Abstract
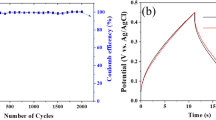
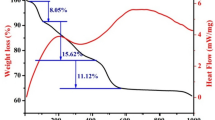
LiFePO4/C was prepared by the carbothermal reduction method with FePO4/polymer prepared by in situ polymerization-precipitation method as raw material. The effects of carbonization temperature of carbon source and its graphitization degree on the electrochemical properties of LiFePO4/C were investigated. Particle size, morphology, structure, and electrochemical properties of the prepared material were characterized. The carbonization temperature of the coated carbon source should be higher than the lithium reaction temperature and the carbonization temperature of the reduction carbon source to ensure that the carbonization product of the coated carbon source was not consumed by the lithium reaction. The addition of monomer should be beneficial to control the particle size and morphology of the precursor, as well as the thickness and uniformity of coated carbon layer on the LiFePO4/C. The initial discharge specific capacity was 163.7 mAh g−1 at 0.1 C, and the specific capacity retention rate was 93.7% after 50 cycles for the LiFePO4/C prepared.












Similar content being viewed by others
References
Le Poul N, Baudrin E, Morcrette M, Gwizdala S, Masquelier C, Tarascon JM (2003) Development of potentiometric ion sensors based on insertion materials as sensitive element. Solid State Ionics 159:149–158
Liang Y, Wen K, Mao Y, Liu Z, Zhu G, Yang F, He W (2015) Shape and size control of LiFePO4 for high-performance lithium-ion batteries. ChemElectroChem 2:1227–1237
Wang Y, Meng Y, Guo Y, Xiao D (2021) LiFePO4-covered silicon composite cathode with additional Li storage for lithium-ion batteries. Ionics 27:4983–4993
Okada S, Sawa S, Egashira M, Yamaki J, Tabuchi M, Kageyama H, Konishi T, Yoshino A (2001) Cathode properties of phospho-olivine LiMPO4 for lithium secondary batteries. J Power Sources 97–98:430–432
Ceder G, Van der Ven A, Aydinol MK (1998) Lithium-intercalation oxides for rechargeable batteries. Jom-Journal of the Minerals Metals & Materials Society 50:35–40
Massarotti V, Capsoni D, Bini M, Chiodelli G, Azzoni CB, Mozzati MC, Paleari A (1997) Electric and magnetic properties of LiMn2O4- and Li2MnO3-type oxides. J Solid State Chem 131:94–100
Fang H, Pan Z, Li L, Yang Y, Yan G, Li G, Wei S (2008) The possibility of manganese disorder in LiMnPO4 and its effect on the electrochemical activity. Electrochem Commun 10:1071–1073
Chung S-Y (2012) Comment on “Positive electrode materials for Li-ion and Li-batteries.” Chem Mater 24:2240–2243
Islam MS, Driscoll DJ, Fisher CAJ, Slater PR (2005) Atomic-scale investigation of defects, dopants, and lithium transport in the LiFePO4 olivine-type battery material. Chem Mater 17:5085–5092
Zhang H, Li J, Luo L, Zhao J, He J, Zhao X, Liu H, Qin Y, Wang F, Song J (2021) Hierarchically porous MXene decorated carbon coated LiFePO4 as cathode material for high-performance lithium-ion batteries. Journal of Alloys and Compounds 876:160210
Srinivasan V, Newman J (2006) Existence of path-dependence in the LiFePO4 electrode. Electrochemical and Solid State Letters 9:A110–A114
Park SB, Park CK, Hwang JT, Cho WI, Jang H (2011) Anisotropic lithium ion migration in LiFePO4. Met Mater Int 17:1017–1020
Wang L, Guo Q, Wang J, Li H, Wang G, Yang J, Song Y, Qin Y, Liu L (2014) Improved cycling performance of a silicon anode for lithium ion batteries using carbon nanocoils. RSC Adv 4:40812–40815
Tang M, Huang H-Y, Meethong N, Kao Y-H, Carter WC, Chiang Y-M (2008) Modeling particle size effects on phase stability and transition pathways in nanosized olivine cathode particles. J MRS Online Proceedings Library 1100:JJ03-04
Zhu C, Mu X, Popovic J, Weichert K, van Aken PA, Yu Y, Maier J (2014) Lithium potential variations for metastable materials: case study of nanocrystalline and amorphous LiFePO4. Nano Lett 14:5342–5349
Du W, Gupta A, Zhang X, Sastry AM, Shyy W (2010) Effect of cycling rate, particle size and transport properties on lithium-ion cathode performance. Int J Heat Mass Transf 53:3552–3561
Nan C, Lu J, Li L, Li L, Peng Q, Li Y (2013) Size and shape control of LiFePO4 nanocrystals for better lithium ion battery cathode materials. Nano Res 6:469–477
Ke X, Xiao R-G, Liao X, Ma Z-M, Wang S-D, Xu D (2018) LiFePO4/C cathode material prepared with sphere mesoporous-FePO4 as precursors for lithium-ion batteries. J Electroanal Chem 820:18–23
Armand M, Gauthier M, Magnan JF, Ravet NJC (2009) Method for synthesis of carbon-coated redox materials with controlled size. US patent, US 7601318. DOI:CN1478310 A
Iarchuk AR, Nikitina VA, Karpushkin EA, Sergeyev VG, Antipov EV, Stevenson KJ, Abakumov AM (2019) Influence of carbon coating on intercalation kinetics and transport properties of LiFePO4. ChemElectroChem 6:5090–5100
Zhang W-J (2011) Structure and performance of LiFePO4 cathode materials: a review. J Power Sources 196:2962–2970
Wang J, Sun X (2012) Understanding and recent development of carbon coating on LiFePO4 cathode materials for lithium-ion batteries. Energy Environ Sci 5:5163–5185
Luo G, Liu W, Yu X (2013) Effect of pyrrole additive on structure and properties of LiFePO4/C cathode materials prepared by in situ polymerization restriction method. Journal of the Chinese Silicate Society 41:19–23
Shi M, Li R, Liu Y (2021) In situ preparation of LiFePO4/C with unique copolymer carbon resource for superior performance lithium-ion batteries. Journal of Alloys and Compounds 854:157162
Chen W-M, Qie L, Yuan L-X, Xia S-A, Hu X-L, Zhang W-X, Huang Y-H (2011) Insight into the improvement of rate capability and cyclability in LiFePO4/polyaniline composite cathode. Electrochim Acta 56:2689–2695
Li Y, Wang L, Zhang KY, Liang F, Yao YC, Kong LX (2022) High performance of LiFePO4 with nitrogen and phosphorus dual-doped carbon layer for lithium-ion batteries. Journal of Alloys and Compounds 890:161617
Zaghib K, Julien CM (2005) Structure and electrochemistry of FePO4·2H2O hydrate. J Power Sources 142:279–284
Wang Y-G, Li H-Q, Xia Y-Y (2006) Ordered whiskerlike polyaniline grown on the surface of mesoporous carbon and its electrochemical capacitance performance. Adv Mater 18:2619–2623
Rozlivkova Z, Trchova M, Exnerova M, Stejskal J (2011) The carbonization of granular polyaniline to produce nitrogen-containing carbon. Synth Met 161:1122–1129
Zhang N, Fu T, Yang F, Kan H, Wang X, Long H (2013) The effect of sucrose on adsorption properties of hexagonal boron nitride powders. Proc. 8th China International Conference on High-Performance Ceramics (CICC-8), Chongqing, Peoples R China, 602–603:134–137. https://doi.org/10.4028/www.scientific.net/KEM.602-603.134
Song YN, Yang SF, Zavalij PY, Whittingham MS (2002) Temperature-dependent properties of FePO4 cathode materials. Mater Res Bull 37:1249–1257
Funding
This work was supported by the Natural Science Foundation of Inner Mongolia (grant no. 2021MS05031).
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Yan, X., Yang, Y., Li, C. et al. The synthesis of LiFePO4/C with polyaniline as coated carbon source and sucrose as reducing carbon source. Ionics 28, 1559–1571 (2022). https://doi.org/10.1007/s11581-021-04430-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11581-021-04430-1