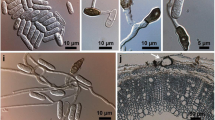
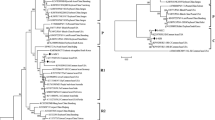
Abstract
Soybean purple seed stain (S-PSS) is a destructive, worldwide distributed fungal disease caused by several Cercospora species. This work aims to shed light on the nature of the genealogical and genetic relationships amongst S-PSS causal agents. Fungal isolates were obtained from Argentina and Brazil, which belong to the leading countries in soybean production worldwide. DNA sequences were obtained from eight loci across the collection of isolates. Relationships were evaluated through Bayesian phylogenetic inferences, and distance and character-based network analyses and discriminant analyses. The occurrence of reticulate evolutionary events was tested with recombination tests. The high haplotype diversity (H = 1.0) was arranged in four validated haplogroups. Reticulate network topologies were evident, and 11 recombination events were validated through several tests. Five of these events occurred across species boundaries. Comparison with sequences from 70 Cercospora species indicated that at least five monophyletic groups of S-PSS-causing agents are currently present in South America. The provided evidence supports the hypothesis that interspecific genetic exchange plays a significant role in the evolutionary dynamics of Cercospora species in this region. The occurrence of interspecific recombination has implications for understanding epidemiological threats to soybean production that appear to be more serious than previously anticipated.



Similar content being viewed by others
Abbreviations
- S-PSS:
-
Soybean purple seed stain
- GCPSR:
-
Genealogical Concordance Phylogenetic Species Recognition
References
Akaike H (1973) Information theory and an extension of the maximum likelihood principle. In: Petrov BN, Csaki F (eds) Second international symposium on information theory. Akademiai Kiado, Budapest, pp 267–281
Albu S, Schneider RW, Price PP, Doyle VP (2016) Cercospora cf. flagellaris and Cercospora cf. sigesbeckiae are associated with Cercospora leaf blight and purple seed stain on soybean in North America. Phytopathology 106:1376–1385
Almeida ÁMR, Piuga FF, Marin SRR, Binneck E, Sartori F, Costamilan LM, Teixeira MR, Lopes M (2005) Pathogenicity, molecular characterization, and cercosporin content of Brazilian isolates of Cercospora kikuchii. Fitopatol Bras 30:594–602
Álvarez-Pérez S, de Vega C, Herrera CM (2013) Multilocus sequence analysis of nectar pseudomonads reveals high genetic diversity and contrasting recombination patterns. PLoS One 8:e75797
Bakhshi M, Arzanlou M, Babai-ahari A, Groenewald JZ, Braun U, Crous PW (2015) Application of the consolidated species concept to Cercospora spp. from Iran. Persoonia 34:65–86
Bandelt HJ, Forster P, Röhl A (1999) Median-joining networks for inferring intraspecific phylogenies. Mol Biol Evol 16:37–48
Bolton MD, Secor GA, Rivera V, Weiland JJ, Rudolph K, Birla K, Rengifo J, Campbell LG (2012) Evaluation of the potential for sexual reproduction in field populations of Cercospora beticola from USA. Fungal Biol 116:511–521
Boni MF, Posada D, Feldman MW (2007) An exact nonparametric method for inferring mosaic structure in sequence triplets. Genetics 176:1035–1047
Braun U, Nakashima C, Crous PW (2013) Cercosporoid fungi (Mycosphaerellaceae) 1. species on other fungi, Pteridophyta and Gymnospermae. Int Mycol Assoc Fungus 4:265–345
Braun U, Crous PW, Nakashima C (2014) Cercosporoid fungi (Mycosphaerellaceae) 2. species on monocots (Acoraceae to Xyridaceae, excluding Poaceae). Int Mycol Assoc Fungus 5:203–390
Braun U, Crous PW, Nakashima C (2015) Cercosporoid fungi (Mycosphaerellaceae) 3. species on monocots (Poaceae, true grasses). Int Mycol Assoc Fungus 6:25–97
Bruen TC, Philippe H, Bryant D (2006) A simple and robust statistical test for detecting the presence of recombination. Genetics 172:2665–2681
Bryant D, Moulton V (2004) Neighbor-Net: an agglomerative method for the construction of phylogenetic networks. Mol Biol Evol 21:255–265
Cai G, Schneider RW (2008) Population structure of Cercospora kikuchii, the causal agent of Cercospora leaf blight and purple seed stain in soybean. Phytopathology 98:823–829
Campbell V, Legendre P, Lapointe FJ (2011) The performance of the Congruence Among Distance Matrices (CADM) test in phylogenetic analysis. BMC Evol Biol 11:64
Chupp C (1954) A monograph of the fungus genus Cercospora. Ithaca, New York, 667 pp
Corlett M (1991) An annotated list of the published names in Mycosphaerella and Sphaerella. Mycological Memoir no. 18
Croll D, Sanders IR (2009) Recombination in Glomus intraradices, a supposed ancient asexual arbuscular mycorrhizal fungus. BMC Evol Biol 9:13
Crous PW, Braun U (2003) Mycosphaerella and its anamorphs: 1. Names published in Cercospora and Passalora. CBS Biodivers Ser 1:1–571
Crous PW, Aptroot A, Kang J-C, Braun U, Wingfield MJ (2000) The genus Mycosphaerella and its anamorphs. Stud Mycol 45:107–121
Crous PW, Groenewald JZ, Mansilla JP, Hunter GC, Wingfield MJ (2004a) Phylogenetic reassessment of Mycosphaerella spp. and their anamorphs occurring on Eucalyptus. Stud Mycol 50:195–214
Crous PW, Groenewald JZ, Pongpanich K, Himaman W, Arzanlou M, Wingfield MJ (2004b) Cryptic speciation and host specificity among Mycosphaerella spp. occurring on Australian Acacia species grown as exotics in the tropics. Stud Mycol 50:457–469
De Queiroz K (2007) Species concepts and species delimitation. Syst Biol 56:879–886
Dettman JR, Jacobson DJ, Turner E, Pringle A, Taylor JW (2003) Reproductive isolation and phylogenetic divergence in Neurospora: comparing methods of species recognition in a model eukaryote. Evolution 57:2721–2741
Douhan GW, Huryn KL, Douhan LAI (2007) Significant diversity and potential problems associated with inferring population structure within the Cenococcum geophilum species complex. Mycologia 99:812–819
Edgar RC (2004) MUSCLE: multiple sequence alignment with high accuracy and high throughput. Nucleic Acids Res 32:1792–1797
Edwards SV, Xi Z, Janke A, Faircloth BC, McCormack JE, Glenn TC, Zhong B, Wu S, Lemmon EM, Lemmon AR, Leaché AD, Liu L, Davis CC (2016) Implementing and testing the multispecies coalescent model: a valuable paradigm for phylogenomics. Mol Phylogenet Evol 94:447–462
Ellis MB (1971) Dematiaceous hyphomycetes. Commonwealth Mycological Institute, Kew, Surrey, UK, pp 1–608
Gams W, van der Aa HA, van der Plaats-Niterink AJ, Samsom RA, Stalpers JA (1987) CBS course of mycology. Centraalbureau voor Schimmelcultures, Institute of the Royal Netherlands
Gams W, Verkleij GJM, Crous PW (2007) CBS course of mycology, 5th edn. Centraalbureau voor Schimmelcultures, The Netherlands
Gibbs MJ, Armstrong JS, Gibbs AJ (2000) Sister-scanning: a Monte Carlo procedure for assessing signals in recombinant sequences. Bioinformatics 16:573–582
Giribet G, Wheeler WC (1999) On gaps. Mol Phylogenet Evol 13:132–143
González AM, Turino L, Latorre Rapela MG, Lurá MC (2008) Cercospora kikuchii aislada en la provincia de Santa Fe (Argentina): variabilidad genética y producción de cercosporina in vitro. Rev Iberoam Micol 25:237–241
Groenewald M, Groenewald JZ, Harrington TC, Abeln ECA, Crous PW (2006) Mating type gene analysis in apparently asexual Cercospora species is suggestive of cryptic sex. Fungal Genet Biol 43:813–825
Groenewald M, Linde CC, Groenewald JZ, Crous PW (2008) Indirect evidence for sexual reproduction in Cercospora beticola populations from sugar beet. Plant Pathol 57:25–32
Groenewald JZ, Nakashima C, Nishikawa J, Shin H-D, Park J-H, Jama AN, Groenewald M, Braun U, Crous PW (2013) Species concepts in Cercospora: spotting the weeds among the roses. Stud Mycol 75:115–170
Hasegawa M, Kishino H, Yano TA (1985) Dating of the human-ape splitting by a molecular clock of mitochondrial DNA. J Mol Evol 22:160–174
Hudson RR, Kaplan NL (1985) Statistical properties of the number of recombination events in the history of a sample of DNA sequences. Genetics 111:147–164
Huson DH, Bryant D (2006) Application of phylogenetic networks in evolutionary studies. Mol Biol Evol 23:254–267
Hyten DL, Song Q, Zhu Y, Choi I-Y, Nelson RL, Costa JM, Specht JE, Shoemaker RC, Cregan PB (2006) Impacts of genetic bottlenecks on soybean genome diversity. Proc Natl Acad Sci U S A 103:16666–16671
Jombart T (2008) adegenet: a R package for the multivariate analysis of genetic markers. Bioinformatics 24:1403–1405
Jombart T, Devillard S, Balloux F (2010) Discriminant analysis of principal components: a new method for the analysis of genetically structured populations. BMC Genet 11:94
Jukes TH, Cantor CR (1969) Evolution of protein molecules. In: Munro HN (ed) Mammalian protein metabolism. Academic Press, New York, pp 21–132
Kimura M (1980) A simple method for estimating evolutionary rates of base substitutions through comparative studies of nucleotide sequences. J Mol Evol 16:111–120
Latorre Rapela MG, Colombini MA, González AM, Vaira SM, Maumary R, Mattio MC, Carrera E, Lurá MC (2011) Phenotypic and genotypic variability in Cercospora kikuchii isolates from Santa Fe province, Argentina. In: Krezhova D (ed) Soybean—genetics and novel techniques for yield enhancement. InTech Open Access Publisher, Croatia, pp 97–112
Le Gac M, Hood ME, Fournier E, Giraud T (2007) Phylogenetic evidence of host-specific cryptic species in the anther smut fungus. Evolution 61:15–26
Lee G-A, Crawford GW, Liu L, Sasaki Y, Chen X (2011) Archaeological soybean (Glycine max) in East Asia: does size matter? PLoS One 6:e26720
Lepage BA, Currah RS, Stockey RA (1994) The fossil fungi of the Princeton chert. Int J Plant Sci 155:828–836
Lian S, Lee J-S, Cho WK, Yu J, Kim M-K, Choi H-S, Kim K-H (2013) Phylogenetic and recombination analysis of tomato spotted wilt virus. PLoS One 8:e63380
Librado P, Rozas J (2009) DnaSP v5: a software for comprehensive analysis of DNA polymorphism data. Bioinformatics 25:1451–1452
Liu F, Wang M, Damm U, Crous PW, Cai L (2016) Species boundaries in plant pathogenic fungi: a Colletotrichum case study. BMC Evol Biol 16:81
Lurá MC, Latorre Rapela MG, Vaccari MC, Maumary R, Soldano A, Mattio M, González AM (2011) Genetic diversity of Cercospora kikuchii isolates from soybean cultured in Argentina as revealed by molecular markers and cercosporin production. Mycopathologia 171:361–371
Mantel N (1967) The detection of disease clustering and a generalized regression approach. Cancer Res 27:209–220
Martin D, Rybicki E (2000) RDP: detection of recombination amongst aligned sequences. Bioinformatics 16:562–563
Martin DP, Murrell B, Golden M, Khoosal A, Muhire B (2015) RDP4: detection and analysis of recombination patterns in virus genomes. Virus Evol 1:1–5
Maynard Smith J (1992) Analyzing the mosaic structure of genes. J Mol Evol 34:126–129
McDonald BA, Linde C (2002) Pathogen population genetics, evolutionary potential, and durable resistance. Annu Rev Phytopathol 40:349–379
McVean GAT, Myers SR, Hunt S, Deloukas P, Bentley DR, Donnelly P (2004) The fine-scale structure of recombination rate variation in the human genome. Science 304:581–584
Morrison DA (2011) Introduction to phylogenetic networks. Available online at: http://www.rjr-productions.org/Networks/index.html
Nylander JAA (2004) MrModeltest v2. Program distributed by the author. Evolutionary Biology Centre, Uppsala University, Uppsala
O’Donnell K, Cigelnik E, Nirenberg HI (1998) Molecular systematics and phylogeography of the Gibberella fujikuroi species complex. Mycologia 90:465–493
Padidam M, Sawyer S, Fauquet CM (1999) Possible emergence of new geminiviruses by frequent recombination. Virology 265:218–225
Peterson SW (2008) Phylogenetic analysis of Aspergillus species using DNA sequences from four loci. Mycologia 100:205–226
Posada D, Crandall KA (2001) Evaluation of methods for detecting recombination from DNA sequences: computer simulations. Proc Natl Acad Sci U S A 98:13757–13762
Quaedvlieg W, Binder M, Groenewald JZ, Summerell BA, Carnegie AJ, Burgess TI, Crous PW (2014) Introducing the consolidated species concept to resolve species in the Teratosphaeriaceae. Persoonia 33:1–40
Rambaut A (2006–2014). FigTree v1.4.2. Available online at: http://tree.bio.ed.ac.uk/software/figtree/
Rambaut A, Suchard MA, Xie D, Drummond AJ (2014) Tracer v1.6. Available online at: http://beast.bio.ed.ac.uk/Tracer
Ronquist F, Huelsenbeck JP (2003) MrBayes 3: Bayesian phylogenetic inference under mixed models. Bioinformatics 19:1572–1574
Salminen MO, Carr JK, Burke DS, McCutchan FE (1995) Identification of breakpoints in intergenotypic recombinants of HIV type 1 by bootscannning. AIDS Res Hum Retrovir 11:1423–1425
Siboe GM, Murray J, Kirk PM (2000) Genetic similarity among Cercospora apii-group species and their detection in host plant tissue by PCR/RFLP analyses of the rDNA internal transcribed spacer (ITS). J Gen Appl Microbiol 46:69–78
Soares APG, Guillin EA, Borges LL, da Silva ACT, de Almeida ÁMR, Grijalba PE, Gottlieb AM, Bluhm BH, de Oliveira LO (2015) More Cercospora species infect soybeans across the Americas than meets the eye. PLoS One 10:e0133495
Starkey DE, Ward TJ, Aoki T, Gale LR, Kistler HC, Geiser DM, Suga H, Toth B, Varga J, O’Donnell K (2007) Global molecular surveillance reveals novel Fusarium head blight species and trichothecene toxin diversity. Fungal Genet Biol 44:1191–1204
Stewart EL, Liu Z, Crous PW, Szabo LJ (1999) Phylogenetic relationships among some cercosporoid anamorphs of Mycosphaerella based on rDNA sequence analysis. Mycol Res 103:1491–1499
Stewart JE, Timmer LW, Lawrence CB, Pryor BM, Peever TL (2014) Discord between morphological and phylogenetic species boundaries: incomplete lineage sorting and recombination results in fuzzy species boundaries in an asexual fungal pathogen. BMC Evol Biol 14:38
Tamura K (1992) Estimation of the number of nucleotide substitutions when there are strong transition-transversion and G + C-content biases. Mol Biol Evol 9:678–687
Tamura K, Stecher G, Peterson D, Filipski A, Kumar S (2013) MEGA6: molecular evolutionary genetics analysis version 6.0. Mol Biol Evol 30:2725–2729
Taylor JW, Jacobson DJ, Kroken S, Kasuga T, Geiser DM, Hibbett DS, Fisher MC (2000) Phylogenetic species recognition and species concepts in fungi. Fungal Genet Biol 31:21–32
To-Anun C, Hidayat I, Meeboon J (2011) Genus Cercospora in Thailand: taxonomy and phylogeny (with a dichotomous key to species). Plant Pathol Quar 1:11–87
Wen D, Yu Y, Nakhleh L (2016) Bayesian inference of reticulate phylogenies under the multispecies network coalescent. PLoS Genet 12:e1006006
Acknowledgements
We would like to express our sincere gratitude to the farmers and field technicians who provided assistance during field trips and sampling. We also wish to express our gratitude to B. Ferrari, M. Quiroga, E. Pozzo, D. Girotti, N. García Dutriez and M. Franco from “Don Mario Semillas” for supplying infected materials from different locations in Argentina. This research was financially supported by “Programa de Proyectos Conjuntos de Investigación SPU-CAPES MERCOSUR” (2011-PPC015), “Instituto Nacional de Tecnología Agropecuaria (INTA, Argentina)”, the Minas Gerais State Foundation of Research Aid—FAPEMIG (PPM 00561-15), the National Council of Scientific and Technological Development—CNPq (478752/2013-0 and 305827/2015-4) and the CAPES Foundation—Brazilian Ministry of Education (PPCP-Mercosul 015/2011 and AUX-PE PNPD 2927/2010).
Author information
Authors and Affiliations
Corresponding author
Additional information
Section Editor: Hans-Josef Schroers
Electronic supplementary material
Below are the links to the electronic supplementary material.
Supplementary Table S1
Materials used in this study, including geographic origin of isolates and GenBank accession numbers; the tentative identification of isolates follows Soares et al. (2015) where possible (DOC 131 kb)
Supplementary Table S2
Additional materials used in this study, including numbers of isolates, host names, geographic origin and GenBank accession numbers. Species names are those proposed in Groenewald et al. (2013). The numbers of ex-type isolates are indicated (DOC 250 kb)
Supplementary Table S3
Results of CADM tests performed pairwise amongst single loci matrices from dataset 1. Kendall test values (below the diagonal) and Mantel probabilities (above the diagonal) are shown. All probabilities are based upon 999 permutations (DOC 37 kb)
Supplementary Table S4
Results of CADM tests performed pairwise amongst single loci matrices from dataset 2. Kendall test values (below the diagonal) and Mantel probabilities (above the diagonal) are shown. All probabilities are based upon 999 permutations (DOC 28.5 kb)
Fig. S1
Median-joining network derived from dataset 1, excluding C. sojina (484 sites; 66 terminals). The colours in terminals indicate species identification: C. kikuchii and C. aff. kikuchii (blue); C. cf. flagellaris (red); C. cf. sigesbeckiae (purple); Cercospora sp. Q (green); C. alchemillicola (pink); Cercospora sp. H (orange); Cercospora sp. (black). The black squares represent putative or not sampled haplotypes (GIF 22.2 kb)
Fig. S2
Graphical haplotypes and haplogroups defined for dataset 1. The top line shows the gene order and the approximate size of each gene locus, as follows: act (bases 1–151), cal (152–395), cfp (396–1265), cyt-b (1266–1918), ef (1919–2154) and tub1/2 (2155–3351). Each grey block represents a unique haplotype; variable sites are depicted by vertical lines. The coloured small dots indicate species identification: C. kikuchii and C. aff. kikuchii (blue); C. cf. flagellaris (red); C. cf. sigesbeckiae (purple); Cercospora sp. Q (green); C. alchemillicola (pink); Cercospora sp. H (orange); Cercospora sp. (black). The black arrows indicate interlocus recombinants and the pink arrow shows a putative intralocus recombinant. Haplogroups (H) are delimited on the right, with the equivalent DAPC group assignment shown in parentheses (G) (GIF 148 kb)
Fig. S3
DAPC analyses of dataset 1 (excluding C. sojina; 3351 sites; 66 terminals). a Scatter plot depicting relative intragroup and among-group genetic distances. The first three axes explain 74%, 15.7% and 10.3% of the variation, respectively. The colours correspond to those used to delimit haplogroups. Individual isolates cannot be distinguished within each group and inertia ellipses are not apparent in the scatter plot due to the scale defined by the intragroup/intergroup variation rate. b Group assignment. The red blocks indicate full assignment probability (p = 1.0). Isolates are coloured according to their species identification: C. kikuchii and C. aff. kikuchii (blue); C. cf. flagellaris (red); C. cf. sigesbeckiae (purple); Cercospora sp. Q (green); C. alchemillicola (pink); Cercospora sp. H (orange); Cercospora sp. (black) (GIF 50.3 kb)
Rights and permissions
About this article
Cite this article
Guillin, E.A., de Oliveira, L.O., Grijalba, P.E. et al. Genetic entanglement between Cercospora species associating soybean purple seed stain. Mycol Progress 16, 593–603 (2017). https://doi.org/10.1007/s11557-017-1289-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11557-017-1289-x