the effects are mediated by 5-HT4 receptors on enterocytes.




Similar content being viewed by others
References
Gershon MD. 5-Hydroxytryptamine (serotonin) in the gastrointestinal tract. Curr Opin Endocrinol Diabetes Obes. 2013 Feb;20(1):14–21. https://doi.org/10.1097/MED.0b013e32835bc703. Review.
Hedinger C, Langemann H. [Pronounced thrombocytosis in rats under treatment with 5-oxytryptamin; experimental contribution to the question of endocrine activity of carcinoids]. Schweiz Med Wochenschr. 1955 Apr 16;85(16):368–70. German
Pelagio-Flores R, Ortíz-Castro R, Méndez-Bravo A, Macías-Rodríguez L, López-Bucio J. Serotonin, a tryptophan-derived signal conserved in plants and animals, regulates root system architecture probably acting as a natural auxin inhibitor in Arabidopsis thaliana. Plant Cell Physiol. 2011 Mar;52(3):490–508. https://doi.org/10.1093/pcp/pcr006
Lesurtel M, Graf R, Aleil B, Walther DJ, Tian Y, Jochum W, Gachet C, Bader M, Clavien PA. Platelet-derived serotonin mediates liver regeneration. Science. 2006 Apr 7;312(5770):104–7
MacDonald RA, Schmid R, Hakala TR, Mallory GK. Effect of serotonin upon liver cells of young rats. Proc Soc Exp Biol Med. 1959 May;101(1):83–6
Pakala R, Willerson JT, Benedict CR. Mitogenic effect of serotonin on vascular endothelial cells. Circulation. 1994 Oct;90(4):1919–26
Fiorica-Howells E, Maroteaux L, Gershon MD. Serotonin and the 5-HT(2B) receptor in the development of enteric neurons. J Neurosci. 2000 Jan 1;20(1):294–305
Gross ER, Gershon MD, Margolis KG, Gertsberg ZV, Li Z, Cowles RA. Neuronal serotonin regulates growth of the intestinal mucosa in mice. Gastroenterology. 2012 Aug; 143(2):408–17.e2. https://doi.org/10.1053/j.gastro.2012.05.007. Erratum in Gastroenterology. 2013 Jan;144(1):249. Li, Zhishan [added]
Tackett JJ, Gandotra N, Bamdad MC, Muise ED, Cowles RA. Enhanced serotonin signaling stimulates ordered intestinal mucosal growth. J Surg Res. 2017 Feb;208:198–203. https://doi.org/10.1016/j.jss.2016.09.036
De Ponti F. Pharmacology of serotonin: what a clinician should know. Gut. 2004 Oct;53(10):1520–35. Review
Hoffman JM, Tyler K, MacEachern SJ, Balemba OB, Johnson AC, Brooks EM, Zhao H, Swain GM, Moses PL, Galligan JJ, Sharkey KA, Greenwood-Van Meerveld B, Mawe GM. Activation of colonic mucosal 5-HT(4) receptors accelerates propulsive motility and inhibits visceral hypersensitivity. Gastroenterology. 2012 Apr;142(4):844–854.e4. https://doi.org/10.1053/j.gastro.2011.12.041
Liu MT, Kuan YH, Wang J, Hen R, Gershon MD. 5-HT4 receptor-mediated neuroprotection and neurogenesis in the enteric nervous system of adult mice. J Neurosci. 2009 Aug 5;29(31):9683–99. https://doi.org/10.1523/JNEUROSCI.1145-09.2009.
Takaki M, Goto K, Kawahara I. The 5-hydroxytryptamine 4 Receptor Agonist-induced Actions and Enteric Neurogenesis in the Gut. J Neurogastroenterol Motil. 2014 Jan;20(1):17–30. https://doi.org/10.5056/jnm.2014.20.1.17
Briejer MR, Bosmans JP, Van Daele P, Jurzak M, Heylen L, Leysen JE, Prins NH, Schuurkes JA. The in vitro pharmacological profile of prucalopride, a novel enterokinetic compound. Eur J Pharmacol. 2001 Jun 29;423(1):71–83
Greig CJ, Cowles RA. Improved capacity to evaluate changes in intestinal mucosal surface area using mathematical modeling. Microsc Res Tech. 2017 Jul;80(7):793–798. https://doi.org/10.1002/jemt.22866
Mezei T, Szakács M, Dénes L, Jung J, Egyed-Zsigmond I. Semiautomated Image Analysis of High Contrast Tissue Areas Using Hue/Saturation/Brightness Based Color Filtering. 2011Jan;57(6):679–684.
Tappenden KA. Intestinal adaptation following resection. JPEN J Parenter Enteral Nutr. 2014 May;38(1 Suppl):23S–31S. https://doi.org/10.1177/0148607114525210. Review
Martin CA, Perrone EE, Longshore SW, Toste P, Bitter K, Nair R, Guo J, Erwin CR, Warner BW. Intestinal resection induces angiogenesis within adapting intestinal villi. J Pediatr Surg. 2009 Jun;44(6):1077–82; discussion 1083. https://doi.org/10.1016/j.jpedsurg.2009.02.036
Rowland KJ, Yao J, Wang L, Erwin CR, Maslov KI, Wang LV, Warner BW. Immediate alterations in intestinal oxygen saturation and blood flow after massive small bowel resection as measured by photoacoustic microscopy. J Pediatr Surg. 2012 Jun;47(6):1143–9. https://doi.org/10.1016/j.jpedsurg.2012.03.020
Sigalet DL, Martin GR. Mechanisms underlying intestinal adaptation after massive intestinal resection in the rat. J Pediatr Surg. 1998 Jun;33(6):889–92
Hines OJ, Bilchik AJ, Zinner MJ, Skotzko MJ, Moser AJ, McFadden DW, Ashley SW. Adaptation of the Na+/glucose cotransporter following intestinal resection. J Surg Res. 1994 Jul;57(1):22–7
Musch MW, Bookstein C, Rocha F, Lucioni A, Ren H, Daniel J, Xie Y, McSwine RL, Rao MC, Alverdy J, Chang EB. Region-specific adaptation of apical Na/H exchangers after extensive proximal small bowel resection. Am J Physiol Gastrointest Liver Physiol. 2002 Oct;283(4):G975–85
Menge H, Sepúlveda FV, Smith MW. Cellular adaptation of amino acid transport following intestinal resection in the rat. J Physiol. 1983 Jan;334:213–23
Chaves M, Smith MW, Williamson RC. Increased activity of digestive enzymes in ileal enterocytes adapting to proximal small bowel resection. Gut. 1987 Aug;28(8):981–7
Greig CJ, Gandotra N, Tackett JJ, Bamdad MC, Cowles RA. Enhanced serotonin signaling increases intestinal neuroplasticity. J Surg Res. 2016 Nov;206(1):151–158. https://doi.org/10.1016/j.jss.2016.07.021
Thompson JS, Quigley EM, Adrian TE. Factors affecting outcome following proximal and distal intestinal resection in the dog: an examination of the relative roles of mucosal adaptation, motility, luminal factors, and enteric peptides. Dig Dis Sci. 1999 Jan;44(1):63–74
Appleton GV, Bristol JB, Williamson RC. Proximal enterectomy provides a stronger systemic stimulus to intestinal adaptation than distal enterectomy. Gut. 1987;28 Suppl:165–8
Carbonnel F, Cosnes J, Chevret S, Beaugerie L, Ngô Y, Malafosse M, Parc R, Le Quintrec Y, Gendre JP. The role of anatomic factors in nutritional autonomy after extensive small bowel resection. JPEN J Parenter Enteral Nutr. 1996 Jul-Aug;20(4):275–80
Prins NH, Akkermans LMA, Lefebvre RA, Schuurkes JAJ. 5-HT4 receptors on cholinergic nerves involved in contractility of canine and human large intestine longitudinal muscle. British Journal of Pharmacology. 2000;131(5):927–932. https://doi.org/10.1038/sj.bjp.0703615.
Cellek S, John AK, Thangiah R, Dass NB, Bassil AK, Jarvie EM, Lalude O, Vivekanandan S, Sanger GJ. 5-HT4 receptor agonists enhance both cholinergic and nitrergic activities in human isolated colon circular muscle. Neurogastroenterol Motil. 2006 Sep;18(9):853–61.
Kilbinger H, Wolf D. Effects of 5-HT4 receptor stimulation on basal and electrically evoked release of acetylcholine from guinea-pig myenteric plexus. Naunyn Schmiedebergs Arch Pharmacol.1992 Mar;345(3):270–5
Kilbinger H, Gebauer A, Haas J, Ladinsky H, Rizzi CA. Benzimidazolones and renzapride facilitate acetylcholine release from guinea-pig myenteric plexus via 5-HT4 receptors. Naunyn Schmiedebergs Arch Pharmacol. 1995 Mar;351(3):229–36
Ren J, Zhou X, Galligan JJ. 5-HT4 receptor activation facilitates recovery from synaptic rundown and increases transmitter release from single varicosities of myenteric neurons. Am J Physiol Gastrointest Liver Physiol. 2008 Jun;294(6):G1376–83. https://doi.org/10.1152/ajpgi.00078.2008
Budhoo MR, Harris RP, Kellum JM. The role of the 5-HT4 receptor in Cl- secretion in human jejunal mucosa. Eur J Pharmacol. 1996 Oct 24;314(1–2):109–14
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Park, C.J., Armenia, S.J., Zhang, L. et al. The 5-HT4 Receptor Agonist Prucalopride Stimulates Mucosal Growth and Enhances Carbohydrate Absorption in the Ileum of the Mouse. J Gastrointest Surg 23, 1198–1205 (2019). https://doi.org/10.1007/s11605-018-3907-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11605-018-3907-6