Abstract
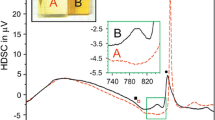
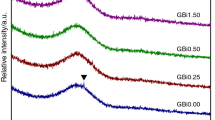
The crystallization behavior of Li2O-Al2O3-SiO2 glasses with various concentration of Li2O were investigated by DSC, XRD, and FE-SEM. The h/l-quartz s.s. is the main crystalline phase for all of the glass with the heat treatment of 720 °C/3 h+800°C/1.5 h while the crystallinity and crystal size increase with the increase of Li2O contents. Due to the negative thermal expansion coefficient of h/l-quartz s.s., the thermal expansion coefficient of glass-ceramics decreases with the increasing of Li2O contents. When the Li2O content is 9mol%, the near-zero CTE20–700 °C= −0.09 × 10−6 °C−1 is obtained upon the heat treatment of 720 °C/3 h+800 °C/1.5 h. The crystallization activation energy of the glass with 9mol% Li2O is about 351 kJ/mol, and the crystallization mechanism is volume crystallization.
Similar content being viewed by others
References
Soares VO, Peitl O, Zanotto ED. New Sintered Li2O-Al2O3-SiO2 Ultra-low Expansion Glass-ceramic[J]. Journal of the American Ceramic Society, 2013, 96(4): 1 143–1 149
Venkateswaran C, Sharma SC, Chauhan VS. Near-zero Thermal Expansion Transparent Lithium Alumino Silicate Glass-ceramic by Microwave Hybrid Heat-treatment[J]. Journal of the American Ceramic Society, 2018, 101(1): 140–150
Karmakar B, Kundu P, Jana S, et al. Crystallization Kinetics and Mechanism of Low-expansion Lithium Aluminosilicate Glass-ceramics by Dilatometry[J]. Journal of the American Ceramic Society, 2010, 85(10): 2 572–2 574
Feng D, Zhu Y, Li F, et al. Influence Investigation of CaF2 on the LAS Based Glass-ceramics and the Glass-ceramic/Diamond Composites[J]. Journal of the European Ceramic Society, 2016, 36(10): 2 579–2 585
Roy R, Roy DM, Osborn EF. Compositional and Stability Relationships among the Lithium Aluminosilicates: Eucryptite, Spodumene, and Petalite[J]. Journal of the American Ceramic Society, 1950, 33(5): 152–159
Li B, Duan D, Long Q. Effect of Cr2O3 Addition on Crystallization, Microstructure and Properties of Li2O-Al2O3-SiO2 Glass-ceramics[J]. Journal of Alloys and Compounds, 2017, (693): 9–15
Kleebusch E, Patzig C, Höche T, et al. Phase Formation during Crystallization of a Li2O-Al2O3-SiO2, Glass with ZrO2, as Nucleating Agentan X-ray Diffraction and STEM-study[J]. Ceramics International, 2017 (43): 9 769–9 777
Wange P, Höche T, Rüssel C, et al. Microstructure-property Relationship in High-strength MgO-Al2O3-SiO2-TiO2 Glass-ceramics[J]. Journal of Non-crystalline Solids, 2002 (298): 137–145
Dittmer M, Müller M, Rüssel C. Self-organized Nanocrystallinity in MgO-Al2O3-SiO2 Glasses with ZrO2 as Nucleating Agent[J]. Materials Chemistry Physics, 2010 (124): 1 083–1 088
Hunger A, Carl G, Rüssel C. Formation of Nanocrystalline Quartz Crystals from ZnO/MgO/Al2O3/TiO2/ZrO2/SiO2 Glasses[J]. Solid State Sci., 2010 (12): 1 570–1 574
Dittmer M, Müller M, Rüssel C. Self-organized Nanocrystallinity in MgO-Al2O3-SiO2 Glasses with ZrO2 as Nucleating Agent[J]. Materials Chemistry and Physics, 2010 (124): 1 083–1 088
Gaddam A, Fernandes HR, Tulyaganov DU, et al. The Roles of P2O5 and SiO2/Li2O Ratio on the Network Structure and Crystallization Kinetics of Non-stoichiometric Lithium Disilicate based Glasses[J]. Journal of Non-Crystalline Solids, 2018, (481): 512–521
Hu AM, Liang KM, Zhou F, et al. Phase Transformations of Li2O-Al2O3-SiO2 Glasses with CeO2 Addition[J]. Ceramics International, 2005, 31(1): 11–14
Hu AM, Liang KM, Li M, et al. Effect of Nucleation Temperatures and Time on Crystallization Behavior and Properties of Li2O-Al2O3-SiO2 Glasses[J]. Materials Chemistry and Physics, 2006, 98(2–3): 430–433
Author information
Authors and Affiliations
Corresponding author
Additional information
Funded by the Opening Project of State Key Laboratory of Special Glass and the National Natural Science Foundation of China (No. 52072278)
Rights and permissions
About this article
Cite this article
Han, J., Hong, W., Jiang, H. et al. Crystallization Behavior and Kinetics of Lithium Aluminosilicate Glasses with Various Li2O Contents. J. Wuhan Univ. Technol.-Mat. Sci. Edit. 36, 243–247 (2021). https://doi.org/10.1007/s11595-020-2401-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11595-020-2401-2