Abstract
Purpose
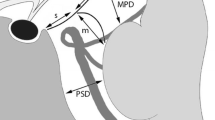
To clarify the detectability of the choroid plexus of the third ventricle (ChPl3V) with magnetic resonance ventriculography (MRVn) employing a steady-state free precession (SSFP) sequence in comparison to surgical endoscopic movies as a golden standard, as we encountered some clinical cases of total agenesis of corpus callosum (ACC) where we could not recognize the choroid plexus of the third ventricle and found no previous article addressing this problem.
Materials and methods
This retrospective study included consecutive patients from 2010 to 2016 for whom endoscopic evaluation of the third ventricle was conducted. The anterior portion of the right and left streaks of ChPl3V was evaluated in 8 patients on 16 sites, while the posterior portion of both streaks of ChPl3V was evaluated in 13 patients on 26 sites. Sensitivity of MRVn to visualize ChPl3V with endoscopic movies as the golden standard was calculated.
Results
Sensitivity of MRVn in visualizing the anterior portion of ChPl3V was 0.813, and that for the posterior portion 0.692. The anterior portion of ChPl3V was visualized in all cases where no tumor contacted the foramen of Monro.
Conclusion
MRVn visualizes the anterior portion of ChPl3V with significant sensitivity and the posterior portion with lower one.




Similar content being viewed by others
References
Loeser JD, Alvord EC Jr. Agenesis of the corpus callosum. Brain. 1968;91(3):553–70.
Netsky MG, Shuangshoti S. I origin of choroid plexus and ependyma. In: Netsky MG, Shuangshoti S, editors. The choroid plexus in health and disease. John Wright and Sons Ltd: Bristol; 1975. p. 3–18.
Bayer SA, Altman J. The human brain during the early first trimester. 1st ed., Atlas of Human Central Nervous System DevelopmentBoca Raton: CRC Press; 2008. p. 432–4. ISBN 978-08-4931-424-7.
Rakic P, Yakovlev PI. Development of the corpus callosum and cavum septi in man. J Comp Neurol. 1968;132(1):45–72.
Madhukar M, Choudhary AK, Boal DK, Dias MS, Iantosca MR. Choroid plexus: normal size criteria on neuroimaging. Surg Radiol Anat. 2012;34:887–95.
Casselman JW, Kuhweide R, Deimling M, Ampe W, Dehaene I, Meeus L. Constructive interference in steady state-3DFT MR imaging of the inner ear and cerebellopontine angle. AJNR Am J Neuroradiol. 1993;14:47–57.
Aleman J, Jokura H, Higano S, Akabane A, Shirane R, Yoshimoto T. Value of constructive interference in steady-state three-dimensional, Fourier transformation magnetic resonance imaging for the neuroendoscopic treatment of hydrocephalus and intracranial cysts. Neurosurgery. 2001;48(6):1291–5 (discussion 1295–6).
Seitz J, Held P, Strotzer M, Volk M, Nitz WR, Dorenbeck U, et al. MR imaging of cranial nerve lesions using six different high-resolution T1- and T2(*)-weighted 3D and 2D sequences. Acta Radiol. 2002;43(4):349–53.
Kocaoglu M, Bulakbasi N, Ucoz T, Ustunsoz B, Pabuscu Y, Tayfun C, et al. Comparison of contrast-enhanced T1-weighted and 3D constructive interference in steady state images for predicting outcome after hearing-preservation surgery for vestibular schwannoma. Neuroradiology. 2003;45:476–81.
Hatipoğlu HG, Durakoğlugil T, Ciliz D, Yüksel E. Comparison of FSE T2W and 3D FIESTA sequences in the evaluation of posterior fossa cranial nerves with MR cisternography. Diagn Interv Radiol. 2007;13(2):56–60.
Awaji M, Okamoto K, Nishiyama K. Magnetic resonance cisternography for preoperative evaluation of arachnoid cysts. Neuroradiology. 2007;49:721–6.
Linn J, Moriggl B, Schwarz F, Naidich TP, Schmid UD, Wiesmann M, et al. Cisternal segments of the glossopharyngeal, vagus, and accessory nerves: detailed magnetic resonance imaging-demonstrated anatomy and neurovascular relationships. J Nerosurg. 2009;110:1026–41.
Rhoton AL Jr. The lateral and third ventricles. Neurosurgery. 2002;51(4 Suppl):S207–71 (see Figure 5.12).
Oresković D, Klarica M. The formation of cerebrospinal fluid: nearly a hundred years of interpretations and misinterpretations. Brain Res Rev. 2010;64(2):241–62. https://doi.org/10.1016/j.brainresrev.2010.04.00.
Orešković D, Radoš M, Klarica M. Role of choroid plexus in cerebrospinal fluid hydrodynamics. Neuroscience. 2017;23(354):69–87. https://doi.org/10.1016/j.neuroscience.2017.04.025.
Spector R, Snodgrass SR, Johanson CE. A balanced view of the cerebrospinal fluid composition and functions: focus on adult humans. Exp Neurol. 2015;273:57–68. https://doi.org/10.1016/j.expneurol.2015.07.027.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical statement
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
About this article
Cite this article
Sato, K., Awaji, M., Inagawa, S. et al. Detectability of the choroid plexus of the third ventricle with magnetic resonance ventriculography. Jpn J Radiol 37, 449–457 (2019). https://doi.org/10.1007/s11604-019-00834-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11604-019-00834-2