Abstract
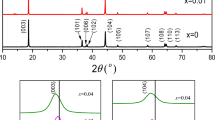
Na3V2-xBix(PO4)3/C (0 ≤ x ≤ 0.07) samples were prepared by a facile sol-gel method. Dependence of their physical and electrochemical properties on the amount of Bi3+ doping are investigated by X-ray diffraction (XRD) analysis, thermogravimetric analysis (TGA), X-ray photoelectron spectroscopy (XPS), inductively coupled plasma optical emission spectrometer (ICP-OES), impedance spectroscopy (EIS), cyclic voltammetry (CV), scanning electron microscopy (SEM), and high resolution transmission electron microscopy (HRTEM). It is found that Bi3+ is substituted on the vanadium site without damage to the structure of Na3V2(PO4)3 compound. The Na3V1.97Bi0.03(PO4)3/C cathode materials demonstrate superior rate capability and delivered specific capacity of 116.3 and 77.4 mAh∙g−1 at charge-discharge of 0.1 C and 20 C (1 C =117.6 mA∙g−1), respectively. Circulating at a high current rate of 20 C, the discharge capacity of 67.3 mAh∙g−1 can be still reached, and the capacity can remain at 86.68% over 100 cycles. Na3V2-xBix(PO4)3/C composites possess an excellent electrochemical characteristic, which mainly ascribes to its nano-sized particles, stability in structure, and enhancement in ionic and electronic conductivity arose from Bi3+ doping.









Similar content being viewed by others
References
Palomares V, Serras P, Villaluenga I, Hueso KB, Carretero-González J, Rojo T (2012) Na - ion batteries, recent advances and present challenges to become low cost energy storage systems. Energy Environ Sci 5:5884–5901
Whittingham MS (2004) Lithium batteries and cathode materials. Chem Rev 104:4271–4302
Etacheri V, Marom R, Elazari R, Salitra G, Aurbach D (2011) Challenges in the development of advanced Li - ion batteries: a review. Energy Environ Sci 4:3243–3262
Risacher F, Fritz B (2009) Origin of salts and brine evolution of Bolivian and Chilean salars. Aquat Geochem 15:23–157
Taracson JM, Armand M (2001) Issues and challenges facing lithium ion batteries. Nature 414:59–367
Seyfried WE Jr, Janecky DR, Mottl MJ (1984) Alteration of the oceanic crust: implications for geochemical cycles of lithium and boron. Geochim Cosmochim Acta 48:557–569
Dunn B, Kamath H, Tarascon JM (2011) Electrical energy storage for the grid: a battery of choices. Science 334:928–935
Cheng F, Liang J, Tao Z, Chen J (2011) Functional materials for rechargeable batteries. Adv Mater 23:1695–1715
Lee KT, Ramesh TN, Nan F, Botton G, Nazar LF (2011) Topochemical synthesis of sodium metal phosphate olivines for sodium - ion batteries. Chem Mat 23:3593–3600
Zhu Y, Xu Y, Liu Y, Luo C, Wang C (2012) Comparison of electrochemical performances of olivine nafepo4 in sodium-ion batteries and olivine LiFePO4 in lithium-ion batteries. Nanoscale 5:780–787
Oh SM, Myung ST, Hassoun J, Scrosati B, Sun YK (2012) Reversible NaFePO4 electrode for sodium secondary batteries. Electrochem Commun 22:149–152
Ivana H, Jusef YK, Scrosati B (2014) Sodium-ion battery based on an electrochemically converted NaFePO4 cathode and nanostructured tin-carbon anode. ChemPhysChem 15:2152–2155
Li G, Jiang D, Hui W, Lan X, Zhong H, Yang J (2014) Glucose-assisted synthesis of Na3V2(PO4)3/C composite as an electrode material for high-performance sodium-ion batteries. J Power Sources 265:325–334
Lim SY, Kim H, Shakoor RA, Jung Y, Choi JW (2012) Electrochemical and thermal properties of NASICON structured Na3V2(PO4)3 as a sodium rechargeable battery cathode: a combined experimental and theoretical study. J Electrochem Soc 159:A1393–A1397
Saravanan K, Mason CW, Rudola A, Wong KH, Balaya P (2013) The first report on excellent cycling stability and superior rate capability of Na3V2(PO4)3 for sodium ion batteries. Adv Energy Mater 3:444–450
Jung YH, Lim CH, Kim DK (2013) Graphene-supported Na3V2(PO4)3 as a high rate cathode material for sodium-ion batteries. J Mater Chem A 1:11350–11354
Fu L, Tang K, Song K, Van Aken PA, Yu Y, Maier J (2014) Nitrogen doped porous carbon fibres as anode materials for sodium ion batteries with excellent rate performance. Nanoscale 6:1384–1389
Li S, Dong Y, Xu L, Xu X, He L, Mai L (2014) Effect of carbon matrix dimensions on the electrochemical properties of Na3V2(PO4)3 nanograins for high-performance symmetric sodium-ion batteries. Adv Mater 26:3545–3553
Zhu C, Song K, Van Aken PA, Maier J, Yu Y (2014) Carbon-coated Na3V2(PO4)3 embedded in porous carbon matrix: an ultrafast Na-storage cathode with the potential of outperforming Li cathodes. Nano Lett 14:2175–2180
Jian Z, Zhao L, Pan H, Hu YS, Li H, Chen W, Chen L (2012) Carbon coated Na3V2(PO4)3 as novel electrode material for sodium ion batteries. Electrochem Commun 14:86–89
Liu J, Tang K, Song K, Van Aken PA, Yu Y, Maier J (2014) Electrospun Na3- V2(PO4)3/C nanofibers as stable cathode materials for sodium-ion batteries. Nanoscale 6:5081–5086
Cheng Y, Feng K, Zhou W, Zhang H, Li X, Zhang H (2015) A Bi-doped Li3V- 2(PO4)3/C cathode material with an enhanced high-rate capacity and long cycle stability for lithium ion batteries. Dalton Trans 44:17579–17586
Subburaj T, Prasanna K, Kim KJ, Ilango PR, Jo YN, Lee CW (2015) Structural and electrochemical evaluation of bismuth doped lithium titanium oxides for lithium ion batteries. J Power Sources 280:23–29
Zhang Z, Qiu J, Yu M, Jin C, Yang B, Guo G (2020) Performance of Al-doped LiNi1/3Co1/3Mn1/3O2 synthesized from spent lithium ion batteries by sol-gel method. Vacuum. https://doi.org/10.1016/j.vacuum.2019.109105
Ruan Y, Wang K, Song S, Liu J, Han X (2017) Improved structural stability and electrochemical performance of Na3V2(PO4)3 cathode material by Cr doping. Ionics 23:1097–1105
Song J, Park S, Mathew V, Gim J, Kim S, Jo J, Kim J (2016) An enhanced high-rate Na3V2(PO4)3-Ni2P nanocomposite cathode with stable lifetime for sodium-ion batteries. ACS Appl Mater Interfaces 8:35235–35242
Li H, Yu X, Bai Y, Wu F, Wu C, Liu LY, Yang XQ (2015) Effects of Mg doping on the remarkably enhanced electrochemical performance of Na3V2(PO4)3 cathode materials for sodium ion batteries. J Mater Chem A 3:9578–9586
Zhu X, Fang Y, Ai X, Yang H, Cao Y (2015) Na3V2(PO4)3/C nanocomposite synthesized via pre-reduction process as high-performance cathode material for sodium-ion batteries. J Alloy Compd 646:170–174
Fang J, Wang S, Li Z, Chen H, Xia L, Ding L, Wang H (2016) Porous Na3V- 2(PO4)3@C nanoparticles enwrapped in three-dimensional graphene for high performance sodium-ion batteries. J Mater Chem A 4:1180–1185
Masquelier C, Wurm C, Rodriguez-Carvajal J, Gaubicher J, Nazar L (2000) A powder neutron diffraction investigation of the two rhombohedral NASICON analogues: γ-Na3Fe2(PO4)3 and Li3Fe2(PO4)3. Chem Mat 12:525–532
Xu G, Sun G (2016) Mg2+-doped Na3V2(PO4)3/C decorated with graphene sheets: An ultrafast Na-storage cathode for advanced energy storage. Ceram Int 42:14774–14781
Greczynski G, Hultman L (2020) X-ray photoelectron spectroscopy: towards reliable binding energy referencing. Prog Mater Sci 107:100591
Li H, Wu C, Bai Y, Wu F, Wang M (2016) Controllable synthesis of high-rate and long cycle-life Na3V2(PO4)3 for sodium-ion batteries. J Power Sources 326:14–22
Kang J, Baek S, Mathew V, Gim J, Song J, Park H, Kim J (2012) High rate performance of a Na3V2(PO4)3/C cathode prepared by pyro-synthesis for sodium-ion batteries. J Mat Chem 22:20857–20860
Masquelier C, Croguennec L (2013) Polyanionic (phosphates, silicates, sulfates) frameworks as electrode materials for rechargeable Li (or Na) batteries. Chem Rev 113:6552–6591
Ong SP, Chevrier VL, Hautier G, Jain A, Moore C, Kim S, Ceder G (2011) Voltage, stability and diffusion barrier differences between sodium-ion and lithium-ion intercalation materials. Energy Environ Sci 4:3680–3688
Pivko M, Arcon I, Bele M, Dominko R, Gaberscek M (2012) A3V2(PO4)3 (A= Na or Li) probed by in situ X-ray absorption spectroscopy. J Power Sources 216:145–151
Yang J, Han DW, Jo MR, Song K, Kim YI, Chou SL, Kang YM (2015) Na3V2(PO4)3 particles partly embedded in carbon nanofibers with superb kinetics for ultra - high power sodium ion batteries. J Mater Chem A 3:1005–1009
Shen W, Wang C, Xu Q, Liu H, Wang Y (2015) Nitrogen - doping - induced defects of a carbon coating layer facilitate Na - storage in electrode materials. Adv Energy Mater 5:1400982
Guo JZ, Wu XL, Wan F, Wang J, Zhang XH, Wang RS (2015) A superior Na3V2(PO4)3 - based nanocomposite enhanced by both N - doped coating carbon and graphene as the cathode for sodium - ion batteries. Chem-Eur J 21:17371–17378
Aragón MJ, Lavela P, Alcántara R, Tirado JL et al (2015) Effect of aluminum doping on carbon loaded Na3V2(PO4)3 as cathode material for sodium-ion batteries. Electrochim Acta 180:824–830
Aragón MJ, Lavela P, Ortiz GF, Tirado JL (2015) Effect of iron substitution in the electrochemical performance of Na3V2(PO4)3 as cathode for Na-ion batteries. J Electrochem Soc 162(2):A3077–A3083
Aragón MJ, Lavela P, Ortiz GF, Tirado JL (2015) Benefits of chromium substitution in Na3V2(PO4)3 as a potential candidate for sodium-ion batteries. ChemElectroChem 2(7):995–1002
Liu H, Cao Q, Fu LJ, Li C, Wu YP, Wu HQ (2006) Doping effects of zinc on LiFePO4 cathode material for lithium ion batteries. Electrochem Commun 8:1553–1557
Liu H, Li C, Zhang HP, Fu LJ, Wu YP, Wu HQ (2006) Kinetic study on LiFePO4/C nanocomposites synthesized by solid state technique. J Power Sources 159:717–720
Liu W, Yi H, Zheng Q, Li X, Zhang H (2017) Y-Doped Na3V2(PO4)2F3 compounds for sodium ion battery cathodes: electrochemical performance and analysis of kinetic properties. J Mater Chem A 5:10928–10935
Funding
This work was supported by the Science and Technology Pillar Program of Sichuan Province (No. 2016GZ0393) and the Opening Foundation of Sichuan Engineering Research Center for Powder Metallurgy, Chengdu University (No. SC-FMYJ 2017-01 and No. SC-FMYJ 2017-03).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Chai, Z., Zhu, X., Chen, S. et al. Effects of bismuth doping on the physical and electrochemical properties of Na3V2-xBix (PO4)3/C (0 ≤ x ≤ 0.07) composite cathode materials. Ionics 27, 2409–2419 (2021). https://doi.org/10.1007/s11581-021-04028-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11581-021-04028-7