Abstract
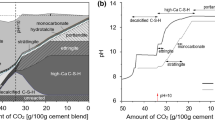
The experimental and modeling approaches were taken to study the deterioration mechanism of concrete under acid rain attack. Concrete specimens were prepared and exposed to the simulated acid rain environment. The neutralization depth of concrete was measured, and the mineralogical composition and microstructure of concrete were analyzed using X-ray diffraction (XRD) and scanning electron microscope (SEM). The experimental results show that the degradation of concrete increases with the corrosion time and the decrease in pH value of acidic solution from 3.5 to 1.5. Concrete was corroded by H+ and SO42− in acid rain, producing gypsum and leading to the neutralization of concrete. The acid rain exposure also resulted in the decomposition of hydration products of cement, such as C-S-H and ettringite, forming the main corrosion products of gypsum and SiO2·nH2O. Based on the second Fick diffusion law, a model was developed to describe the deterioration mechanism of concrete exposed to acid rain mathematically, coupling the diffusion of reactive ions and the corrosion reaction. The simulation results and the experimental results were compared and discussed.
Similar content being viewed by others
References
Tsouli S, Lekatou A G, Kleftakis S, et al. Corrosion Behavior of 304L Stainless Steel Concrete Reinforcement in Acid Rain Using Fly Ash as Corrosion Inhibitor[J]. Procedia Structural Integrity, 2018, 10: 41–48
Okochi H, Kameda H, Hasegawa S, et al. Deterioration of Concrete Structures by Acid Deposition-an Assessment of the Role of Rainwater on Deterioration by Laboratory and Field Exposure Experiments Using Mortar Specimens[J]. Atmospheric Environment, 2000, 34(18): 2 937–2 945
Zhou C L, Zhu Z M, Zhu A J, et al. Deterioration of Mode II Fracture Toughness, Compressive Strength and Elastic Modulus of Concrete Under the Environment of Acid Rain and Cyclic Wetting-Drying[J]. Construction and Building Materials, 2019, 228: 116–130
Tripathi D, Kumar R, Mehta P K, et al. Silica Fume Mixed Concrete in Acidic Environment[J]. Materials Today: Proceedings, 2020, 27(2): 1 001–1 005
Sahoo S, Mahapatra TR, Priyadarshini N, et al. Influence of Water Binder Ratio on Strength and Acid Resistance of Concrete Made Up of Mineral Admixture As Supplementary Cementitious Material[J]. Materials Today: Proceedings, 2020, 26(2): 796–803
Xie S D, Qi L, Zhou D. Investigation of the Effects of Acid Rain on the Deterioration of Cement Concrete Using Accelerated Tests Established in Laboratory[J]. Atmospheric Environment, 2004, 38(2): 4 457–4 466
Chen MC, Wang K, Xie L. Deterioration Mechanism of Cementitious Materials Under Acid Rain Attack[J]. Engineering Failure Analysis, 2013, 27: 272–285
Zhang X M, Chai F H, Wang S L, et al. Research Progress of Acid Precipitation in China[J]. Research of Environmental Sciences, 2010, 23(5): 527–532 (in Chinese)
Fang K H, He Z. Building Material (Seventh Edition)[M]. Beijing: China Water Resources and Hydropower Press, 2015 (in Chinese)
Weng K M, Min P R, Wang G B. A Study on Carbonation of Hydrates of Calcium Aluminate[J]. Journal of the Chinese Ceramic Society, 1989, 17(2): 173–180 (in Chinese)
Li B X, Cai L H, Wang K, et al. Prediction of the Residual Strength for Durability Failure of Concrete Structure in Acidic Environments[J]. Journal of Wuhan University of Technology-Mater. Sci. Ed., 2016, 31(2): 340–344
Gospodinov P N, Kazandjiev R F, Mironova M. The Effect of Sulfate Ion Diffusion on the Structure of Cement Stone[J]. Cement and Concrete Composites, 1996, 18(6): 401–407
Funding
Funded by the National Key Research and Development Program of China (No. 2020YFC1909904) and the National Natural Science Foundation of China (No. 51372185)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Li, B., Yuan, X., Zhang, Y. et al. Deterioration Mechanism of Concrete Subjected to Acid Rain Attack—An Experimental and Modeling Approach. J. Wuhan Univ. Technol.-Mat. Sci. Edit. 37, 849–854 (2022). https://doi.org/10.1007/s11595-022-2606-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11595-022-2606-z