Abstract
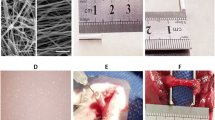
Using nerve guide conduits (NGCs) to promote the regeneration of PNI is a feasible alternative to autograft. Compared with NGCs made of single material, composite NGCs have a greater development prospect. Our previous research has confirmed that poly(D, L-lactic acid)/β-tricalcium phosphate/hyaluronic acid/chitosan/nerve growth factor (PDLLA/β-TCP/HA/CHS/NGF) NGCs have excellent physical and chemical properties, which can slowly release NGF and support cell adhesion and proliferation. In this study, PDLLA/β-TCP/HA/CHS/NGF NGCs were prepared and used to bridge a 10 mm sciatic nerve defect in 200–250 g Sprague-Dawley (SD) rat to verify the performance of the NGCs in vivo. Substantial improvements in nerve regeneration were observed after using the PDLLA/β-TCP/HA/CHS/NGF NGCs based on gross post-operation observation, triceps wet weight analysis and nerve histological assessment. In vivo studies illustrate that the PDLLA/β-TCP/HA/CHS/NGF sustained-release NGCs can effectively promote peripheral nerve regeneration, and the effect is similar to that of autograft.
Article PDF
Similar content being viewed by others
References
Lim EMF, Nakanishi ST, Hoghooghi V, et al. AlphaB-crystallin Regulates Remyelination After Peripheral Nerve Injury[J]. Proc. Natl. Acad. Sci., 2017, 114(9): 1 707–1 716
Vijayavenkataraman S. Nerve Guide Conduits for Peripheral Nerve Injury Repair: A Review on Design, Materials and Fabrication Methods[J]. Acta Biomater., 2020, 106: 54–69
Wiberg M, Terenghi GJSTI. Will it be Possible to Produce Peripheral Nerves?[J]. Surg. Technol. Int., 2003, 11: 303–310
Yegiyants S, Dayicioglu D, Kardashian G, et al. Traumatic Peripheral Nerve Injury: A Wartime Review[J]. J. Craniofac. Surg., 2010, 21(4): 998–1 001
Li R, Liu Z, Pan Y, et al. Peripheral Nerve Injuries Treatment: a Systematic Review[J]. Cell Biochem. Biophys., 2014, 68(3): 449–454
Ray WZ, Mackinnon SE. Management of Nerve Gaps: Autografts, Allografts, Nerve Transfers, and End-to-side Neurorrhaphy[J]. Exp. Neurol., 2010, 223(1): 77–85
Lin YC, Marra KG. Injectable Systems and Implantable Conduits for Peripheral Nerve Repair[J]. Biomed. Mater., 2012, 7(2): SI
Moore AM, Ray WZ, Chenard KE, et al. Nerve Allotransplantation as It Pertains to Composite Tissue Transplantation[J]. Hand, 2009, 4(3): 239–244
Grinsell D, Keating CP. Peripheral Nerve Reconstruction after Injury: A Review of Clinical and Experimental Therapies[J]. Biomed Res. Int., 2014, 698256
Muheremu A, Ao Q. Past, Present, and Future of Nerve Conduits in the Treatment of Peripheral Nerve Injury[J]. Biomed Res. Int., 2015, 237507
Zhang ES, Zhu CS, Yang J, et al. Electrospun PDLLA/PLGA Composite Membranes for Potential Application in Guided Tissue Regeneration[J]. Mater. Sci. Eng., 2016, 58(Jan.): 278–285
Guo XL, Liu G, He QD, et al. The Study of Nerve Conduit with Biocompatibility and Electrical Stimulation Effect[J]. Journal of Wuhan University of Technol.-Mater. Sci. Ed., 2018, 33(6):1 530–1 539
Kim K, Yu M, Zong XH, et al. Control of Degradation Rate and Hydrophilicity in Electrospun Non-woven Poly(d,l-lactide) Nanofiber Scaffolds for Biomedical Applications[J]. Biomaterials, 2003, 24(27): 4 977–4 985
Nair LS, Laurencin CT. Biodegradable Polymers as Biomaterials[J]. Prog. Polym. Sci., 2007, 32(8–9): 762–798
Cui ZQ, Zhang YK, Cheng YL, et al. Applications, Microstructure, mechanical, Corrosion Properties and Cytotoxicity of Beta-calcium Polyphosphate Reinforced ZK61 Magnesium Alloy Composite by Spark Plasma Sintering[J]. Mater. Sci. Eng. C: Mater. Biol. Appl., 2019, 99: 1 035–1 047
Gilarska A, Lewandowska-Lancucka J, Guzdek-Zajac K, et al. Bioactive Yet Antimicrobial Structurally Stable Collagen/Chitosan/Lysine Functionalized Hyaluronic Acid-based Injectable Hydrogels for Potential Bone Tissue Engineering Applications[J]. Int. J. Biol. Macromol., 2019, 155: 938–950
Xu X, Jha AK, Harrington DA, et al. Hyaluronic Acid-based Hydrogels: From a Natural Polysaccharide to Complex Networks[J]. Soft Matter, 2012, 8(12): 3 280–3 294
LogithKumar R, KeshavNarayan A, Dhivya S, et al. A Review of Chitosan and Its Derivatives in Bone Tissue Engineering[J]. Carbohydr. Polym., 2016, 151: 172–188
Zhang Z, Li X, Li Z, et al. Collagen/nano-sized β-tricalcium Phosphate Conduits Combined with Collagen Filaments and Nerve Growth Factor Promote Facial Nerve Regeneration in Miniature Swine: An in vivo Study[J]. Oral Surg., Oral Med., Oral Pathol. Oral Radiol., 2019, 128(5): 472–478
Mcmahon SB, Bennett DLH, Priestley JV, et al. The Biological Effects of Endogenous Nerve Growth Factor on Adult Sensory Neurons Re-Vealed by a TrkA-IgG Fusion Molecule[J]. Nat. Med., 1995, 1(8): 774–780
Eggers R, Winter FD, Hoyng SA, et al. Lentiviral Vector-Mediated Gradients of GDNF in the Injured Peripheral Nerve: Effects on Nerve Coil Formation, Schwann Cell Maturation and Myelination[J]. Plos One, 2013, 8(8): e71076
Moattari M, Kouchesfehani HM, Kaka G, et al. Evaluation of Nerve Growth Factor (NGF) Treated Mesenchymal Stem Cells for Recovery in Neurotmesis Model of Peripheral Nerve Injury[J]. J. Cranio-Maxillo-Facial Surg., 2018, 46(6): 898–904
Granholm AC, Reyland M, Albeck D, et al. Glial Cell Line-Derived Neurotrophic Factor Is Essential for Postnatal Survival of Midbrain Dopamine Neurons[J]. J. Neurosci., 2000, 20(9): 3 182–3 190
Tria MA, Fusco M, Vantini G, et al. Pharmacokinetics of Nerve Growth Factor (NGF) Following Different Routes of Administration to Adult Rats[J]. Exp. Neurol., 1994, 127(2): 178–183
Kearney CJ, Mooney DJ. Macroscale Delivery Systems for Molecular and Cellular Payloads[J]. Nat. Mater., 2013, 12(11): 1 004–1 017
Lackington WA, Ryan AJ, O’Brien FJ. Advances in Nerve Guidance Conduit-Based Therapeutics for Peripheral Nerve Repair[J]. ACS Biomater. Sci. Eng., 2016, 3(7): 1 221–1 235
Li YP, Liu H, Xu HX, et al. Preparation and Characterization of a Conduit for Peripheral Nerve Regeneration[J]. Cur. Appl. Polym. Sci., 2017, 1(1): 35–44
Author information
Authors and Affiliations
Corresponding authors
Additional information
Funded by the Chinese National Natural Science Foundation (No. 51572206), and the National Innovation and Entrepreneurship Training Program for College (Nos. 202110497032, 202110497033, and S202110497080)
Rights and permissions
About this article
Cite this article
Yan, X., Wang, J., He, Q. et al. PDLLA/β-TCP/HA/CHS/NGF Sustained-release Conduits for Peripheral Nerve Regeneration. J. Wuhan Univ. Technol.-Mat. Sci. Edit. 36, 600–606 (2021). https://doi.org/10.1007/s11595-021-2450-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11595-021-2450-6