Abstract
Objective
The characteristics of oxaliplatin-induced hypersensitivity reactions (HSRs) in Chinese patients were investigated to provide a reference for patients treated with oxaliplatin.
Methods
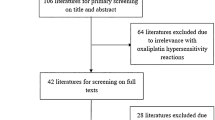
The study reviewed the records of patients who developed oxaliplatin-induced HSRs in 17 hospitals from May 2016 to May 2017. We collected and analyzed the basic information, history of oxaliplatin administration and premedication treatments, chemotherapy cycles, HSR symptoms, and the management and outcomes of these patients.
Results
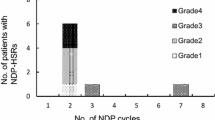
Oxaliplatin-induced HSRs were recorded in 137 patients who had been treated with oxaliplatin-containing regimens. Five different chemotherapy regimens were applied. The median infusion cycle when oxaliplatin-induced HSRs occurred was 7, and HSRs occurred during or shortly after oxaliplatin infusion. Most of the patients experienced grade 1 or grade 2 HSRs with mild symptoms of pruritis (49.64%), flushing (46.72%), chest discomfort (26.28%), and urticaria (25.55%). The majority of the patients completely recovered from HSRs following treatment with antihistamines and dexamethasone. Seven patients completed chemotherapy with oxaliplatin after the symptoms resolved with proper management.
Conclusion
The results indicate that oxaliplatin-induced HSRs remain an important issue in safely and successfully fulfilling oxaliplatin-containing chemotherapy. Further studies are needed to analyze the risk factors and establish prophylaxis for such reactions.
Similar content being viewed by others
References
Ohta H, Hayashi T, Murai S, et al. Comparison between hypersensitivity reactionstocyclesofmodifiedFOLFOX6 and XELOX therapies in patients with colorectal cancer. Cancer Chemother Pharmacol, 2017,79(5):1021–1029
Lee SY, Kang HR, Song WJ, et al. Overcoming oxaliplatin hypersensitivity: different strategies are needed according to the severity and previous exposure. Cancer Chemother Pharmacol, 2014,73(2014):1021–1029
Yamauchi H, Goto T, Takayoshi K, et al. A retrospective analysis of the risk factors for allergic reactions induced by the administration of oxaliplatin. Eur J Cancer Care, 2015,24(1):111–116
Maindrault-Goebel F, André T, Tournigand C, et al. Allergic-type reactions to oxaliplatin: Retrospective analysis of 42 patients. Eur J Cancer, 2005,41(15):2262–2267
Park SJ, Lee KY, Park WS, et al. Clinical Outcomes of Reintroducing Oxaliplatin to Patients with Colorectal Cancer after Mild Hypersensitivity Reactions. Oncology, 2013,85(6):323–327
Parel M, Ranchon F, Nosbaum A, et al. Hypersensitivity to oxaliplatin: clinical features and risk factors. BMC Pharmacol Toxicol, 2014,15:1
Aroldi F, Prochilo T, Bertocchi P, et al. Oxaliplatin-induced hypersensitivity reaction: underlying mechanisms and management. J Chemother, 2015,27(2):63–66
Kolomeyevskaya NV, Lele SB, Miller A, et al. Oxaliplatin Is a Safe Alternative Option for Patients with Recurrent Gynecologic Cancers After Hypersensitivity Reaction to Carboplatin. Int J Gynecol Cancer, 2015,25(1):42–48
Kitada N, Dan T, Takara K, et al. CASE REPORT Oxaliplatin-induced hypersensitivity reaction displaying marked elevation of immunoglobulin E. J Oncol Pharm Practice, 2007,13(2007):233–235
Kobayashi T, Masaki T, Kogawa K, et al. Hemoptysis and Acute Respiratory Syndrome (ARDS) as Delayed-Type Hypersensitivity After FOLFOX4 Plus Bevacizumab Treatment. Int Surg, 2013,98(2013):445–449
Bautista MA, Bautista MA, Stevens WT, et al. Hypersensitivity reaction and acute immune mediated thrombocytopenia from oxaliplatin: two case reports and a review of the literature. J Hematol Oncol, 2010, 12(3):1–8
Bano N, Najam R, Qazi F, et al. Clinical Features of Oxaliplatin Induced Hypersensitivity Reactions and Therapeutic Approaches. Asian Pac J Cancer Prev, 2016, 17(4):1637–1641
Saif MW. Hypersensitivity reactions associated with oxaliplatin. Expert Opion Drug Saf, 2006,5(5):687–694
Kim BH, Bradley T, Tai J, et al. Hypersensitivity to Oxaliplatin: An Investigation of Incidence and Risk Factors, and Literature Review. Oncology, 2009,76:231–238
Lenza G, Hackera UT, Kern W, et al. Adverse reactions to oxaliplatin: a retrospective study of 25 patients treated in one institution. Anti-Cancer Drugs, 2003,14(2013):731–733
Polyzos A, Tsavaris N, Gogas H, et al. Clinical Features of Hypersensitivity Reactions to Oxaliplatin: A 10-Year Experience. Oncology, 2008,76(1):36–41
Makrilia N, Syrigou E, Kaklamanos I, et al. Hypersensitivity reactions associated with platinum antineoplastic agents: a systematic review. Met Based Drugs, 2010,2010:207084
Nozawa H, Muto Y, Yamada Y. Desensitization to Oxaliplatin with Two Stages of Premedication in a Patient with Metastatic Rectal Cancer. Clin Therap, 2008,30(6):1160–1165
Toki MI, Saif MW, Syrigos KN. Hypersensitivity reactions associated with oxaliplatin and their clinical management. Expert Opion Drug Saf, 2014,13(11):1545–1554
Gammon D, Bhargava P, Mccormick MJ. Hypersensitivity Reactions to Oxaliplatin and the Application of a Desensitization Protocol. Oncologist, 2004,9:546–549
Syrigou EI, Karapanagiotou EM, Alamara CV, et al. Hypersensitivity Reactions to Oxaliplatin: A Retrospective Study and the Development of a Desensitization Protocol. Clin Colorectal Cancer, 2009, 8(2):106–109
Siu SWK, Chan RTT, Au GKH. Hypersensitivity reactions to oxaliplatin: experience in a single institute. Ann Oncol, 2006,17:259–261
Waddle M, Irvin M, Gupta E, et al. Acute Disseminated Intravascular Coagulation after Oxaliplatin Infusion. Chemotherapy, 2017,62(5):295–300
Lai J, Wang W. Acute hemolysis after receiving oxaliplatin treatment: a case report and literature review. Pharm World Sci, 2009,31(5):538–541
Garciaz S, Oziel-Taieb S, Dermeche S, et al. Acute immune hematological complication of oxaliplatin. A series of 3 cases. Tumori, 2014,100(1):e17–e19
Phull P, Quillen K, Hartshorn KL. Acute Oxaliplatin-induced Hemolytic Anemia, Thrombocytopenia, and Renal Failure: Case Report and a Literature Review. Clin Colorectal Cancer, 2017,16(2):e105–e107
González-Mahave I, Lobera Iabairu T, Blasco Sarramián A, et al. Anaphylaxis produced by oxaliplatin. J Investig Allergol Clin immunol, 2005,15(1):75
Shao Y, Hong R. Fatal Thrombocytopenia after Oxaliplatin-based Chemotherapy. Anticancer Res, 2008, 28(2008):3115–3118
Kidera Y, Satoh T, Ueda S, et al. High-dose dexamethasone plus antihistamine prevents colorectal cancer patients treated with modified FOLFOX6 from hypersensitivity reactions induced by oxaliplatin. Int J Clin Oncol, 2011,16(2011):244–249
Pagani M, Bonadonna P. Skin Test Protocol for the Prevention of Hypersensitivity Reactions to Oxaliplatin. Anticancer Res, 2014,34(2014):537–540
Garufi C, Cristaudo A, Vanni B, et al. Skin testing and hypersensitivity reactions to oxaliplatin. Ann Oncol, 2003,14:497–498
Zhang X, Zhao Y, Zheng Y, et al. The Effects of Prolonged Infusion on Reducing Oxaliplatin Hypersensitivity Reactions. J Investig Allergol Clin Immunol, 2017,27(1):65–66
Yanai T, Iwasa S, Hashimoto H, et al. Successful Rechallenge for Oxaliplatin Hypersensitivity Reactions in Patients with Metastatic Colorectal Cancer. Anticancer Res, 2012,32:5521–5526
Rose PG, Metz C, Link N. Desensitization With Oxaliplatin in Patients Intolerant of Carboplatin Desensitization. Int J Gynecoll Cancer, 2014,24(9):1603–1606
Edmondson DA, Gruling BJ, Urmanski AM, et al. Oxaliplatin Hypersensitivity: Case Report and Successful Repeat Desensitization. Am J Therapeut, 2007,14(2007):116–118
Wong JT, Ling M, Patil S, et al. Oxaliplatin Hypersensitivity: Evaluation, Implications of Skin Testing, and Desensitization. J Allergy Clin Immunol Pract, 2013,2(1):40–45
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
There is no conflict of interest in the submitted work.
Additional information
This work was supported by grants from the Hubei Center for Adverse Drug Reaction Monitoring (No. 20160422), and the Research-oriented Clinician Plan of Tongji Medical College, Huazhong University of Science and Technology (No. 5001540076).
Rights and permissions
About this article
Cite this article
Li, M., Jiang, C., Yang, Jw. et al. Clinical Features of Oxaliplatin-induced Hypersensitivity Reactions in Chinese Patients: A Retrospective Multicenter Analysis. CURR MED SCI 41, 827–831 (2021). https://doi.org/10.1007/s11596-021-2387-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11596-021-2387-1