Abstract
Aim
Our aim was to assess MRI findings in the acute phase of ON and their correlation with visual acuity at presentation, visual outcome (VO) and MS development, to analyze a possible correlation between lesions number and diagnosis, and to assess correlation between orbits MRI and OCT.
Materials and methods
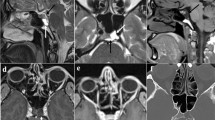
We retrospectively studied 37 patients, who presented to our Emergency Department with an ON first episode from January 2015 to January 2017. Patients underwent immediately a complete neuro-ophthalmological evaluation, blood test, CSF analysis. MRI of brain, orbits, cervical spine was executed within 7 days from ON onset. Brain MRI was classified as: normal, non-specific, suspected demyelination, lesions with dissemination in space and time. Optic nerves findings were localized in three sites (intra-orbital, canalicular and chiasmal) and classified as: normal, STIR- alteration, altered contrast enhancement. Patients underwent neuro-ophthalmological follow-up and MRI at 6 months to assess VO (complete recovery, partial recovery, deficit persistence). Another follow-up at 1 year was performed to identify MS or clinically isolated syndrome (CIS).
Results
64.8% patients received a diagnosis of MS; 35% of CIS. Lesions of the optic nerve were found in 65.8%. We observed statistically significant correlation between brain MRI pattern and diagnosis and between lesions number and diagnosis. We observed a statistically significant correlation between orbital MRI pattern and optical coherence tomography (OCT) results. MRI brain findings correlate with development of MS. MRI brain features and lesions number can predict the risk of MS conversion.



Similar content being viewed by others
References
Petzold A, Wattjes MP, Costello F, Flores-Rivera J et al (2014) The investigation of acute optic neuritis: a review and proposed protocol. Nat Rev Neurol 10(8):447–458. https://doi.org/10.1038/nrneurol.2014.108
Dooley MC, Foroozan R (2010) Optic neuritis. J Ophthalmic Vis Res 5(3):182–187
Soelberg K, Jarius S, Skejoe H, Engberg H, Mehlsen JJ, Nilsson AC, Madsen JS, Reindl M, Wildemann B, Grauslund J et al (2017) A population-based prospective study of optic neuritis. Mult Scler 23(14):1893–1901. https://doi.org/10.1177/1352458517734070
Soelberg K, Skejoe HPB, Grauslund J et al (2018) Magnetic resonance imaging findings at the first episode of acute optic neuritis. Mult Scler Relat Disord 20:30–36. https://doi.org/10.1016/j.msard.2017.12.018
Hirst CL, Ingram G, Pickersgill TP et al (2012) Temporal evolution of remission following multiple sclerosis relapse and predictors of outcome. Mult Scler 18(8):1152–1158. https://doi.org/10.1177/1352458511433919
Hickman SJ, Toosy AT, Jones SJ (2004) A serial MRI study following optic nerve mean area in acute optic neuritis. Brain 127(Pt 11):2498–2505. https://doi.org/10.1093/brain/awh284
Hickman SJ, Toosy AT, Miszkiel KA et al (2004) Visual recovery following acute optic neuritis-a clinical, electrophysiological and magnetic resonance imaging study. J Neurol 251(8):996–1005. https://doi.org/10.1007/s00415-004-0477-1
Kupersmith MJ, Mandel G, Anderson S et al (2011) Baseline, one and three month changes in the peripapillary retinal nerve fiber layer in acute optic neuritis: relation to baseline vision and MRI. J Neurol Sci 308(1–2):117–123. https://doi.org/10.1016/j.jns.2011.05.039
Jenkins TM, Toosy AT, Ciccarelli O (2010) Neuroplasticity predicts outcome of optic neuritis independent of tissue damage. Ann Neurol 67(1):99–113. https://doi.org/10.1002/ana.21823
Berg S, Kaschka I, Utz KS et al (2015) Baseline magnetic resonance imaging of the optic nerve provides limited predictive information on short-term recovery after acute optic neuritis. PLoS ONE 10(1):e0113961. https://doi.org/10.1371/journal.pone.0113961
Toosy AT, Mason DF, Miller DH (2014) Optic neuritis. Lancet Neurol 13(1):83–99. https://doi.org/10.1016/S1474-4422(13)70259-X
Behbehani R, Adnan H, Al-Hassan AA et al (2018) Predictors of retinal atrophy in multiple sclerosis: a longitudinal study using spectral domain optical coherence tomography with segmentation analysis. Mult Scler Relat Disord 11(21):56–62. https://doi.org/10.1016/j.msard.2018.02.010
Cellina M, Fetoni V, Ciocca M, Pirovano M, Oliva G (2018) Anti-myelin oligodendrocyte glycoprotein antibodies: magnetic resonance imaging findings in a case series and a literature review. Neuroradiol J 31(1):69–82. https://doi.org/10.1177/1971400917698856
Swanton JK, Fernando KT, Dalton CM et al (2010) Early MRI in optic neuritis: the risk for clinically definite multiple sclerosis. Mult Scler 16(2):156–165. https://doi.org/10.1177/1352458509353650
Polman CH, Reingold SC, Banwell B et al (2011) Diagnostic criteria for multiple sclerosis: 2010 revisions to the McDonald criteria. Ann Neurol 69(2):292–302. https://doi.org/10.1002/ana.22366
Thompson AJ, Banwell BL, Barkhof F et al (2018) Diagnosis of multiple sclerosis: 2017 revisions of the McDonald criteria. Lancet Neurol 17(2):162–173. https://doi.org/10.1016/S1474-4422(17)30470-2
Brusa A, Jones SJ, Plant GT (2001) Long-term remyelination after optic neuritis: A2-year visual evoked potential and psychophysical serial study. Brain 124(Pt 3):468–479
Smith SA, Williams ZR, Ratchford JN et al (2011) Diffusion tensorimaging of the optic nerve in multiple sclerosis: association with retinal damageand visual disability. AJNR Am J Neuroradiol 32(9):1662–1668. https://doi.org/10.3174/ajnr.A2574
Shams PN, Plant GT (2009) Optic neuritis: a review. Int MS J 16:82–89
Miller DH, Newton MR, van der Poel JC, du Boulay EP, Halliday AM, Kendall BE, Johnson G, MacManus DG, Moseley IF, McDonald WI (1988) Magnetic resonance imaging of the optic nerve in optic neuritis. Neurology 38(2):175–179
Deschamps R, Gout O, Fontaine B, Rigolet MH, Cabanis EA, Lyon-Caen O, Tourbah A (2002) Acute optic neuritis: clinical and MRI prognostic factors. Study of fifty patients. Rev Neurol (Paris) 158(4):446–452
Ramanathan S, Prelog K, Barnes EH, Tantsis EM, Reddel SW, Henderson AP, Vucic S, Gorman MP, Benson LA, Alper G, Riney CJ, Barnett M, Parratt JD, Hardy TA, Leventer RJ, Merheb V, Nosadini M, Fung VS, Brilot F, Dale RC (2016) Radiological differentiation of optic neuritis with myelin oligodendrocyte glycoprotein antibodies, aquaporin-4 antibodies, and multiple sclerosis. Mult Scler 22(4):470–482. https://doi.org/10.1177/1352458515593406
Kim HJ, Paul F, Lana-Peixoto MA, Tenembaum S, Asgari N, Palace J, Klawiter EC, Sato DK, de Seze J, Wuerfel J, Banwell BL, Villoslada P, Saiz A, Fujihara K, Kim SH, Guthy-Jackson Charitable Foundation NMO International Clinical Consortium & Biorepository (2015) MRI characteristics of neuromyelitis optica spectrum disorder: an international update. Neurology 84(11):1165–1173. https://doi.org/10.1212/WNL.0000000000001367
Kupersmith MJ, Alban T, Zeiffer B, Lefton D (2002) Contrast-enhanced MRI in acute optic neuritis: relationship to visual performance. Brain 125(Pt4):812–822
Dunker S, Wiegand W (1996) Prognostic value of magnetic resonance imaging in monosymptomatic optic neuritis. Ophthalmology 103(11):1768–1773
Zhang Y, Metz LM, Scott JN, Trufyn J, Fick GH, Costello F (2014) MRI texture heterogeneity in the optic nerve predicts visual recovery after acute optic neuritis. Neuroimage Clin 4:302–307
Ruet A, Deloire MS, Ouallet JC, Molinier S, Brochet B (2011) Predictive factors for multiple sclerosis in patients with clinically isolated spinal cord syndrome. Mutl Scler 19(12):1610–1617
Tintore M, Rovira A, Arrambide G et al (2010) Brainstem lesions in clinically isolated syndromes. Neurology 75(21):1933–1938
Sombekke MH, Wattjes MP, Balk LJ et al (2013) Spinal cord lesions in patients with clinically isolated syndrome: a powerful tool in diagnosis and prognosis. Neurology 80(1):69–75
Jenkins T, Ciccarelli O, Toosy A et al (2010) Dissecting structure-function interactions in acute optic neuritis to investigate neuroplasticity. Hum Brain Mapp 31(2):276–286. https://doi.org/10.1002/hbm.20863
Waldman AT, Hiremath G, Avery RA et al (2013) Monocular and binocular low-contrast visual acuity and optical coherence tomography in pediatric multiple sclerosis. Mult Scler Relat Disord 3(3):326–334. https://doi.org/10.1016/j.msard.2013.10.008
Jankowska-Lec I, Wasyluk J, Palasik W et al (2018) Peripapillary retinal nerve fiber layer thickness measured by optical coherence tomography in different clinical subtypes of multiple sclerosis. Mult Scler Relat Disord 27:260–268. https://doi.org/10.1016/j.msard.2018.11.003
Cettomai D, Hiremath G, Ratchford J et al (2010) Associations between retinal nerve fiber layer abnormalities and optic nerve examination. Neurology 75(15):1318–1325. https://doi.org/10.1212/WNL.0b013e3181f735bd
Costello F, Coupland S, Hodge W et al (2006) Quantifying axonal loss after optic neuritis with optical coherence tomography. Ann Neurol 59(6):963–969. https://doi.org/10.1002/ana.20851.37
Ramsaransing G, Maurits N, Zwanikken C et al (2001) Early prediction of a benign course of multiple sclerosis on clinical grounds: a systematic review. Mult Scler 7(5):345–347
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical standards
As stated in Materials and Methods, this retrospective study has been approved by our Institutional Review Board (Institutional Review Board Area 1 Milan; reference number: 2018/ST/116). All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and national research committee and with the 1964 Helsinki Declaration and its later amendments.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Cellina, M., Floridi, C., Rosti, C. et al. MRI of acute optic neuritis (ON) at the first episode: Can we predict the visual outcome and the development of multiple sclerosis (MS)?. Radiol med 124, 1296–1303 (2019). https://doi.org/10.1007/s11547-019-01073-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11547-019-01073-1