Abstract
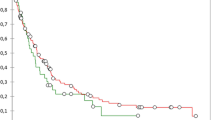
Previous studies examining short- and long-term outcomes of pancreaticoduodenectomy with vascular resection for pancreatic adenocarcinoma have not graded perioperative complication severity. These studies may provide incomplete assessments of the efficacy of vascular resection. In the current study, we evaluated 36 patients who had pancreaticoduodenectomy with major vascular resection. These were matched 1:3 by tumor stage and age to patients who had pancreaticoduodenectomy without vascular resection. Charts were reviewed to identify all complications and 90-day readmissions. Complications were graded as either severe or minor adverse postoperative outcomes, taking into account the total length of stay. There were no statistical differences in patient demographics, comorbidities, or symptoms between the groups. Patients who had vascular resection had significantly increased rates of severe adverse postoperative outcomes, readmissions, lengths of hospital stay, as well as higher hospital costs. Hypoalbuminemia and major vascular resection were independent predictors of severe adverse postoperative outcomes. On multivariate Cox-regression survival analysis, patients who had vascular resection had decreased recurrence-free (12 vs. 17 months) and overall (17 vs. 29 months) survival. Major vascular resection was a predictor of mortality, may be an independent prognostic factor for survival, and may warrant incorporation into future staging systems.

Similar content being viewed by others
References
Worni M, Castleberry AW, Clary BM, Gloor B, Carvalho E, Jacobs DO, Pietrobon R, Scarborough JE, White RR. Concomitant vascular reconstruction during pancreatectomy for malignant disease: a propensity score-adjusted, population-based trend analysis involving 10,206 patients. JAMA Surg 2013;148(4):331-8.
Banz VM, Croagh D, Coldham C, Taniere P, Buckels J, Mayer D, Muiesan P, Bramhall S, Mirza DF. Factors influencing outcome in patients undergoing portal vein resection for adenocarcinoma of the pancreas. Eur J Surg Oncol 2012;38(1):72-9.
Hwang JW, Kim SC, Song KB, Yoon JH, Nam JS, Lee JS, Park KM, Lee YJ. Significance of radiologic location and extent of portal venous involvement on prognosis after pancreatic adenocarcinoma. Pancreas 2015;44(4):665-71.
Jahromi AH, Jaferimehr E, Dabbous HM, Chu Q, D’Agostino H, Shi R, Wellman GP, Zibari GB, Shokouh-Amiri H. Curative resection of pancreatic adenocarcinoma with major venous resection/repair is safe procedure but will not improve survival. JOP 2014;15(5):433-41.
Tseng JF, Raut CP, Lee JE, Pisters PW, Vauthey JN, Abdalla EK, Gomez HF, Sun CC, Crane CH, Wolff RA, Evans DB. Pancreaticoduodenectomy with vascular resection: margin status and survival duration. J Gastrointest Surg 2004;8(8):935-49.
Riediger H, Makowiec F, Fischer E, Adam U, Hopt UT. Postoperative morbidity and long-term survival after pancreaticoduodenectomy with superior mesenterico-portal vein resection. J Gastrointest Surg 2006;10(8):1106-15.
Menon VG, Puri VC, Annamalai AA, Tuli R, Nissen NN. Outcomes of vascular resection in pancreaticoduodenectomy: single-surgeon experience. Am Surg 2013;79(10):1064-7.
Ravikumar R, Sabin C, Abu Hilal M, Bramhall S, White S, Wigmore S, Imber CJ, UK Vascular Resection in Pancreatic Cancer Study Group. Portal vein resection in borderline resectable pancreatic cancer: A United Kingdom multicenter study. J Am Coll Surg 2014;218(3):401-11.
Castleberry AW, White RR, De La Fuente SG, Clary BM, Blazer DG, McCann RL, Pappas TN, Tyler DS, Scarborough JE. The impact of vascular resection on early postoperative outcomes after pancreaticoduodenectomy: an analysis of the American College of Surgeons National Surgical Quality Improvement Program Database. Ann Surg Oncol 2012;19(13):4068-77.
Varadhachary GR, Tamm EP, Crane C, Evans DB, Wolff RA. Borderline resectable pancreatic cancer. Curr Treat Options Gastroenterol 2005;8(5):377-84.
Clavien PA, Barkun J, de Oliveira ML, Vauthey JN, Dindo D, Schulick RD, de Santibañes K, Bassi C, Graf R, Vonlanthen R, Padbury R, Cameron JL, Makuuchi M. The Clavien-Dindo classification of surgical complications: five-year experience. Ann Surg 2009;250(2):187-96.
Baker MS, Sherman KL, Stocker SJ, Hayman AV, Bentren DJ, Prinz RA, Talamonti MS. Using a modification of the Clavien-Dindo system accounting for readmissions and multiple interventions: Defining quality for pancreaticoduodenectomy. J Surg Oncol 2014;110(4):400-6.
Yu XZ, Li J, Fu DL, Di Y, Yang F, Hao SJ, Jin C. Benefit from synchronous portal-superior mesenteric vein resection during pancreaticoduodenectomy for cancer: a meta-analysis. Eur J Surg Oncol 2014;40(4):371-8.
Chua TC, Saxena A. Extended pancreaticoduodenectomy with vascular resection for pancreatic cancer: a systemic review. J Gastrointest Surg 2010;14(9):1442-52.
Mollberg N, Rahbari NN, Koch M, Hartwig W, Hoegger Y, Buchler MW, Weitz J. Arterial resection during pancreatectomy for pancreatic cancer: A systemic review and meta-analysis. Ann Surg 2011;254(6):882-93.
Müller SA, Hartel M, Mehrabi A, Welsch T, Martin DJ, Hinz U, Schied BM, Büchler MW. Vascular resection in pancreatic cancer surgery: survival determinants. J Gastrointest Surg 2008;13(4):784-92.
Mbah N, Brown RE, St Hill CR, Bower MR, Ellis SF, Scoggins CR, McMasters KM, Martin RC. Impact of post-oeprative complications on quality of life after pancreatectomy. JOP 2012;13(4):387-93.
Merkow RP, Bilimoria KY, Tomlinson JS, Paruch JL, Fleming JB, Talamonti MS, Ko CY, Bentrem DJ. Postoperative complications reduce adjuvant chemotherapy use in resectable pancreatic cancer. Ann Surg 2014;260(2);372-7.
Wang J, Estrella JS, Peng L, Rashid A, Varadhachary GR, Wang H, Lee JE, Pisters PW, Vauthey JN, Katz MH, Gomez HF, Evans DB, Abbruzzese JL, Fleming JB, Wang H. Histologic tumor involvement of superior mesenteric vein/portal vein predicts poor prognosis in patients with stage II pancreatic adenocarcinoma treated with neoadjuvant chemoradiation. Cancer 2012;118:3801-11.
Fukuda S, Oussoultzoglou E, Bachellier P, Rosso E, Nakano H, Audet M, Jaeck D. Significance of the depth of portal vein wall invasion after curative resection for pancreatic adenocarcinoma. Arch Surg 2007;142(2):172-9.
Mathur A, Ross SB, Luberice K, Kurian T, Vice M, Toomey P, Rosemurgy AS. Margin status impacts survival after pancreaticoduodenectomy but negative margins should not be pursued. Am Surg 2014;80(4):353-60.
Dusch N, Weiss C, Ströbel P, Kienle P, Post S, Niedergethmann M. Factors predicting long-term survival following pancreatic resection for ductal adenocarcinoma of the pancreas: 40 years of experience. J Gastrointest Surg 2014;18(4):674-81.
Merkow RP, Billimoria KY, Bentrem DJ, Pitt HA, Winchester DP, Posner MC, Ko CY, Pawlik TM. National assessment of margin status as a quality indicator after pancreatic cancer surgery. Ann Surg Oncol 2014;21(4):1067-74.
National Comprehensive Cancer Network (NCCN). NCCN Clinical Practice Guidelines in Oncology: Pancreatic Adenocarcinoma Version 1.2015. http://www.nccn.org/professionals/physician_gls/pdf/pancreatic.pdf
Marti JL, Hochster HS, Hiotis SP, Donahue B, Ryan T, Newman E. Phase I/II trial of induction chemotherapy followed by concurrent chemoradiotherapy and surgery for locoregionally advanced pancreatic cancer. Ann Surg Oncol 2008;15(12):3521-31.
O’Rielly EM, Perelshteyn A, Jarnagin WR, Schattner M, Gerdes H, Capanu M, Tang LH, LaValle J, Winston C, DeMatteo RP, D’Angelica M, Kurtz RC, Abou-Alfa GK, Kimstra DS, Lowery MA, Brennan MF, Coit DG, Reidy DL, Kingham TP, Allen PJ. A single-arm, nonrandomized phase II trial of neoadjuvant gemcitabine and oxaliplatin in patients with resectable pancreas adenocarcinoma. Ann Surg 2014;260(1):142-8.
Christians KK, Tsai S, Mahmoud A, Ritch P, Thomas JP, Wiebe L, Kelly T, Erickson B, Wang H, Evans DB, George B. Neoadjuvant FULFIRINOX for borderline resectable pancreas cancer: a new treatment paradigm? Oncologist 2014;19(3)266-74.
Ferrone CR, Marchegiani G, Hong TS, Ryan DP, Deshpande V, McDonnel EI, Sabbatino F, Santos DD, Allen JN, Blaszkowsky LS, Clark JW, Faris JE, Goyal L, Kwak EL, Murphy JE, Ting DT, Wo JY, Zhu AX, Warshaw AL, Lillemoe KD, Fernandex-del Castillo C. Radiological and surgical implications of neoadjuvant treatment with FOLFIRINOX for locally advanced and borderline resectable pancreatic cancer. Ann Surg 2015;261(1):12-7.
Tzeng CW, Fleming JB, Lee JE, Xiao L, Pisters PW, Vauthey JN, Abdalia EK, Wolff RA, Varadhachary GR, Fogelman DR, Crane CH, Balachandran A, Katz MH. Defined clinical classifications are associated with outcome of patients with anatomically resectable pancreatic adenocarcinoma treated with neoadjuvant therapy. Ann Surg Oncol 2012;19(6):2045-53.
Author information
Authors and Affiliations
Corresponding author
Additional information
Primary Discussant
Jennifer F. Tseng, M.D. MPH (Boston, MA)
Dr Kantor, Dr. Baker, congratulations on your talk on an important subject in pancreatic surgery. I have 2 questions.
1). 50 % of your 36 patients underwent primary transverse closure and 31 % underwent end-to-end anastomosis over the 7 years of the study. This 81 % rate of primary reconstruction without patch or graft is higher than that reported in many studies and may suggest intraoperative decision making rather than preoperative planning for venous resection necessitating reconstruction. Were these vascular reconstructions planned a priori based on imaging or intraoperative necessity due to unexpected tumor involvement and/or bleeding at the time of operation? This might influence the increased complications and worse outcomes from the VR group.
2). You had a 56 % rate of neoadjvuant therapy in the VR group versus 13 % in the matched control group. Was survival (time to recurrence) calculated from diagnosis, as would be appropriate in a study where some patients received neoadjuvant therapy, or from surgery, which would bias the survival against the VR group?
Thank you for the opportunity to review.
Closing Discussant
Dr. Kantor
Dr. Tseng, thank you for reviewing our manuscript and your thoughtful comments.
1). With regard to your first question, for this study we identified all patients that had vascular resections by a retrospective query of our institutional pancreatic database. We then reviewed operative notes and had additional separate discussion with the operating surgeons to clarify the indications/reasons for the vein resection. For our analysis, we excluded vein resections that were done for accidental vascular injuries as well as unplanned vascular resections. .
2). With regard to your second question, overall survival was calculated from time of surgery. We felt that either choice for the point of origin for the survival analysis (from date of surgery or date of diagnosis) would have the potential to introduce bias. We did have many patients that received neoadjuvant chemotherapy in our vascular resection group. All of these patients were selected retrospectively and thus were by nature patients who survived through neoadjuvant therapy to surgery. We felt that using the time of diagnosis would create bias in the sense that none of these patients would have had any possibility of mortality during the course of neoadjuvant chemotherapy. We also do not have data on how many patients would have dropped out (developed metastatic disease) during neoadjuvant treatment. For these reasons, in an analysis starting at time of diagnosis, patients in this cohort would have thus had a built in survival advantage. On the other hand, using date of surgery, “takes” time away from these individuals in the sense that it excludes the time in chemotherapy from the survival calculations. At our institution we typically give short courses of neoadjuvant therapy: approximately two months of treatment with a third month left for recovery prior to surgery. While using the starting point for the survival analysis as the date of surgery could certainly introduce a bias that would reflect negatively on the survival statistics of patients in the vascular resection cohort, the observed difference in overall survival between the groups was wide enough (9 months) that it would be expected to continue to be significant even if neoadjuvant treatment time was added back to all affected vascular resection patients. In the end, we felt the least problematic method was to use the date of surgery as the start point for our survival calculation.
Plenary Presentation at 2015 SSAT Annual Meeting on May 19, 2015 in Washington, DC
Rights and permissions
About this article
Cite this article
Kantor, O., Talamonti, M.S., Stocker, S.J. et al. A Graded Evaluation of Outcomes Following Pancreaticoduodenectomy with Major Vascular Resection in Pancreatic Cancer. J Gastrointest Surg 20, 284–292 (2016). https://doi.org/10.1007/s11605-015-2957-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11605-015-2957-2