Abstract
Background
Systemic therapy using immune checkpoint inhibitors (ICIs) has recently become prevalent in the treatment of patients with various types of advanced cancers; however, difficulties are still associated with predicting the outcomes of patients receiving ICIs due to heterogenous responses to these agents.
Objective
To develop a prognostic model for advanced cancer patients treated with ICIs.
Patients and Methods
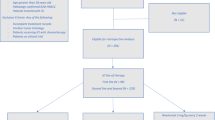
This study retrospectively analyzed the impact of clinical parameters on overall survival (OS) in 329 patients with several advanced solid malignant tumors who received systemic therapy using ICIs.
Results
The primary tumors of 329 patients were as follows: lung (n = 89), kidney (n = 70), urinary tract (n = 52), skin (n = 50), stomach (n = 30), esophagus (n = 21), and head and neck (n = 17). Median OS after the introduction of ICIs was 17.3 months. Among the factors that correlated with OS in a univariate analysis, body mass index, C-reactive protein, hemoglobin, lymphocytes, and platelets were identified as independent predictors of OS in a multivariate analysis. Following the classification of patients into 3 groups based on positive numbers of these independent risk factors, median OS was not reached in the favorable risk group with 0 or 1 risk factor (n = 76), 19.5 months in the intermediate-risk group with 2 or 3 risk factors (n = 182), and 7.2 months in the poor risk group (n = 71) with 4 or 5 risk factors.
Conclusions
Although this is a simple and objective model, it may be used as a reliable tool to predict the outcomes of advanced cancer patients receiving ICIs across multiple tumor types.


Similar content being viewed by others
References
Kalbasi A, Ribas A. Tumour-intrinsic resistance to immune checkpoint blockade. Nat Rev Immunol. 2020;20:25–39.
Lee JB, Ha SJ, Kim HR. Clinical insights Into novel immune checkpoint inhibitors. Front Pharmacol. 2021;12: 681320.
Hu-Lieskovan S, Malouf GG, Jacobs I, Chou J, Liu L, Johnson ML, et al. Addressing resistance to immune checkpoint inhibitor therapy: an urgent unmet need. Future Oncol. 2021;17:1401–39.
Chen JA, Ma W, Yuan J, Li T. Translational biomarkers and rationale strategies to overcome resistance to immune checkpoint inhibitors in solid tumors. Cancer Treat Res. 2020;180:251–79.
Lei Y, Li X, Huang Q, Zheng X, Liu M. Progress and challenges of predictive biomarkers for immune checkpoint blockade. Front Oncol. 2021;11: 617335.
Borghaei H, Paz-Ares L, Horn L, Spingel DR, Steins M, Ready NE, et al. Nivolumab versus docetaxel in advanced nonsquamous non-small-cll lung cancer. N Engl J Med. 2015;373:1627–39.
Rini BI, Plimack ER, Stus V, Gafanov R, Hawkins R, Nosov D, et al. Pembrolizumab plus axitinib versus sunitinib for advanced renal-cell carcinoma. N Engl J Med. 2019;380:1116–27.
Bellmunt J, de Wit R, Vaughn DJ, Fradet Y, Lee JL, Fong L, et al. Pembrolizumab as second-line therapy for advanced urothelial carcinoma. N Engl J Med. 2017;376:1015–26.
Postow MA, Chesney J, Pavlick AC, Robert C, Grossman K, McDermott D, et al. Nivolumab and ipilimumab versus ipilimumab in untreated melanoma. N Engl J Med. 2015;372:2006–17.
Shitara K, Ajani JA, Moehler M, Garrido M, Gallardo C, Shen L, et al. Nivolumab plus chemotherapy or ipilimumab in gastro-oesophageal cancer. Nature. 2022;603:942–8.
Ortega-Franco A, Hodgson C, Raja H, Carter M, Lindsay C, Hughes S, et al. Real-world data on pembrolizumab for pretreated non-small-cell lung cancer: clinical outcome and relevance of the lung immune prognostic index. Target Oncol. 2022;17:453–65.
Fucà G, Beninato T, Bini M, Mazzeo L, Di Guardo L, Cimminiello C, et al. The pan-immune-inflammation value in patients with metastatic melanoma receiving first-line therapy. Target Oncol. 2021;16:529–36.
Formica V, Morelli C, Patrikidou A, Murias C, Butt S, Nardecchia A, et al. Gastric inflammatory prognostic index (GIPI) in patients with metastatic gastro-esophageal junction/gastric cancer treated with PD-1/PD-L1 immune checkpoint inhibitors. Target Oncol. 2020;15:327–36.
Yamada T, Hayashi T, Inokuchi Y, Hayashi K, Watanabe H, Komori K, et al. Impact of the neutrophil-to-lymphocyte ratio on the survival of patients with gastric cancer treated with nivolumab monotherapy. Target Oncol. 2020;15:317–25.
Cortellini A, Bersanelli M, Buti S, Cannita K, Santini D, Perrone F, et al. A multicenter study of body mass index in cancer patients treated with anti-PD-1/PD-L1 immune checkpoint inhibitors: when overweight becomes favorable. J Immunother Cancer. 2019;7:57.
Haik L, Gonthier A, Quivy A, Gross-Goupil M, Veillon R, Frison E, et al. The impact of sarcopenia on the efficacy and safety of immune checkpoint inhibitors in patients with solid tumours. Acta Oncol. 2021;60:1597–603.
Valero C, Lee M, Hoen D, Weiss K, Kelly DW, Adusumilli PS, et al. Pretreatment neutrophil-to-lymphocyte ratio and mutational burden as biomarkers of tumor response to immune checkpoint inhibitors. Nat Commun. 2021;12:729.
Tjokrowidjaja A, Lord SJ, John T, Lewis CR, Kok PS, Marschner IC, et al. Pre- and on-treatment lactate dehydrogenase as a prognostic and predictive biomarker in advanced non-small cell lung cancer. Cancer. 2022;128:1574–83.
Ayers KL, Ma M, Debussche G, Corrigan D, McCafferty J, Lee K, et al. A composite biomarker of neutrophil-lymphocyte ratio and hemoglobin level correlates with clinical response to PD-1 and PD-L1 inhibitors in advanced non-small cell lung cancers. BMC Cancer. 2021;21:441.
Niu Z, Wang X, Xu Y, Li Y, Gong X, Zeng Q, et al. Development and validation of a novel survival model for cutaneous melanoma based on necroptosis-related genes. Front Oncol. 2022;12: 852803.
Zheng Y, Tang L, Liu Z. Multi-omics analysis of an immune-based prognostic predictor in non-small cell lung cancer. BMC Cancer. 2021;21:1322.
Kao C, Powers E, Wu Y, Datto MB, Green MF, Strickler JH, et al. Predictive value of combining biomarkers for clinical outcomes in advanced non-small cell lung cancer patients receiving immune checkpoint inhibitors. Clin Lung Cancer. 2021;22:500–9.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
No external funding was used in the preparation of this manuscripts.
Conflict of interest
AS, YI, HK, Kensuke F, Kazuhito F, AI, YM, KT, MK and HM declare that they have no conflicts of interest that might be relevant to the contents of this manuscript.
Ethics approval
This study was approved by the Research Ethics Committee of our institution.
Consent to participate
Informed consent from all of the patients has been exempted due to its retrospective design.
Consent for publication
Not applicable.
Availability of data and material
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Code availability
Not applicable.
Author contributions
AK was involved in data collection, contributed to data interpretation, and wrote the manuscript. YI, HK, Kensuke F, Kazuhito F, AI, YM and KT were involved in data collection. MK conceived the idea of the study.HM was involved in data collection and contributed to the interpretation of the results. All authors reviewed the manuscript draft and revised it critically on intellectual content. All authors approved the final version of the manuscript to be published.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Sano, A., Inoue, Y., Kikuchi, H. et al. Development of a Simple and Objective Prognostication Model for Patients with Advanced Solid Malignant Tumors Treated with Immune Checkpoint Inhibitors: A Pan-Cancer Analysis. Targ Oncol 17, 583–589 (2022). https://doi.org/10.1007/s11523-022-00911-z
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11523-022-00911-z