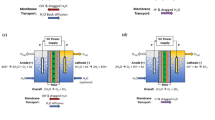
Abstract
One kind of acid-base high temperature proton exchange membranes has been prepared from amino trimethylene phosphonic acid (ATMP), epoxycyclohexyethyltrimethoxysilane (EHTMS), and 3-aminopropyltriethoxysilane (APTES) by sol-gel process. The structural characteristics of these membranes with different amount of APTES were investigated by FT-IR, XRD, and SEM. These membranes showed excellent dimensional stability in water with the contribution of flexible ionic network structure and were thermally stable up to about 200 °C. In addition, the proton conductivity of the membranes increased with increasing temperature over the range of 20 to 140 °C, up to a maximum of 2.63 × 10−2 S cm−1 at 140 °C under anhydrous condition. The high proton conductivity was attributed to the formation of hydrogen bond network through the synergistic effect of N and P. The activation energy value of membranes became lower from 0.46 to 0.30 eV because of the acid-base pairs. The variable-temperature FT-IR further proved the formation of hydrogen bond network in the membrane.










Similar content being viewed by others
References
Park CH, Lee CH, Guiver MD, Lee YM (2011) Sulfonated hydrocarbon membranes for medium-temperature and low-humidity proton exchange membrane fuel cells (PEMFCs). Prog Polym Sci 36:1443–1498
Jiang ZY, Zheng XH, Wu H, Pan FS (2008) Proton conducting membranes prepared by incorporation of organophosphorus acids into alcohol barrier polymers for direct methanol fuel cells. J Power Sources 185:85–94
Napoli L, Franco J, Fasoli H, Sanguinetti A (2014) Conductivity of Nafion 117 membrane used in polymer electrolyte fuel cells. Int J Hydrogen Energ 39:8656–8660
Schuster M, Rager T, Noda A, Maier J (2005) About the choice of the protogenic group in PEM separator materials for intermediate, low humidity operation: a critical comparison of sulfonic acid, phosphonic acid and imidazole functionalized model compounds. Fuel Cells 5:355–365
Wieser C (2004) Novel polymer electrolyte membranes for automotive applications-requirements and benefits. Fuel Cells 4:245–250
Aparicio M, Durán A (2004) Hybrid organic/inorganic sol-gel materials for proton conducting membranes. J Sol-Gel Sci Techn 31:103–107
Lin HL, Tang TH, Hu CR, Yu TL (2012) Poly(benzimidazole)/silica-ethyl-phosphoric acid hybrid membranes for proton exchange membrane fuel cells. J Power Sources 201:72–80
Souquet-Grumey J, Perrin R, Cellier J, Bigarré J, Buvat P (2014) Synthesis and fuel cell performance of phosphonated hybrid membranes for PEMFC applications. J Membrane Sci 466:200–210
Li W, Shen CH, Gao SJ, Yin SS, Li HL (2016) Preparation and characterization of phosphonic acid functionalized siloxane/polyimide composite proton exchange membranes. Solid State Ionics 287:1–7
Zhang L, He HQ, Rasheed RKA, Zhou WJ, Xue YH, Ding OL, Chan SH (2013) Fabrication of novel phosphotungstic acid functionalized mesoporous silica composite membrane by alternative gel-casting technique. J Power Sources 221:318–327
Wang C, Paddison SJ (2010) Proton transfer in functionalized phosphonic acid molecules. Phys Chem Chem Phys 12:970–981
Li W, Gao SS, Fang JH (2014) Synthesis and properties of sulfonated polyimide–polybenzimidazole copolymers as proton exchange membranes. J Mater Sci 49:2745–2753
Pan HY, Zhang YY, Pu HT, Chang ZH (2014) Organic-inorganic hybrid proton exchange membrane based on polyhedral oligomeric silsesquioxanes and sulfonated polyimides containing benzimidazole. J Power Sources 293:195–202
Chen C, Shen CH, Kong GJ, Gao SJ (2013) High temperature proton exchange membranes prepared from epoxycyclohexylethyltrimethoxysilane and amino trimethylene phosphonic acid as anhydrous proton conductors. Mater Chem Phys 140:24–30
Huang SJ, Lee HK, Lee YS, Kang WH (2005) Proton-conductive membranes doped with orthophosphoric acid based on inorganic-organic hybrid materials. J Am Ceram Soc 88:3427–3432
Chippar P, Ju H (2012) Evaluating cold-start behaviors of end and intermediate cells in a polymer electrolyte fuel cell (PEFC) stack. Solid State Ionics 225:85–91
Lakshminarayana G, Nogami M (2009) Synthesis and characterization of proton conducting inorgani-organic hybrid nanocomposite membranes based on mixed PWA-PMA-TEOS-GPTMS-H3PO4-APTES for H2/O2 fuel cells. J Phys Chem C 113:14540–14550
Zeng SJ, Hu SA, Pan SJ, Wu GL, Xu WJ (2010) Effects of acids and water addition on morphology and proton conduction in sol-gel derived acid-base polysiloxane. Solid State Ionics 181:1408–1414
Yuan SS, Tang QW, He BL (2014) Three-dimensional hydrogel frameworks for high-temperature proton exchange membrane fuel cells. J Mater Sci 49:5481–5491
Doyle M, Choi SK, Proulx G (2000) High-temperature proton conducting membranes based on perfluorinated ionomer membrane-ionic liquid composites. J Electrochem Soc 147:34–37
Tanaka Y, Nourisuye T, Hirayama S, Takemori T, Tran-Cong-Miyata Q, Nomura S (2007) DLS and AFM studies on the cluster evolution of organically modified silica gels catalyzed by a super strong acid. Macromolecules 40:3773–3778
Yue BH, Yan LM, Han SY, Xie LQ (2013) Proton transport pathways in an acid-base complex consisting of a phosphonic acid group and a 1,2,3-triazolyl group. J Phys Chem B 117:7941–7949
Acknowledgments
This work was supported by the National Natural Science Foundation of China (21276202).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The manuscript is approved by all authors for publication and no conflict of interest exits in the submission of this manuscript. The research did not involve any human or animal participant.
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Xiang, J., Shen, C., Gao, S. et al. Acid-base high temperature proton exchange membranes prepared from phosphonic acid functionalized siloxane. Ionics 23, 949–958 (2017). https://doi.org/10.1007/s11581-016-1900-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11581-016-1900-1