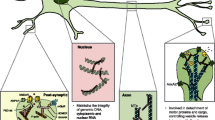
Summary
As a major microtubule-associated protein, tau plays an important role in promoting microtubule assembly and stabilizing microtubules. In Alzheimer’s disease (AD) and other tauopathies, the abnormally hyperphosphorylated tau proteins are aggregated into paired helical filaments and accumulated in the neurons with the form of neurofibrillary tangles. An imbalanced regulation in protein kinases and protein phosphatases is the direct cause of tau hyperphosphorylation. Among various kinases and phosphatases, glycogen synthase kinase-3β (GSK-3β) and protein phosphatase 2A (PP2A) are the most implicated. Accumulation of the hyperphosphorylated tau induces synaptic toxicity and cognitive impairments. Here, we review the upstream factors or pathways that can regulate GSK-3β or PP2A activity mainly based on our recent findings. We will also discuss the mechanisms that may underlie tau-induced synaptic toxicity.
Similar content being viewed by others
References
Li HL, Wang HH, Liu SJ, et al. Phosphorylation of tau antagonizes apoptosis by stabilizing beta-catenin, a mechanism involved in Alzheimer’s neurodegeneration. Proc Natl Acad Sci USA, 2007,104(9):3591–3596
Liu XA, Song J, Jiang Q, et al. Expression of the hyperphosphorylated tau attenuates ER stress-induced apoptosis with upregulation of unfolded protein response. Apoptosis, 2012,17(10):1039–1049
Cohen TJ, Friedmann D, Hwang AW, et al. The microtubule-associated tau protein has intrinsic acetyltransferase activity. Nat Struct Mol Biol, 2013,20(6):756–762
Grundke-Iqbal I, Iqbal K, Tung YC, et al. Abnormal phosphorylation of the microtubule-associated protein tau (tau) in Alzheimer cytoskeletal pathology. Proc Natl Acad Sci USA, 1986,83(13):4913–4917
Thal DR, Holzer M, Rüb U, et al. Alzheimer-related tau-pathology in the perforant path target zone and in the hippocampal stratum oriens and radiatum correlates with onset and degree of dementia. Exp Neurol, 2000,163(1):98–110
Wang JZ, Liu F. Microtubule-associated protein tau in development, degeneration and protection of neurons. Prog Neurobiol, 2008,85(2):148–175
Wang JZ, Grundke-Iqbal I, Iqbal K. Kinases and phosphatases and tau sites involved in Alzheimer neurofibrillary degeneration. Eur J Neurosci, 2007,25(1):59–68
Jacobs KM, Bhave SR, Ferraro DJ, et al. GSK-3ß: A bifunctional role in cell death pathways. Int J Cell Biol, 2012,2012:930710
Dominguez I, Itoh K, Sokol SY. Role of glycogen synthase kinase 3 beta as a negative regulator of dorsoventral axis formation in Xenopus embryos. Proc Natl Acad Sci USA, 1995,92(18):8498–8502
Jiang J, Wang ZH, Qu M, et al. Stimulation of EphB2 attenuates tau phosphorylation through PI3K/Akt-mediated inactivation of glycogen synthase kinase-3ß. Sci Rep, 2015,5:11765
Liu D, Wei N, Man HY, et al. The MT2 receptor stimulates axonogenesis and enhances synaptic transmission by activating Akt signaling. Cell Death Differ, 2015,22(4):583–596
Adi S, Wu NY, Rosenthal SM. Growth factor-stimulated phosphorylation of Akt and p70(S6K) is differentially inhibited by LY294002 and Wortmannin. Endocrinology, 2001,142(1):498–501
Liu SJ, Zhang JY, Li HL, et al. Tau becomes a more favorable substrate for GSK-3 when it is prephosphorylated by PKA in rat brain. J Biol Chem, 2004,279(48):50078–50088
Liu SJ, Zhang AH, Li HL, et al. Overactivation of glycogen synthase kinase-3 by inhibition of phosphoinositol-3 kinase and protein kinase C leads to hyperphosphorylation of tau and impairment of spatial memory. J Neurochem, 2003,87(6):1333–1344
Wang XL, Zeng J, Yang Y, et al. Helicobacter pylori filtrate induces Alzheimer-like tau hyperphosphorylation by activating glycogen synthase kinase-3ß. J Alzheimers Dis, 2015,43(1):153–165
Wu YY, Wang X, Tan L, et al. Lithium attenuates scopolamine-induced memory deficits with inhibition of GSK-3ß and preservation of postsynaptic components. J Alzheimers Dis, 2013,37(3):515–527
Liu ZC, Fu ZQ, Song J, et al. Bip enhanced the aßsociation of GSK-3ß with tau during ER stress both in vivo and in vitro. J Alzheimers Dis, 2012,29(4):727–740
Li XH, Xie JZ, Jiang X, et al. Methylglyoxal induces tau hyperphosphorylation via promoting AGEs formation. Neuromolecular Med, 2012,14(4):338–348
Li XH, Du LL, Cheng XS, et al. Glycation exacerbates the neuronal toxicity of ß-amyloid. Cell Death Dis, 2013,4: e673
Hu J, Huang HZ, Wang X, et al. Activation of glycogen synthase kinase-3 mediates the olfactory deficit-induced hippocampal impairments. Mol Neurobiol, 2015,52(3):1601–1617
Xiong YS, Wang DL, Tan L, et al. Inhibition of glycogen synthase kinase-3 reverses tau hyperphosphorylation induced by Pin1 down-regulation. CNS Neurol Disord Drug Targets, 2013,12(3):436–443
Xu ZP, Yang SL, Zhao S, et al. Biomarkers for early diagnostic of mild cognitive impairment in type-2 diabetes patients: a multicentre, retrospective, nested case-control study. EBioMedicine, 2016,5:105–113
Wang JZ, Gong CX, Zaidi T, et al. Dephosphorylation of Alzheimer paired helical filaments by protein phosphatase-2A and -2B. J Biol Chem, 1995,270(9):4854–4860
Gong CX, Singh TJ, Grundke-Iqbal I, et al. Phosphoprotein phosphatase activities in Alzheimer disease brain. J Neurochem, 1993,61(3):921–927
Chai GS, Jiang X, Ni ZF, et al. Betaine attenuates Alzheimer-like pathological changes and memory deficits induced by homocysteine. J Neurochem, 2013,124(3):388–396
Zhou P, Chen Z, Zhao N, et al. Acetyl-L-carnitine attenuates homocysteine-induced Alzheimer-like histopathological and behavioral abnormalities. Rejuvenation Res, 2011,14(6):669–679
Wei W, Liu YH, Zhang CE, et al. Folate/vitamin-B12 prevents chronic hyperhomocysteinemia-induced tau hyperphosphorylation and memory deficits in aged rats. J Alzheimers Dis, 2011,27(3):639–650
Zhang CE, Tian Q, Wei W, et al. Homocysteine induces tau phosphorylation by inactivating protein phosphatase 2A in rat hippocampus. Neurobiol Aging, 2008,29(11):1654–1665
Liu X, Wang Z, Xia Y, et al. Losartan-induced hypotension leads to tau hyperphosphorylation and memory deficit. J Alzheimers Dis, 2014,40(2):419–427
Xiong Y, Jing XP, Zhou XW, et al. Zinc induces protein phosphatase 2A inactivation and tau hyperphosphorylation through Src dependent PP2A (tyrosine 307) phosphorylation. Neurobiol Aging, 2013,34(3):745–756
Sun XY, Wei YP, Xiong Y, et al. Synaptic released zinc promotes tau hyperphosphorylation by inhibition of protein phosphatase 2A ( PP2A). J Biol Chem, 2012,287(14):11174–11182
Yu G, Yan T, Feng Y, et al. Ser9 phosphorylation causes cytoplasmic detention of I2PP2A/SET in Alzheimer disease. Neurobiol Aging, 2013,34(7):1748–1758
Luo Y, Nie YJ, Shi HR, et al. PTPA activates protein phosphatase-2A through reducing its phosphorylation at tyrosine-307 with upregulation of protein tyrosine phos-phatase 1B. Biochim Biophys Acta, 2013,1833(5):1235–1243
Luo DJ, Feng Q, Wang ZH, et al. Knockdown of phospho-tyrosyl phosphatase activator induces apoptosis via mito-chondrial pathway and the attenuation by simultaneous tau hyperphosphorylation. J Neurochem, 2014,130(6):816–825
Cheng XS, Zhao KP, Jiang X, et al. Nmnat2 attenuates Tau phosphorylation through activation of PP2A. J Alzheimers Dis, 2013,36(1):185–195
Chen NN, Luo DJ, Yao XQ, et al. Pesticides induce spatial memory deficits with synaptic impairments and an imbal-anced tau phosphorylation in rats. J Alzheimers Dis, 2012,30(3):585–594
Zhao HH, Di J, Liu WS, et al. Involvement of GSK3 and PP2A in ginsenoside Rb1’s attenuation of alumi-num-induced tau hyperphosphorylation. Behav Brain Res, 2013,241:228–234
Zhang CE, Yang X, Li L, et al. Hypoxia-induced tau phosphorylation and memory deficit in rats. Neurodegener Dis, 2014,14(3):107–116
Cui B, Zhu L, She X, et al. Chronic noise exposure causes persistence of tauhyperphosphorylation and formation of NFT tau in the rat hippocampus and prefrontal cortex. Exp Neurol, 2012,238(2):122–129
Huang C, Ng OT, Ho YS, et al. Effect of continuous propofol infusion in rat on tau phosphorylation with or without temperature control. J Alzheimers Dis, 2016,51(1):213–226
Nicolia V, Fuso A, Cavallaro RA, et al. B vitamin deficiency promotes tau phosphorylation through regulation of GSK3beta and PP2A. J Alzheimers Dis, 2010,19(3):895–907
Bereczki E, Re F, Maßserini ME, et al. Liposomes functionalized with acidic lipids rescue Aß-induced toxicity in murine neuroblastoma cells. Nanomedicine, 2011,7(5):560–571
Killick R, Scales G, Leroy K, et al. Deletion of Irs2 reduces amyloid deposition and rescues behavioural deficits in APP transgenic mice. Biochem Biophys Res Commun, 2009, 386(1):257–262
Xiong YS, Liu FF, Liu D, et al. Opposite effects of two estrogen receptors on tau phosphorylation through disparate effects on the miR-218/PTPA pathway. Aging Cell, 2015,14(5):867–877
Wu YY, Wang X, Tan L, et al. Lithium attenuates scopolamine-induced memory deficits with inhibition of GSK-3ß and preservation of postsynaptic components. J Alzheimers Dis, 2013,37(3):515–527
Liu GP, Zhang Y, Yao XQ, et al. Activation of glycogen synthase kinase-3 inhibits protein phosphatase-2A and the underlying mechanisms. Neurobiol Aging, 2008,29(9):1348–1358
Yao XQ, Zhang XX, Yin YY, et al. Glycogen synthase kinase-3ß regulates Tyr307 phosphorylation of protein phosphatase-2A via protein tyrosine phosphatase 1B but not Src. Biochem J, 2011,437(2):335–344
Yao XQ, Li XC, Zhang XX, et al. Glycogen synthase kinase-3ß regulates leucine-309 demethylation of protein phosphatase-2A via PPMT1 and PME-1. FEBS Lett, 2012,586(16):2522–2528
Zhang Y, Ma RH, Li XC, et al. Silencing rescues tau pathologies and memory deficits through rescuing pp2a and inhibiting gsk-3ß signaling in human tau transgenic mice. Front Aging Neurosci, 2014,6:123
Qian W, Shi J, Yin X, et al. PP2A regulates tau phosphorylation directly and also indirectly via activating GSK-3beta. J Alzheimers Dis, 2010, 19(4):1221–1229
Martin L, Magnaudeix A, Esclaire F, et al. Inhibition of glycogen synthase kinase-3beta downregulates total tau proteins in cultured neurons and its reversal by the blockade of protein phosphatase-2A. Brain Res, 2009, 1252:66–75
Arif M, Wei J, Zhang Q, et al. Cytoplasmic retention of protein phosphatase 2A inhibitor 2 (I2PP2A) induces Alzheimer-like abnormal hyperphosphorylation of Tau. J Biol Chem, 2014,289(40):27677–91.
Liu GP, Wei W, Zhou X, et al. I(2)(PP2A) regulates p53 and Akt correlatively and leads the neurons to abort apoptosis. Neurobiol Aging, 2012,33(2):254–264
Liu GP, Wei W, Zhou X, et al. Silencing PP2A inhibitor by lenti-shRNA interference ameliorates neuropathologies and memory deficits in tg2576 mice. Mol Ther, 2013,21(12):2247–2257
Gräff J, Kim D, Dobbin MM, et al. Epigenetic regulation of gene expression in physiological and pathological brain processes. Physiol Rev, 2011,91(2):603–649
Seo SB, McNamara P, Heo S, et al. Regulation of histone acetylation and transcription by INHAT, a human cellular complex containing the set oncoprotein. Cell, 2001,104(1):119–130
Seo SB, Macfarlan T, McNamara P, et al. Regulation of histone acetylation and transcription by nuclear protein pp32, a subunit of the INHAT complex. J Biol Chem, 2002,277(16):14005–14010
Dickstein DL, Brautigam H, Stockton SD, et al. Changes in dendritic complexity and spine morphology in transgenic mice expressing human wild-type tau. Brain Struct Funct, 2010,214(2–3):161–179
Sydow A, Van der Jeugd A, Zheng F, et al. Tau-induced defects in synaptic plasticity, learning, and memory are reversible in transgenic mice after switching off the toxic Tau mutant. J Neurosci, 2011,31(7):2511–2525
Van der Jeugd A, Hochgräfe K, Ahmed T, et al. Cognitive defects are reversible in inducible mice expressing pro-aggregant full-length human Tau. Acta Neuropathol, 2012,123(6):787–805
Hu Y, Li XC, Wang ZH, et al. Tau accumulation impairs mitophagy via increasing mitochondrial membrane potential and reducing mitochondrial Parkin. Oncotarget, 2016,7(14):17356–17368
Mao K, Klionsky DJ. Mitochondrial fission facilitates mitophagy in Saccharomyces cerevisiae. Autophagy, 2013, 9(11):1900–1901
Li XC, Hu Y, Wang ZH, et al. Human wild-type full-length tau accumulation disrupts mitochondrial dynamics and the functions via increasing mitofusins. Sci Rep, 2016,6:24756
Luo HB, Xia YY, Shu XJ, et al. SUMOylation at K340 inhibits tau degradation through deregulating its phosphorylation and ubiquitination. Proc Natl Acad Sci USA, 2014,111(46):16586–16591
Yin Y, Gao D, Wang Y, et al. Tau accumulation induces synaptic impairment and memory deficit by calcineurin-mediated inactivation of nuclear CaMKIV/CREB signaling. Proc Natl Acad Sci USA, 2016,113(26): E3773–E3781
Wang L, Jiang Q, Chu J, et al. Expression of Tau40 induces activation of cultured rat microglial cells. PLoS One, 2013,8(10):e76057
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Ma, Rh., Zhang, Y., Hong, Xy. et al. Role of microtubule-associated protein tau phosphorylation in Alzheimer’s disease. J. Huazhong Univ. Sci. Technol. [Med. Sci.] 37, 307–312 (2017). https://doi.org/10.1007/s11596-017-1732-x
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11596-017-1732-x