Abstract
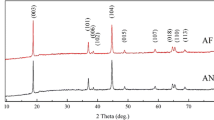
The combustion method for preparation of compound material like Li Co 0.5 Ni 0.45 Ag0.05 O2 cathodes is widely selected because it has the virtue of simplicity and lower cost. It was prepared by firing a mixture of stoichiometric amounts lithium nitrate (LiNO3), cobalt nitrate (Co(NO3)2.6H2O), nickel nitrate (Ni(NO3)2. 6H2O) and silver nitrates (AgNO3). The as-synthesized material was subjected to (TGA/DSC) analysis to determine the optimum range of annealing temperatures at 800, 900, and 1000 °C for 8 h. The effect of annealing on the structural and morphological features can be represented by X-ray diffraction (XRD), scanning electron microscopy (SEM), energy dispersive scattering (EDS), and transmutation electric microscopy (TEM), powders data from this method showed the coexistence of cubic Li Ni2O4 spinel structures at 400 °C. The optimum annealing result at 900 °C with constant duration 8 hours showed a single rhombohedra layered type Li Co 0.5 Ni 0.45 Ag0.05 O2 and polycrystalline structures.








Similar content being viewed by others
References
Christian J., Alain M., Ashok V. and Karim Z., Lithium batteries science and technology, (2016),ISBN 978-3-319-19108-9.
Keun K, Tae T, Joongpyo S (2008) Characteristics of LiNi1/3Co1/3Mn1/3O2 cathode powder prepared by different method in lithium rechargeable batteries. Int. J. lectrochem. Sci. 3:1504–1511
Petrucci R.H., Harwood W.S. and Herring F.G, General chemistry, 8th ed., (2002),pp. 341-2.
Patil VP, Pawar S, Chougule M, Godse P, Sakhare R, Sen S, Joshi P (2011) Effect of annealing on structural, morphological, electrical and optical studies of nickel oxide thin films. Journal of Surface Engineered Materials and Advanced Technology 1(02):35
Hosono E, Matsuda H, Honma I, Ichihara M, Zhou HJ, (2007) High-Rate Lithium Ion Batteries with Flat Plateau Based on Self-Nanoporous Structure of Tin Electrode. Electrochem Soc 154(2):A146–A149
Santiago EI, Andrade AVC, Paiva-Santos CO, Bulhoes LOS (2003) Structural and electrochemical properties of LiCoO2 prepared by combustion synthesis. Solid State Ionics 158(1):91–102
Kinoshita A, Yanagida K, Yanai A (2001) Electrochemical characteristics of LiNi1-xCoxO2 as positive electrode material for lithium secondary batteries. J Powder Sources 102:283–287
Fang HS, Wang ZX, Li XH, Guo HJ, Peng WJ (2006) Exploration of high capacity LiNi0.5Mn1.5O4 synthesized by solid-state reaction. J Power Sources 153(1):174–176
Andouni N, Zaghib K, Gendron F, Mauger A, Julien CM (2007) Magnetic properties of LiNi0.5Mn1.5O4 spinels prepared by wetchemical methods. J Magnetism Magnetic Materials 309:100–105
Fan Y, Wang J, Zhang J (2005) Physical properties and electrochemical performance of LiNi0.5Mn1.5O4 cathode material prepared by a coprecipitation method. [J]. Materials Chemistry and Physics, Acta 103:19–23
Liug Q, Wang Y, Chen H (2005) Synthesis and electrochemical performance of LiNi0.5Mn1.5O4 spinel compound. [J].Electrochimica, Acta 50:1965–1968
Kim J, Myung ST, Sun YK (2004) Molten salt synthesis of LiNi0.5Mn1.5O4 spinel for 5V class cathode material of Li-ion secondary battery. Electrochimica, Acta 49:219–227
Myung ST, Komaba S, Kumagai N, Yashiro H, Chung HT, Cho TH (2002) Nano-crystalline LiNi0.5Mn1.5O4 synthesized by emulsion drying method. Electrochim Acta 47(15):2543–2549
Sun Q, Li X, Wang Z, Ji Y (2009) Synthesis and electrochemical properties of 5V spinel LiNi0.5Mn1.5O4 cathode materials prepared by ultrasonicspray pyrolysis method. [J]. Nonferrous Met. Soc. China 19:176–181
Arrebola JC, Caballero A, Cruz M, Hernán L, Morales J, Castellón ER (2006) Crystallinity control of a nanostructured LiNi0.5 Mn1.5 O4 spinel via polymer‐assisted synthesis: A method for improving its rate capability and performance in 5 V lithium batteries. Adv Funct Mater 16(14):1904–1912
Alcantara R, Jaraba M, Lavela P, Tirada J (2002) Optimizing preparation conditions for 5V electrode performance, and structural changes in LiNi0.5Mn1.5O4 spinel [J]. Electrochimica Acta 47:1829–1835
Zhang B, Wang Z, Guo H (2007) Effect of annealing treatment on electrochemical property of LiNi0.5Mn1.5O4 spinel. [J].Trans Nonferrous Met Soc China 17(2):287–290
Ahmed T, Norlida K, Aseel Z (2015) Synthesis and properties of a spinel cathode material for lithium ion battery with at potential plateau. Turk J. Phys 39:187–198
Antolini E (2004) LiCoO2: formation, structure, lithium and oxygen nonstoichiometry, electrochemical behaviour and transport properties. Solid State Ionics 170:159–171
Feng C, Li H, Zhang C, Guo Z, Wu H, Tang J (2012) Synthesis and electrochemical properties of nonstoichiometric Li–Mn-spinel (Li1.02 Mx Mn1.95 O4− y Fy) for lithium ion battery application. Electrochimica Acta 61:87–93
Ohzuku T, Ariyoshi K, Takeda S, Sakai Y (2001) Synthesis and characterization of 5 V insertion material of Li [Fey Mn2−y] O4 for lithium-ion batteries. Electrochimica Acta 46(15):2327–2336
David W.I.F.,Shankland K., McCusker, L.B. and Baerlocher CH. (Eds), Structure determination from powder diffraction data, Oxford, (2002) ISBN 0-19-920553-1.
Kim D., Gim H. Lim J., Park, S. Kim J. Mater. Res. Bull. (2010), 45, 252.
Markova D.,Journal of the University of Chemical Technology and Metallurgy, (2010),45, 4, 351.
Yoo K, Hoang TK, Chen P (2014) Recent developments in synthesis of xLi2MnO3·(1− x) LiMO2 (M= Ni, Co, Mn) cathode powders for high-energy lithium rechargeable batteries. Frontiers Energy Res 2:36
Zhang X (2011) Aerosol Synthesis Of Cathode Materials For Li-Ion Batteries. Ph.D. Thesis, Washington University in St. Louis, Paper 674
Acknowledgments
The authors gratefully acknowledge the technical support provided by the Applied Science Department, Laser and Chemical Branch, University of Technology, and Baghdad, Iraq.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Haider, A.J., AL-Rsool, R.A. & Haider, M.J. Morphological and Structural Properties of Cathode Compound Material for Lithium-Ion Battery. Plasmonics 13, 1649–1657 (2018). https://doi.org/10.1007/s11468-017-0674-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11468-017-0674-2