Abstract
Objective
Ticagrelor is a widely used anti-platelet drug. However, the mechanisms by which ticagrelor protects against sepsis-induced acute kidney injury (AKI) have not been clearly demonstrated. We designed this study to explore the protective effect of ticagrelor on sepsis-induced AKI and to explore the underlying mechanisms.
Methods
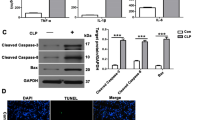
C57BL6J mice received oral ticagrelor (20 mg/kg and 50 mg/kg) for 7 days, and then caecal ligation and puncture (CLP) were performed. An adenosine receptor antagonist, CGS15943, was administered (10 mg/kg, intraperitoneal injection) to block the adenosine pathway 2 h before CLP. After 24 h, serum creatinine levels were measured. Periodic acid-Schiff (PAS) staining and terminal deoxynucleotidyl transferase-mediated dUTP nick end labeling (TUNEL) staining were employed to analyze pathological changes and cell apoptosis. Serum concentrations of tumor necrosis factor-α (TNF-α), interleukin-1β (IL-1β), and mRNA expression of tissue TNF-α and IL-1β were detected. Western blotting analysis was used to determine AKT and mammalian target of rapamycin (mTOR) protein expression in the kidney.
Results
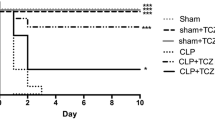
PAS staining showed less swelling of renal tubules, and TUNEL staining revealed less cell apoptosis in the ticagrelor group than in the CLP group. Serum creatinine levels were significantly lower in the ticagrelor group than in the CLP group. Moreover, significantly lower serum and kidney levels of TNF-α and IL-1β were observed in the ticagrelor group. CGS15943 blocked the effects of ticagrelor. Western blotting analysis showed increased phosphorylation of AKT and mTOR in the kidneys of the 50 mg/kg ticagrelor group. The adenosine receptor antagonist inhibited the activation of AKT and mTOR.
Conclusion
This study demonstrates that the protective effect of ticagrelor on sepsis-induced AKI depends on adenosine receptor activation and the subsequent increase of AKT and mTOR phosphorylation.
Similar content being viewed by others
References
Singer M, Deutschman CS, Seymour CW, et al. The Third International Consensus Definitions for Sepsis and Septic Shock (Sepsis-3). JAMA, 2016,315(8):801–810
Uchino S. The epidemiology of acute renal failure in the world. Curr Opin Crit Care, 2006,12(6):538–543
Langenberg C, Bagshaw SM, May CN. The histopathology of septic acute kidney injury: a systematic review. Crit Care, 2008,12(2):R38
Gomez H, Ince C, De Backer D, et al. A unified theory of sepsis-induced acute kidney injury: inflammation, microcirculatory dysfunction, bioenergetics, and the tubular cell adaptation to injury. Shock, 2014,41(1):3–11
Ortega-Paz L, Brugaletta S, Ariotti S, et al. Adenosine and ticagrelor plasma levels in patients with and without ticagrelor-related dyspnea. Circulation, 2018,138(6): 646–648
Nylander S, Femia EA, Scavone M, et al. Ticagrelor inhibits human platelet aggregation via adenosine in addition to P2Y12 antagonism. J Thromb Haemost, 2013,11(10):1867–1876
Armstrong D, Summers C, Ewart L, et al. Characterization of the adenosine pharmacology of ticagrelor reveals therapeutically relevant inhibition of equilibrative nucleoside transporter 1. J Cardiovasc Pharmacol Ther, 2014,19(2):209–219
Jiang Z, Zhang R, Sun M, et al. Effect of Clopidogrel vs. Ticagrelor on Platelet Aggregation and Inflammation Markers After Percutaneous Coronary Intervention for ST-Elevation Myocardial Infarction. Can J Cardiol, 2018,34(12):1606–1612
Sexton TR, Zhang G, Macaulay TE, et al. Ticagrelor Reduces Thromboinflammatory Markers in Patients With Pneumonia. JACC Basic Transl Sci, 2018,3(4):435–449
Li X, Li Y, Shen K, et al. The protective effect of ticagrelor on renal function in a mouse model of sepsis-induced acute kidney injury. Platelets, 2019,30(2):199–205
Nanhwan MK, Ling S, Kodakandla M, et al. Birnbaum. Chronic treatment with ticagrelor limits myocardial infarct size: an adenosine and cyclooxygenase-2-dependent effect. Arterioscler Thromb Vasc Biol, 2014,34(9):2078–2085
Harrois A, Grillot N, Figueiredo S, et al. Duranteau. Acute kidney injury is associated with a decrease in cortical renal perfusion during septic shock. Crit Care, 2018,22(1):161
Ma S, Evans RG, Iguchi N, et al. Sepsis-induced acute kidney injury: A disease of the microcirculation. Microcirculation, 2019,26(2):e12483
Gomez H, Kellum JA. Sepsis-induced acute kidney injury. Curr Opin Crit Care, 2016,22(6):546–553
Wallentin L, Becker RC, Budaj A, et al. Ticagrelor versus clopidogrel in patients with acute coronary syndromes. N Engl J Med, 2009,361(11):1045–1057
Ortega-Paz L, Brugaletta S, Ariotti S, et al. Adenosine and Ticagrelor Plasma Levels in Patients With and Without Ticagrelor-Related Dyspnea. Circulation, 2018,138(6):646–648
Bonello L, Laine M, Kipson N, et al. Ticagrelor increases adenosine plasma concentration in patients with an acute coronary syndrome. J Am Coll Cardiol, 2014,63(9):872–877
Toufektsian MC, Yang Z, Prasad KM, et al. Stimulation of A2A-adenosine receptors after myocardial infarction suppresses inflammatory activation and attenuates contractile dysfunction in the remote left ventricle. Am J Physiol Heart Circ Physiol, 2006,290(4):H1410–1418
Patinha D, Abreu C, Carvalho C, et al. Adenosine A2A and A3 Receptors as Targets for the Treatment of Hypertensive-Diabetic Nephropathy. Biomedicines, 2020,8(11):529
Pak ES, Jeong LS, Hou X, et al. Dual Actions of A2A and A3 Adenosine Receptor Ligand Prevents Obstruction-Induced Kidney Fibrosis in Mice. Int J Mol Sci, 2021,22(11):5667
Kim HJ, Kim SH, Kim M, et al. Inhibition of 15-PGDH prevents ischemic renal injury by the PGE2/EP4 signaling pathway mediating vasodilation, increased renal blood flow, and increased adenosine/A2A receptors. Am J Physiol Renal Physiol, 2020,319(6):F1054–F1066
Shi L, Wu Z, Miao J, et al. Adenosine interaction with adenosine receptor A2a promotes gastric cancer metastasis by enhancing PI3K-AKT-mTOR signaling. Mol Biol Cell, 2019,30(19):2527–2534
Bobermin LD, Roppa R, Quincozes-Santos A. Adenosine receptors as a new target for resveratrol-mediated glioprotection. Biochim Biophys Acta Mol Basis Dis, 2019,1865(3):634–647
Tian Y, Piras BA, Kron IL, et al. Adenosine 2B Receptor Activation Reduces Myocardial Reperfusion Injury by Promoting Anti-Inflammatory Macrophages Differentiation via PI3K/Akt Pathway. Oxid Med Cell Longev, 2015,2015:585297
Huang X, Wu P, Huang F, et al. Baicalin attenuates chronic hypoxia-induced pulmonary hypertension via adenosine A2A receptor-induced SDF-1/CXCR4/PI3K/AKT signaling. J Biomed Sci, 2017,24(1):52
Guo Z, Yu Q. Role of mTOR Signaling in Female Reproduction. Front Endocrinol (Lausanne), 2019,10: 692
Wei X, Luo L, Chen J. Roles of mTOR Signaling in Tissue Regeneration. Cells, 2019,8(9):1075
Correia B, Sousa MI, Ramalho-Santos J. The mTOR Pathway in Reproduction: From Gonadal Function to Developmental Coordination. Reproduction, 2020,159 (4):R173–R188
Chen LJ, Xu YL, Song B, et al. Angiotensin-converting enzyme 2 ameliorates renal fibrosis by blocking the activation of mTOR/ERK signaling in apolipoprotein E-deficient mice. Peptides, 2016,79:49–57
He Y, Lang X, Cheng D, et al. Curcumin Ameliorates Chronic Renal Failure in 5/6 Nephrectomized Rats by Regulation of the mTOR/HIF-1alpha/VEGF Signaling Pathway. Biol Pharm Bull, 2019,42(6):886–891
Y. Cao, Y. Wang, Y. Liu, et al. Decreased Expression of Urinary Mammalian Target of Rapamycin mRNA Is Related to Chronic Renal Fibrosis in IgAN. Dis Markers, 2019:2424751
Selman C, Withers DJ. Mammalian models of extended healthy lifespan. Philos Trans R Soc Lond B Biol Sci, 2011,366(1561):99–107
Temiz-Resitoglu M, Kucukkavruk SP, Guden DS, et al. Activation of mTOR/IkappaB-alpha/NF-kappaB pathway contributes to LPS-induced hypotension and inflammation in rats. Eur J Pharmacol, 2017,802:7–19
Fontana L, Partridge L, Longo VD. Extending healthy life span—from yeast to humans. Science, 2010,328 (5976):321–326
Gui Y, Lu Q, Gu M, et al. Fibroblast mTOR/PPARgamma/HGF axis protects against tubular cell death and acute kidney injury. Cell Death Differ, 2019, 26(12):2774–2789
Author information
Authors and Affiliations
Corresponding author
Additional information
Conflict of Interest Statement
The authors declare that there is no conflict of interest with any financial organization or corporation or individual that can inappropriately influence this work.
This study was supported by grants from the National Natural Science Foundation of China (No. 81700265 to Cong FU and No. 81702092 to Yu-han CAO).
Rights and permissions
About this article
Cite this article
Cao, Yh., Xu, Qc., Wang, Yw. et al. Ticagrelor Protects against Sepsis-Induced Acute Kidney Injury through an Adenosine Receptor-Dependent Pathway. CURR MED SCI 42, 505–512 (2022). https://doi.org/10.1007/s11596-022-2516-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11596-022-2516-5