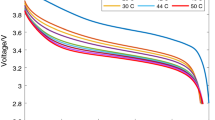
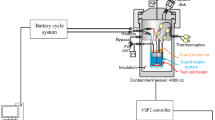
Abstract
Lithium-ion batteries have been widely used in the power-driven system and energy storage system, while overcharge safety for high-capacity and high-power lithium-ion batteries has been constantly concerned all over the world due to the thermal runaway problems by overcharge occurred in recent years. Therefore, it is very important to study the thermal runaway mechanism and improve the safety of the battery during overcharge. In this work, depending on the external appearance, voltage, and temperature changes, the whole overcharge to TR process was divided into 5 stages. By analyzing the side reactions in five stages, the mechanism of heat production during overcharge is summarized, and the order of heat generation is QCa+An > Qanode > Qelectrolyte > Qcathode > QSEI > QISC. Key factors for battery overcharge safety, such as cathode materials, electrolyte safety, and charging current are concluded in this review. Compared to external protection devices (such as BMS, OSD, CID), the internal protection of overcharge additives are more effective. A complex polymer with aromatic functional groups, epoxy or propionate, will become a hot spot in the research of overcharge additives for lithium-ion batteries. This review is expected to offer effective overcharge safety strategies and promote the development of lithium-ion battery with high-energy density.









Similar content being viewed by others
Abbreviations
- VO :
-
The initial value of voltage (V)
- Vip :
-
Voltage value of inflection point of voltage profile (V)
- Vp :
-
Voltage value of voltage plateau (V)
- Vcr :
-
The crest voltage of voltage curve (V)
- TO :
-
Ambient temperature (℃)
- TA :
-
The temperature of the Vip
- TB :
-
The temperature of the Vp
- TONSET :
-
The onset temperature of TR and the start temperature of stage V
- Tmax:
-
The maximum surface temperature of the cell during thermal runaway
- Q:
-
The total heat
- Qside :
-
The side reaction energy
- Qchem :
-
The heat from the released chemical energy
- Qele :
-
The released electrical energy
- Qp :
-
The heat dissipation
- Qrev :
-
The reversible entropy heat
- Qohm:
-
Joule heat
- LTO:
-
Li4Ti5O12 cathode materials
- LCO:
-
LiCoO2 cathode materials
- NCA:
-
LiNi0.8Co0.15Al0.05O2 cathode materials
- LFP:
-
LiFePO4 cathode materials
- NCM111:
-
Li(NiCoMn)1/3O2 cathode materials
- MCMB:
-
Artificial graphite
- MAG10:
-
Artificial graphite
References
Huang WS, Feng XN, Han XB, Zhang WF, Jiang FC (2021) Questions and answers relating to lithium-ion battery safety issues. Cell Rep Phys Sci 2:100285. https://doi.org/10.1016/j.xcrp.2020.100285
Zaghib K, Dube J, Dallaire A, Galoustov K, Guerfifi A, Ramanathan M, Benmayza A, Prakash J, Mauger A, Julien CM (2012) Enhanced thermal safety and high power performance of carbon-coated LiFePO4 olivine cathode for lithium-ion batteries. J Power Sources 219:36–44. https://doi.org/10.1016/j.jpowsour.2012.05.018
Zhu XQ, Wang H, Wang X, Gao YF, Allu S, Cakmak E, Wang ZP (2020) Internal short circuit and failure mechanisms of lithium-ion pouch cells under mechanical indentation abuse conditions: an experimental study. J Power Sources 455:227939. https://doi.org/10.1016/j.jpowsour.2020.227939
ZhuXQ WH, Allu S, Gao YF, Cakmak E, Hopkins EJ, Veith GM, Wang ZP (2020) Investigation on capacity loss mechanisms of lithium-ion pouch cells under mechanical indentation conditions. J Power Sources 465:228314. https://doi.org/10.1016/j.jpowsour.2020.228314
Wen JW, Yan Y, Chen CH (2012) A review on lithium-ion batteries safety issues: existing problems and possible solutions. Mater Express 2:197–212. https://doi.org/10.1166/mex.2012.1075
Lyu P, Liu XJ, Qu J, Zhao T, Huo YT, Qu ZG, Rao ZH (2020) Recent advances of thermal safety of lithium ion battery for energy storage. Energy Storage Materials 31:195–220. https://doi.org/10.1016/j.ensm.2020.06.042
Zeng YQ, Wu K, Wang DY, Wang ZX, Chen LQ (2006) Overcharge investigation of lithium-ion polymer batteries. J Power Sources 160:1302–1307. https://doi.org/10.1016/j.jpowsour.2006.02.009
Chen SC, Wang ZR, Yan W (2020) Identification and characteristic analysis of powder ejected from a lithium ion battery during thermal runaway at elevated temperatures. J Hazard Mater 400:123169. https://doi.org/10.1016/j.jhazmat.2020.123169
Beletskii EV, Fedorova AA, Lukyanov DA, Kalnin AY, Ershov VA, Danilov SE, Spiridonova DV, Alekseeva EV, Levin OV (2021) Switchable resistance conducting-polymer layer for Li-ion battery overcharge protection. J Power Sources 490:229548. https://doi.org/10.1016/j.jpowsour.2021.229548
Doh CH, Kim DH, Kim HS, Shin HM, Jeong YD, Moon SI, Jin BS, Eom SW, Kim HS, Kim KW, Oh DH, Veluchamy A (2008) Thermal and electrochemical behaviour of C/LixCoO2 cell during safety test. J Power Sources 175:881–885. https://doi.org/10.1016/j.jpowsour.2007.09.102
Lin P, Jin P, Hong J, Wang Z (2020) Battery voltage and state of power prediction based on an improved novel polarization voltage model. Energy Rep 6:2299–2308. https://doi.org/10.1016/j.egyr.2020.08.014
Rowden B, Nuria GA (2020) A review of gas evolution in lithiumion batteries. Energy Rep 6:10–18. https://doi.org/10.1016/j.egyr.2020.02.022
Wang SJ, Rafiz K, Liu JL, Jin Y, JerryY LS (2020) Effects of lithium dendrites on thermal runaway and gassing of LiFePO4 batteries. Sustain Energy Fuels 4:2342–2351. https://doi.org/10.1039/d0se00027b
Yuan L, Dubaniewicz T, Zlochower Z, Thomas R, Naseem R (2020) Experimental study on thermal runaway and vented gases of lithium-ion cells. Process Saf Environ Prot 144:186–192. https://doi.org/10.1016/j.psep.2020.07.028
Liu H, Cheng XB, ChongY YH, Huang JQ, Zhang Q (2021) Advanced electrode processing of lithium ion batteries: a review of powder technology in battery fabrication. Particuology 57:56–71. https://doi.org/10.1016/j.partic.2020.12.003
Ouyang MG, Ren DS, Lu L, Li JQ, Feng XN, Han XB, Liu GM (2015) Overcharge-induced capacity fading analysis for large format lithium-ion batteries with LiyNi1/3Co1/3Mn1/3O2 - LiyMn2O4 composite cathode. J Power Sources 279:626–635
Li C, Zhang HP, Fu LJ, Liu H, Wu YP, Rahm E, Holze R, Wu HQ (2006) Cathode materials modified by surface coating for lithium ion batteries. Electrochim Acta 51:3872–3883. https://doi.org/10.1016/j.electacta.2005.11.015
Zhu XQ, Wang ZB, Wang YT, Wang H, Wang C, Tong L, Yi M (2019) Overcharge investigation of large format lithium-ion pouch cells with Li(Ni0.6Co0.2Mn0.2)O2 cathode for electric vehicles: thermal runaway features and safety management method. Energy 169:868–880. https://doi.org/10.1016/j.energy.2018.12.041
Mao N, Wang ZR, Chung YH, Shu CM (2019) Overcharge cycling effect on the thermal behavior, structure, and material of lithium-ion batteries. Appl Therm Eng 163:114147. https://doi.org/10.1016/j.applthermaleng.2019.114147
Wang Z, Yuan J, Zhu X, Wang H, Huang L, Wang Y (2021) Overcharge -to-thermal-runaway behavior and safety assessment of commercial lithium-ion cells with different cathode materials: a comparison study. J Energy Chem 55:484–498. https://doi.org/10.1016/j.jechem.2020.07.028
Huang L, Zhang Z, Wang Z, Zhang L, Zhu X, David DD (2019) Thermal runaway behavior during overcharge for large-format lithium-ion batteries with different packaging patterns. J Energy Storage 25:100811. https://doi.org/10.1016/j.est.2019.100811
Zhitao E, Guo H, Yan G, Wang J, Feng R, Wang Z, Li X (2021) Evolution of the morphology structural and thermal stability of LiCoO2 during overcharge. J Energy Chem 55:524–532. https://doi.org/10.1016/j.jechem.2020.06.071
Choudhari VG, Dhoble DAS, Sathe TM (2020) A review on effect of heat generation and various thermal management systems for lithium ion battery used for electric vehicle. J Energy Storage 32:101729. https://doi.org/10.1016/j.est.2020.101729
Ren D, Feng X, Lu L, Ouyang M, Zheng S, Li F, He X (2017) An electrochemical-thermal coupled overcharge-to-thermal-runaway model for lithium ion battery. J Power Sources 364:328–340. https://doi.org/10.1016/j.jpowsour.2017.08.035
Ren D, Feng X, Lu L, Li J, Ouyang M (2019) Comparison of the overcharge behaviors of lithium-ion batteries under different test conditions. Energy Procedia 158: 4921–4926 http://creativecommons.org/licenses/by-nc-nd/4.0.
Belov D, Yang M (2008) Investigation of the kinetic mechanism in overcharge process for Li-ion battery. Solid State Ionics 179:1816–1821. https://doi.org/10.1016/j.ssi.2008.04.031
Feng XN, Ouyang MG, Liu X et al (2018) Thermal runaway mechanism of lithium ion battery for electric vehicles. Energy Storage Mater 10:246–267. https://doi.org/10.1016/j.ensm.2017.05.013
Burns JC, Stevens DA, Dahn JR (2015) In-situ detection of lithium plating using high precision coulometry. J Electrochem Soc 162:A959–A964 https://iopscience.iop.org/article/https://doi.org/10.1149/2.0621506jes
Doyle M, Fuller TF, Newman J (2020) Modeling of galvanostatic charge and discharge of the lithium polymer insertion cell. J Electrochem Soc 140(1993):1526–1533. https://doi.org/10.1149/1.2221597
Doyle M, Newman J, Gozdz AS, Schmutz CN, Tarascon JM (1996) Comparison of modeling predictions with experimental data from plastic lithium ion cells. J Electrochem Soc 143:1890–1903. https://doi.org/10.1149/1.1836921
Mei WX, Zhang L, Sun JH, Wang QS (2020) Experimental and numerical methods to investigate the overcharge caused lithium plating for lithium ion battery. Energy Storage Mater 32:91–104. https://doi.org/10.1016/j.ensm.2020.06.021
Fleischhammer M, Waldmann T, Bisle G, Hogg B (2015) Interaction of cyclic ageing at high-rate and low temperatures and safety in lithium-ion batteries. J Power Sources 274:432–439. https://doi.org/10.1016/j.jpowsour.2014.08.135
Chen C, Wei Y, Zhao Z, Zou Y, Luo D (2019) Investigation of the swelling failure of lithium-ion battery packs at low temperatures using 2D /3D X-ray computed tomography. Electrochim Acta 305:65–71. https://doi.org/10.1016/j.electacta.2019.03.038
Belov D, Yang MH (2008) Failure mechanism of Li-ion battery at overcharge conditions. J Solid State Electrochem 12: 885–894 https://link.springer.com/article/https://doi.org/10.1007/s10008-007-0449-3.
Tang P, Albertus P, Newman J (2009) Two-dimensional modeling of lithium deposition during cell charging. J Electrochem Soc 156:A390–A399 https://iopscience.iop.org/article/https://doi.org/10.1149/1.3095513.
Fernandes T, Bry A, De S (2018) Identification and quantification of gases emitted during abuse tests by overcharge of a commercial Li-ion battery. J Power Sources 389:106–119. https://doi.org/10.1016/j.jpowsour.2018.03.034
Ohsaki T, Kishi T, Kuboki T, Takami N, Shimura N, Sato Y, Sekino M, Satoh A (2005) Overcharge reaction of lithium-ion batteries. Journal of Power Sources 146 : 97–100 https://doi.org/10.1016/j.jpowsour.2005.03.105.
Yang Y, Wang ZR, Guo PK (2021) Carbon oxides emissions from lithium-ion batteries under thermal runaway from measurements and predictive mode. J Energy Storage 33:101863. https://doi.org/10.1016/j.est.2020.101863
Petibon R, Rotemund LM, Dahn JR (2015) Evaluation of phenyl carbonates as electrolyte additives in lithium-ion batteries. J Power Sources 287:184–195. https://doi.org/10.1016/j.jpowsour.2015.04.012
Gachot G, Ribière P, Mathiron D, Grugeon S, Armand M, Leriche J, Pilard S, Laruelle S (2011) Gas chromatography/mass spectrometry as a suitable tool for the Li-ion battery electrolyte degradation mechanisms study. Anal Chem 83:478–485. https://doi.org/10.1021/ac101948u
Mogi R, Inaba M, Iriyama IY, Abe T, Ogumi Z (2003) Study on the decomposition mechanism of alkyl carbonate on lithium metal by pyrolysis-gas chromatography-mass spectroscopy. J Power Sources 119–121:597–603. https://doi.org/10.1016/S0378-7753(03)00302-1
Renfrew SE, McCloskey BD (2017) Residual lithium carbonate predominantly accounts for first cycle CO2 and CO outgassing of li-stoichiometric and Li-rich layered transition-metal oxides. J Am Chem Soc 139:17853–17860. https://doi.org/10.1021/jacs.7b08461
Metzger M, Marino C, Sicklinger J, Haering DA, Gasteiger HA (2015) Anodic oxidation of conductive carbon and ethylene carbonate in high-voltage Li-ion batteries quantified by on-line electrochemical mass spectrometry. J Electrochem Soc 162:A1123-1134. https://doi.org/10.1149/2.0951506jes
Yang Y, Ramaswamy SG, Jakoby WB (1998) Enzymatic hydrolysis of organic cyclic carbonates. J Biol Chem 273:7814–7817. https://doi.org/10.1074/jbc.273.14.7814
Yuan Q, Zhao F, Wang W, Zhao Y, Liang Z, Yan D (2015) Overcharge failure investigation of lithium-ion batteries. Electrochim Acta 178:682–688. https://doi.org/10.1016/j.electacta.2015.07.147
Kumai k, Miyashiro H, Kobayashi Y, Takei K, Ishikawa R (1999) Gas generation mechanism due to electrolyte decomposition in commercial lithium-ion cell. J Power Sources 81–82:715–719.PII: S0378- 7753 98 00234–1
Ye J, Chen H, Wang Q, Huang P, Sun J, Lo S (2016) Thermal behavior and failure mechanism of lithium ion cells during overcharge under adiabatic conditions. Appl Energy 182:464–474. https://doi.org/10.1016/j.apenergy.2016.08.124
Li H, Duan QL, Zhao CP, Huang ZH, Wang QS (2019) Experimental investigation on the thermal runaway and its propagation in the large format battery module with i(Ni1/3Co1/3Mn1/3)O2 as cathode. J Hazard Mater 375:241–254. https://doi.org/10.1016/j.jhazmat.2019.03.116
Feng XN, Fang MM, He X, Ouyang MG, Lu LG, Wang H, Zhang MX (2014) Thermal runaway features of large format prismatic lithium ion battery using extended volume accelerating rate calorimetry. J Power Sources 255:294–301. https://doi.org/10.1016/j.jpowsour.2014.01.005
Feng X, He X, Ouyang M, Wang L, Lu L, Ren D, Santhanagopalan S (2018) A coupled electrochemical-thermal failure model for predicting the thermal runaway behavior of lithium-ion batteries. J Electrochem Soc 165:A3748–A3765. https://doi.org/10.1149/2.0311816jes
Xu K, Zhuang GV, Allen JL, Lee U, Zhang SS, Ross PN Jr, Jow TR (2006) Syntheses and characterization of lithium alkyl mono- and dicarbonates as components of surface films in li-ion batteries. J Phys Chem B 110:7708–7719. https://doi.org/10.1021/jp0601522
Browning KL, Baggetto L, Unocic RR, Dudney NJ, Veith GM (2013) Gas evolution from cathode materials: a pathway to solvent decomposition concomitant to SEI formation. J Power Sources 239:341–346. https://doi.org/10.1016/j.jpowsour.2013.03.118
Richard MN, Dahn JR (1999) Accelerating rate calorimetry study on the thermal stability of lithium intercalated graphite in electrolyte I experimental. J Electrochem Soc 146:2068–2077. https://doi.org/10.1016/S0140-6701(00)96499-3
Liao Z, Zhang S, Li K, Zhang G, Habetler TG (2019) A survey of methods for monitoring and detecting thermal runaway of lithium-ion batteries. J Power Sources 436:226879. https://doi.org/10.1016/j.jpowsour.2019.226879
Zhu X, Wang Z, Wang C, Huang L (2018) Overcharge investigation of large format lithium-ion pouch cells with Li(Ni0.6Co0.2Mn0.2)O2 cathode for electric vehicles: degradation and failure mechanisms. J Electrochem Soc 165:A3613–A3629. https://doi.org/10.1149/2.0161816jes
Spotnitz R, Franklin J (2003) Abuse behavior of high-power, lithium-ion cells. J Power Sources 113:81–100. https://doi.org/10.1016/S0378-7753(02)00488-3
Jung R, Metzger MF, Stinner C, Gasteiger H (2017) Oxygen release and its effect on the cycling stability of LiNixMnyCozO2(NMC) cathode materials for Li-ion batteries. J Electrochem Soc 164:A1361–A1377. https://doi.org/10.1149/2.0021707jes
Aurbach D, Gofer Y, Ben-Zion M, Aped P (1992) The behaviour of lithium electrodes in propylene and ethylene carbonate: Te major factors that influence Li cycling efficiency. J Electroanal Chem 339:451–471. https://doi.org/10.1016/0022-0728(92)80467-I
Pasquier AD, Disma F, Bowmer T (1998) Differential scanning calorimetry study of the reactivity of carbon anodes in plastic li-ion batteries. J Electrochem Soc 145:472–477. https://doi.org/10.1016/S0140-6701(98)93828-0
Stenzel YP, B€orner M, Preibisch Y, Winter M, Nowak S, (2019) Thermal profiling o lithium ion battery electrodes at different states of charge and aging conditions. J Power Sources 433:226709. https://doi.org/10.1016/j.jpowsour.2019.226709
Liu X, Ren D, Hsu H, He X, Amine K, Ouyang M (2018) Thermal runaway of lithium-ion batteries without internal short circuit. Joule 2:2047–2064. https://doi.org/10.1016/j.joule.2018.06.015
Wang HY, Tang AD, Luang HK (2011) Oxygen evolution in overcharged LixNi1/3Co1/3Mn1/3O2 electrode and its thermal analysis kinetics. Chin J Chem 29:1583–1588. https://doi.org/10.1002/cjoc.201180284
Nitta N, Wu F, Lee JT, Yushin G (2015) Li-ion battery materials: present and future. Mater Today 18 :252–264 ://doi.org/https://doi.org/10.1002/cjoc.201180284.
Wang Q, Jiang L, Yu Y, Sun J (2019) Progress of enhancing the safety of lithium ion battery from the electrolyte aspect. Nano Energy 55:93–114. https://doi.org/10.1016/j.nanoen.2018.10.035
Wang QS, Ping P, Zhao XJ, Chu GQ, Sun JH, Chen CH (2012) Thermal runaway caused fire and explosion of lithium ion battery. J Power Sources 208:210–224. https://doi.org/10.1016/j.jpowsour.2012.02.038
Pasquier AD, Wang Y, Weyhe R, Friedrich B (2018) Gas generation measurement and evaluation during mechanical processing and thermal treatment of spent Li-ion batteries. Waste Manage 84:102–111. https://doi.org/10.1016/j.wasman.2018.11.029
Liu J, Duan Q, Feng L, Ma M, Sun J, Wang Q (2020) Capacity fading and thermal stability of LiNixCoyMnzO2 /graphite battery after overcharging.J Energy Storage 29 :101397 https://doi.org/10.1016/j.est.2020.101397.
Röder P, Stiaszny B, Ziegler J, Lagaly NP, Wiemhöfer HD (2014) The impact of calendar aging on the thermal stability of a LiMn2O4-Li(Ni1/3Mn1/3Co1/3)O2/graphite lithium-ion cell. J Power Sources 268:315–325. https://doi.org/10.1016/j.jpowsour.2014.06.040
Sloop SE, Kerr JB, Kinoshita K (2003) The role of Li-ion battery electrolyte reactivity in performance decline and self-discharge. J Power Sources 119 :330–337, https://doi.org/10.1016/S0378-7753(03)00149-6.
Kawamura T, Kimura A, Egashira M, Okada S, Yamaki JI (2002) Thermal stability of alkyl carbonate mixed- solvent electrolytes for lithium ion cells. J Power Sources 104:260–264,PII: 0378–7753(01)00960–0
Li J, Wang GX, Xu ZM (2016) Environmentally-friendly oxygen-free roasting/wet magnetic separation technology for in situ recycling cobalt, lithium carbonate and graphite from spent LiCoO2/graphite lithium batteries. J Hazard Mater 302:97–104. https://doi.org/10.1016/j.jhazmat.2015.09.050
Botte GG, White RE, Zhang Z (2001) Thermal stability of LiPF6–EC: EMC electrolyte for lithium ion batteries. J Power Sources 97:570–575. https://doi.org/10.1016/S0378-7753(01)00746-7
Wong AY, Lam F (2002) Study of selected thermal characteristics of polypropylene/polyethylene binary blends using DSC and TGA.J Polym Test 21 :691–696 https://doi.org/10.1016/S0142-9418(01)00144-1
Yamada A, Chung SC, Hinokuma K (2001) Optimized LiFePO4 for lithium battery cathodes. J Electrochem Soc 148:A224–A229 101149/1.1348257.
Wong AY, Lam, (2002) Study of selected thermal characteristics of polypropylene/polyethylene binary blends using DSC and TGA. J Polym Test 21:691–696. https://doi.org/10.1016/S0142-9418(01)00144-1
Park JH, Kim JM, Lee CK, Lee SY (2014) Mixed ion/electron-conductive protective soft nanomatter-based conformal surface modification of lithium-ion battery cathode materials. J Power Sources 263:209–216. https://doi.org/10.1016/j.jpowsour.2014.04.028
Belharouak I, Lu W, Vissers D, Amine K (2006) Safety characteristics of Li(Ni0.8Co0.15Al0.05)O2 and Li(Ni1/3Co1/3Mn1/3)O2. Electrochem Commun 8:329–335. https://doi.org/10.1016/j.elecom.2005.12.007
Belharouak I, Sun YK, Lu W, Amine K (2007) On the safety of the Li4Ti5O12/LiMn2O4 lithium-ion battery system. J Electrochem Soc 154 :A1083–A1087 106.114.76.65 on 07/04/2021.
Park YS, Bang HJ, Oh SM, Lee SYK, SM, (2009) Effect of carbon coating on thermal stability of natural graphite spheres used as anode materials in lithium-ion batteries. J Power Sources 190:553–557. https://doi.org/10.1016/j.jpowsour.2009.01.067
Tanaka H, Osawa T, Moriyoshi Y, Kurihara M, Maruyama S, Ishigaki T (2004) Improvement of the anode performance of graphite particles through surface modification in RF thermal plasma. Thin Solid Films 7:209–216. https://doi.org/10.1016/j.tsf.2003.12.024
Roth EP, Doughty DH (2004) Thermal abuse performance of high-power 18650 Li-ion cells. J Power Sources 8:308–318. https://doi.org/10.1016/j.jpowsour.2003.09.068
Zheng S, Wang L, Feng X (2018) Probing the heat sources during thermal runaway process by thermal analysis of different battery chemistries. J Power Sources 378:527–536. https://doi.org/10.1016/j.jpowsour.2017.12.050
Eom J, Kim MG, Cho J (2008) Storage characteristics of LiNi0.8Co0.1 + x Mn0.1 − x O2 (x = 0, 0.03, and 0.06) cathode materials for lithium batteries. J Electrochem Soc 155:A239–A245. https://doi.org/10.1149/1.2830946
Noh HJ, Youn S, Yoon CS, Sun YK (2013) Comparison of the structural and electrochemical properties of layered Li[NixCoyMnz]O2 (x = 1/3, 0.5, 0.6, 0.7, 0.8 and 0.85) cathode material for lithium-ion batteries. J Power Sources 233:121–130. https://doi.org/10.1016/j.jpowsour.2013.01.063
Venkatraman S, Choi J, Manthiram A (2004) Factors influencing the chemical lithium extraction rate from layered LiNi1−y−zCoyMnzO2 cathodes. Electrochem Commun 6:832–837. https://doi.org/10.1016/j.elecom.2004.06.004
Heubner C, Schneider M, Lamme C, Michaelis A (2015) Local heat generation in a single stack lithium ion battery cell. Electrochim. Acta 186 : 404–412 10.1016 /j.electacta. 2015.10.182.
Xiao M, Choe SY (2013) Theoretical and experimental analysis of heat generations of a pouch type LiMn2O4/carbon high power Li-polymer battery. J Power Sources 241:46–55. https://doi.org/10.1016/j.jpowsour.2013.04.062
Malik M, Mathew M, Dincer I, Rosen MA, Fowler M (2018) Experimental investigation and thermal modelling of a series connected LiFePO4 battery pack. Int J Therm Sci 132:466–477. https://doi.org/10.1016/j.ijthermalsci.2018.06.025
Giel H, Henriques D, Bourne G, Markus T (2018) Investigation of the generation of a commercial 2032 (LiCoO2) coin cell with a novel differential scanning battery calorimeter. J Power Sources 390:116–126. https://doi.org/10.1016/j.jpowsour.2018.04.017
Hosseinzadeh E, Genieser R, Worwood D, Barai A, Marco J, Jenningsl PA (2018) Systematic approach for electrochemical-thermal modelling of a large format lithium-ion battery for electric vehicle application. J Power Sources 382:77–94. https://doi.org/10.1016/j.jpowsour.2018.02.027
Xu X, Deng S, Wang H, et al (2017) Research progress in improving the cycling stability of high-voltage Li Ni0.5Mn1.5O4 cathode in lithium-ion battery. Nano-micro Letters 9: 22 https://doi.org/10.1007/s40820-016-0123-3
Manthiram A, Chemelewski K, Lee ES (2014) A perspective on the high-voltage Li Mn1.5Ni0.5O4 spinel cathode for lithium-ion batteries. Energy Environ Sci 7:1339–1350. https://doi.org/10.1039/C3EE42981D
Bai Y, Li L, Li Y, Chen GH, Zhao HC, Wang ZH, Wu C, Ma HY, Wang XQ, Cui HY, Zhou J (2019) Reversible and irreversible heat generation of NCA/Si–C pouch cell during electrochemical energy-storage process. J Energy Chem 29:95–102. https://doi.org/10.1016/j.jechem.2018.02.016
Liang C, Jiang L, Wang Q, Sun J (2020) Dynamic heat generation of LiNi0.5Co0.2Mn0.3O2 half cell under cycling based on an in situ microcalorimetry. Fire Technol 2387–2440 https://doi.org/10.1007/s10694-020-00956-4.
Qi C, Zhu Y, Gao F (2018) Mathematical model for thermal behavior of lithium ion battery pack under overcharge. Int J Heat Mass Transfer 124:552–563. https://doi.org/10.1016/j.ijheatmasstransfer.2018.03.100
Hyundai Motor Company, overcharge safety device. USPTO 20180159185,2018
Li W, Crompton KR, Hacker C (2020) Comparison of current interrupt device and vent design for 18650 format lithium-ion battery caps. J Energy Storage 32:101890. https://doi.org/10.1016/j.est.2020.101890
Dai HF, Zhang XL, Wei XZ, Sun ZC, Wang ZC, Hu F (2013) Cell-BMS validation with a hardware-in-the-loop simulation of lithium-ion battery cells for electric vehicles. Int J Electr Power Energy Syst 52:174–184. https://doi.org/10.1016/j.ijepes.2013.03.037
Re T (2020) Electrolyte additives for improved lithium-ion battery performance and overcharge protection. Curr Opin Electrochem 21:22–30. https://doi.org/10.1016/j.coelec.2020.01.001
Wang ZY, Jiang LH, Liang C, Zhao CP, Wei ZS, Wang QS (2020) Effects of 3-fluoroanisol as an electrolyte additive on enhancing the overcharge endurance and thermal stability of lithium-ion batteries. J Electrochem Soc 167: 30517 https://doi.org/10.1149/1945-7111/ abb8b2
Golovin MN, Wilkinson DP, Dudley JT, Holonko D, Hoo S (1992) Applications of metallocenes in rechargeable lithium batteries for overcharge protection. J Electrochem Soc 139:5–10. https://doi.org/10.1023/A:1024039302020
Ding Y, Zhao Y, Li Y, Goodenough JB, Yu G (2017) A high-performance all-metallocene-based, non-aqueous redox flow battery. Energy Environ Sci 10:491–497. https://doi.org/10.1039/c6ee02057g
Forgie JC, Khakani SE, MacNeil DD, Rochefort D (2013) Electrochemical characterisation of a lithium-ion battery electrolyte based on mixtures of carbonates with a ferrocene-functionalised imidazolium electroactive ionic liquid. Phys Chem Chem Phys 15(2013):7713–7721. https://doi.org/10.1039/C3CP50560J
Abraham K M, Pasquariello D M. Overcharge protection of secondary non-aqueous batteries[P].CA1306003,1992–08–04
Abraham KM, Pasquariello DM, Willstaedt EB (1990) n-Butylferrocene for overcharge protection of secondary lithium batteries. J Electrochem Soc 137:1856–1861
Ates MN, Allen CJ, Mukerjee S, Abraham K (2012) Electronic effects of substituents on redox shuttles for overcharge protection of Li-ion batteries. J Electrochem Soc 159:A1057–A1064. https://doi.org/10.1149/2.064207jes
Cha C, Ai X, Yang H (1995) Polypyridine complexes of iron used as redox shuttles for overcharge protection of secondary lithium batteries. J Power Sources 54:255–258. https://doi.org/10.1016/0378-7753(94)02079-I
Abraham K M, Rohan J F, Foo C C, et al. Chemical overcharge protection of lithium and lithium-ion secondary batteries[P].EP 0825663, 1998–02–25.
Odom SA, Ergun S, Poudel PP, Parkin SR (2014), A fast, inexpensive method for predicting overcharge performance in lithium-ion batteries Energy Environ Sci 7:760–767 https://doi.org/10.1039/C3EE42305K
Kerr J B, Tia M. Electrochemical storage cell containing a substituted anisole or di-anisole redox shuttle additive for overcharge protection and suitable for use in liquid organic and solid polymer electrolytes[P].US 6045952, 2000–04–04
Chen Z, Qin Y, Amine K (2009) Redox shuttles for safer lithium-ion batteries. Electrochim Acta 54:5605–5613. https://doi.org/10.1016/j.electacta.2009.05.017
Behl WK, Chin DT (1998) Electrochemical overcharge protection of rechargeable lithium batteries: II Effect of lithium iodide-iodine additives on the behavior of lithium electrode in LiAsF6-tetrahydrofuran solutions. J Electrochem Soc 135:21–25. https://doi.org/10.1149/1.2095558
Behl WK, Chin DT (1988) Electrochemical overcharge protection of rechargeable lithium batteries: I Kinetics of iodide/tri-iodide/iodine redox reactions on platinum in LiAsF6-tetrahydrofuran solutions. J Electrochem Soc 135:16–21. https://doi.org/10.1149/1.2095545
Allen J (2020) Review of polymers in the prevention of thermal runaway in lithium-ion batteries. Energy Rep 6:217–224. https://doi.org/10.1016/j.egyr.2020.03.027
Buhrmester C, Chen J, Moshurchak L, Jiang J, Wang RL, Dahn J (2005) Studies of aromatic redox shuttle additives for LiFePO4-based Li-ion cells. J Electrochem Soc 152:A2390–A2399. https://doi.org/10.1149/1.2098265
Huang J, Shkrob IA, Wang P, Cheng L, Pan B, He M, Liao C, Zhang Z, Curtiss ZL (2015) 1,4-Bis(trimethylsilyl)- 2,5-dimethoxybenzene: a novel redox shuttle additive for overcharge protection in lithium-ion batteries that doubles as a mechanistic chemical probe. J Mater Chem A3:7332–7337. https://doi.org/10.1039/C5TA00899A
Zhang Z, Zhang L, Schlueter JA, Redfern PC, Curtiss L, Amine K (2010) Understanding the redox shuttle stability of 3,5-di-tert- butyl-1,2-dimethoxybenzene for overcharge protection of lithium-ion batteries. J Power Sources 195: 4957–4962 https://doi.org/10.1016/j.jpowsour.2010.02.0
Zhang L, Zhang Z, Redfern PC, Curtiss LA, Amine K (2012) Molecular engineering towards safer lithium-ion batteries: a highly stable and compatible redox shuttle for overcharge protection. Energy Environ Sci 5:8204–8207. https://doi.org/10.1039/C2EE21977H
Leonet O, Colmenares LC, Kvasha A, Oyarbide M, Mainar AR, Glossmann T, Blázquez JA, Zhang Z (2018) Improving the safety of lithium-ion battery via a redox shuttle additive 2,5-Di-tert-butyl-1,4-bis(2-methoxyethoxy)benzene (DBBB). ACS Appl Mater Interfaces 10:9216–9219. https://doi.org/10.1021/acsami.8b01298
Matadi BP, Geniès S, Delaille A, Waldmann T, Kasper M, Wohlfahrt MM, Aguesse F, Bekaert E, Jiménez-Gordon I, Daniel L (2017) Effects of biphenyl polymerization on lithium deposition in commercial graphite/NMC lithium-ion pouch-cells during calendar aging at high temperature. J Electrochem Soc 164:A1089–A1097. https://doi.org/10.1149/2.0631706jes
Huang T, Zheng X, Wang W, Pan Y, Fang G, Wu M (2017) 2-Chloro-4-methoxy)-phenoxy pentafluorocyclotriphosphazene as a safety additive for lithium-ion batteries. Mater Chem Phys 196:310–314. https://doi.org/10.1016/j.matchemphys.2017.05.005
Vogl U, Schmitz A, Stock C, Badillo JP, Winter GHJ, M, (2014) Investigation of N-ethyl-2-pyrrolidone (NEP) as electrolyte additive in regard to overcharge protecting characteristics. J Electrochem Soc 161:A1407–A1414. https://doi.org/10.1149/2.1021409jes
Xiao L, Ai X, Cao Y, Yang H (2004) Electrochemical behavior of biphenyl as polymerizable additive for overcharge protection of lithium ion batteries. Electrochim Acta 49:4189–4196. https://doi.org/10.1016/j.electacta.2004.04.013
Zhang Q, Qiu C, Fu Y, Ma X (2009) Xylene as a new polymerizable additive for overcharge protection of lithium ion batteries. Chin J Chem 27:1459–1463. https://doi.org/10.1002/cjoc.200990245
Lee H, Lee JH, Ahn S, Kim HJ, Cho JJ (2006) Co-use of cyclohexyl benzene and biphenyl for overcharge protection of lithium-ion batteries. Electrochem Solid-State Lett 9:A307–A310. https://doi.org/10.1149/1.2193072
Yang W, Yang W, Song A, Gao L, Sun G, Shao G (2017) Pyrrole as a promising electrolyte additive to trap polysulfides for lithium-sulfur batteries. J Power Sources 348:175–182. https://doi.org/10.1016/j.jpowsour.2017.03.008
Li S, Xia L, ZhangH AX, Yang H, Cao Y (2011) A poly(3-decyl thiophene)-modified separator with self-actuating overcharge protection mechanism for LiFePO4-based lithium ion battery. J Power Sources 196:7021–7024. https://doi.org/10.1016/j.jpowsour.2010.09.111
Mao H Y, Von S U. Aromatic monomer gassing agents for protecting non-aqueous lithium batteries against overcharge[P].US 5776627,1998–07–07
Abe K, Matsumori Y, Ueki A. Nonaqueous electrolytic solution and lithium secondary batteries [P].EP1361622, 2003–11–12
Roh K, Choi J, Lee J. Electrolyte composition for Lithium secondary battery having high overcharge safety[P]. WO2004040687,2004–05–13
Tobishima S, Ogino Y, Watanabe Y (2003) Influence of electrolyte additives on safety and cycle life of rechargeable lithium cells. J Appl Electrochem 33:143–150. https://doi.org/10.1023/A:1024039302020
He YB, Liu Q, Tang ZY, Chen YH, Song QS (2007) The cooperative effect of tri(-chloromethyl) phosphate and cyclohexyl benzene on lithium ion batteries. Electrochim Acta 52:3534–3540. https://doi.org/10.1016/j.electacta.2006.10.039
Chen YH, Tang ZY, Wang L (2006) Aplication of cyclohexyl benzene and triethyl amine for overcharge protection of Li- ion batteries. Chinese Journal of Power Sources30 :829–832
Mao HY, Wainwright DS. Polymerizable additives for making non-aqueous rechargeable lithium batteries safe after overcharge[P]. US 6074776,2000–06–13
Mao HY. Polymerizable aromatic additives for overcharge protection in non-aqueous rechargeable lithium batteries[P].US 5879834,1999–03–09
Ralph OG, Stephan KL, Lutz BL. Overcharge protection of nonaqueous rechargeable lithium batteries by cyano-substituted thiophenes as electrolyte additives[P].US 20040053138,2004–03–18
Hyeong-Gon NS. Polymer electrolyte composition for improving overcharge safety and lithium battery using the same[P].US20030152837,2004–08–14
Feng J, Cao Y, Ai X, Yang H (2008) Tri-(4-methoxythphenyl) phosphate: a new electrolyte additive with both fifire-retardancy and overcharge protection for Li-ion batteries. Electrochim Acta 53:8265–8268. https://doi.org/10.1016/j.electacta.2008.05.024
Feng J, Lu L (2013) A novel bifunctional additive for safer lithium ion batteries. J Power Sources 243:29–32. https://doi.org/10.1016/j.jpowsour.2013.05.170
Huang T, Zheng X, Fang G, Pan Y, Wang W, Wu M (2017) (4-Methoxy)-phenoxy pentafluorocyclotriphosphazene as a novel flame retardant and overcharge protection additive for lithium-ion batteries. RSC Adv 7:47775–47780. https://doi.org/10.1039/C7RA09416G
Feng J, Gao X, Ci L, Xiong S (2016) A novel bifunctional additive for 5 V-class high-voltage lithium ion batteries. RSC Adv 6:7224–7228. https://doi.org/10.1039/C5RA22547G
Korepp C, Kern W, Lanzer EA (2007) 4-Bromobenzyl isocyanate versus benzyl isocyanate new film-forming electrolyte additives and overcharge protection additives for lithium ion batteries. J Power Sources 174:637–642 0.1016/j.jpowsour.2007.06.142
Funding
This work was funded by Science and Technology Project of Hebei Education Department (No. ZD2020327).
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Chen, Y. Recent advances of overcharge investigation of lithium-ion batteries. Ionics 28, 495–514 (2022). https://doi.org/10.1007/s11581-021-04331-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11581-021-04331-3