Abstract
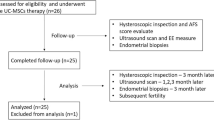
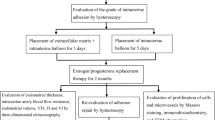
Intrauterine adhesion is a major cause of female reproductive disorders. Although we and others uncontrolled pilot studies showed that treatment with autologous bone marrow stem cells made a few patients with severe intrauterine adhesion obtain live birth, no large sample randomized controlled studies on this therapeutic strategy in such patients have been reported so far. To verify if the therapy of autologous bone marrow stem cells-scaffold is superior to traditional treatment in moderate to severe intrauterine adhesion patients in increasing their ongoing pregnancy rate, we conducted this randomized controlled clinical trial. Totally 195 participants with moderate to severe intrauterine adhesion were screened and 152 of them were randomly assigned in a 1:1 ratio to either group with autologous bone marrow stem cells-scaffold plus Foley balloon catheter or group with only Foley balloon catheter (control group) from February 2016 to January 2020. The per-protocol analysis included 140 participants: 72 in bone marrow stem cells-scaffold group and 68 in control group. The ongoing pregnancy occurred in 45/72 (62.5%) participants in the bone marrow stem cells-scaffold group which was significantly higher than that in the control group (28/68, 41.2%) (RR=1.52, 95%CI 1.08–2.12, P=0.012). The situation was similar in live birth rate (bone marrow stem cells-scaffold group 56.9% (41/72) vs. control group 38.2% (26/68), RR=1.49, 95%CI 1.04–2.14, P=0.027). Compared with control group, participants in bone marrow stem cells-scaffold group showed more menstrual blood volume in the 3rd and 6th cycles and maximal endometrial thickness in the 6th cycle after hysteroscopic adhesiolysis. The incidence of mild placenta accrete was increased in bone marrow stem cells-scaffold group and no severe adverse effects were observed. In conclusion, transplantation of bone marrow stem cells-scaffold into uterine cavities of the participants with moderate to severe intrauterine adhesion increased their ongoing pregnancy and live birth rates, and this therapy was relatively safe.
Similar content being viewed by others
References
AAGL Elevating Gynecologic Surgery. (2017). AAGL practice report: practice guidelines on intrauterine adhesions developed in collaboration with the European Society of Gynaecological Endoscopy (ESGE). J Minim Invasive Gynecol 24, 695–705.
Amer, M.I., Abd-El-Maeboud, K.H.I., Abdelfatah, I., Salama, F.A., and Abdallah, A.S. (2010). Human amnion as a temporary biologic barrier after hysteroscopic lysis of severe intrauterine adhesions: pilot study. J Minim Invasive Gynecol 17, 605–611.
American Fertility Society. (1988). The American Fertility Society classifications of adnexal adhesions, distal tubal occlusion, tubal occlusion secondary to tubal ligation, tubal pregnancies, mullerian anomalies and intrauterine adhesions. Fertil Steril 49, 944–955.
Bosteels, J., Weyers, S., D’Hooghe, T.M., Torrance, H., Broekmans, F.J., Chua, S.J., and Mol, B.W.J. (2017). Anti-adhesion therapy following operative hysteroscopy for treatment of female subfertility. Cochrane Database Syst Rev 11, CD011110.
Chen, Y., Liu, L., Luo, Y., Chen, M., Huan, Y., and Fang, R. (2017). Effects of aspirin and intrauterine balloon on endometrial repair and reproductive prognosis in patients with severe intrauterine adhesion: a prospective cohort study. Biomed Res Int 2017, 1–8.
Ding, L., Li, X., Sun, H., Su, J., Lin, N., Péault, B., Song, T., Yang, J., Dai, J., and Hu, Y. (2014). Transplantation of bone marrow mesenchymal stem cells on collagen scaffolds for the functional regeneration of injured rat uterus. Biomaterials 35, 4888–4900.
Dreyer, K., van Rijswijk, J., Mijatovic, V., Goddijn, M., Verhoeve, H.R., van Rooij, I.A.J., Hoek, A., Bourdrez, P., Nap, A.W., Rijnsaardt-Lukassen, H.G.M., et al. (2017). Oil-based or water-based contrast for hysterosalpingography in infertile women. N Engl J Med 376, 2043–2052.
Feng, L., Wang, L., Ma, Y., Duan, W., Martin-Saldaña, S., Zhu, Y., Zhang, X., Zhu, B., Li, C., Hu, S., et al. (2023). Engineering self-healing adhesive hydrogels with antioxidant properties for intrauterine adhesion prevention. Bioact Mater 27, 82–97.
Hanstede, M.M.F., van der Meij, E., Goedemans, L., and Emanuel, M.H. (2015). Results of centralized Asherman surgery, 2003–2013. Fertil Steril 104, 1561–1568.e1.
Higham, J.M., O’brien, P.M.S., and Shaw, R.W. (1990). Assessment of menstrual blood loss using a pictorial chart. Br J Obstet Gynaecol 97, 734–739.
Hooker, A.B., de Leeuw, R.A., van de Ven, P.M., Brölmann, H.A.M., and Huirne, J.A.F. (2018). Reproductive performance after the application of hyaluronic acid gel after dilation and curettage in women who have experienced at least one previous curettage: long-term results of a multicenter prospective randomized trial. Fertil Steril 110, 1231–1238.
Hooker, A.B., Lemmers, M., Thurkow, A.L., Heymans, M.W., Opmeer, B.C., Brolmann, H.A.M., Mol, B.W., and Huirne, J.A.F. (2014). Systematic review and meta-analysis of intrauterine adhesions after miscarriage: prevalence, risk factors and long-term reproductive outcome. Hum Reprod Update 20, 262–278.
Lin, X.N., Zhou, F., Wei, M.L., Yang, Y., Li, Y., Li, T.C., and Zhang, S.Y. (2015). Randomized, controlled trial comparing the efficacy of intrauterine balloon and intrauterine contraceptive device in the prevention of adhesion reformation after hysteroscopic adhesiolysis. Fertil Steril 104, 235–240.
Mogos, M.F., Salemi, J.L., Ashley, M., Whiteman, V.E., and Salihu, H.M. (2016). Recent trends in placenta accreta in the United States and its impact on maternal-fetal morbidity and healthcare-associated costs, 1998–2011. J Matern Fetal Neonatal Med 29, 1077–1082.
Myers, E.M., and Hurst, B.S. (2012). Comprehensive management of severe Asherman syndrome and amenorrhea. Fertil Steril 97, 160–164.
Nagori, C.B., Panchal, S.Y., and Patel, H. (2011). Endometrial regeneration using autologous adult stem cells followed by conception by in vitro fertilization in a patient of severe Asherman’s syndrome. J Hum Reprod Sci 4, 43–48.
Salzani, A., Yela, D.A., Gabiatti, J.R.E., Bedone, A.J., and Monteiro, I.M.U. (2007). Prevalence of uterine synechia after abortion evacuation curettage. Sao Paulo Med J 125, 261–264.
Santamaria, X., Cabanillas, S., Cervelló, I., Arbona, C., Raga, F., Ferro, J., Palmero, J., Remohí, J., Pellicer, A., and Simón, C. (2016). Autologous cell therapy with CD133+ bone marrow-derived stem cells for refractory Asherman’s syndrome and endometrial atrophy: a pilot cohort study. Hum Reprod 31, 1087–1096.
Schenker, J.G., and Margalioth, E.J. (1982). Intrauterine adhesions: an updated appraisal. Fertil Steril 37, 593–610.
Sher, G., and Fisch, J.D. (2002). Effect of vaginal sildenafil on the outcome of in vitro fertilization (IVF) after multiple IVF failures attributed to poor endometrial development. Fertil Steril 78, 1073–1076.
Shi, X., Saravelos, S.H., Zhou, Q., Huang, X., Xia, E., and Li, T.C. (2019). Prevention of postoperative adhesion reformation by intermittent intrauterine balloon therapy: a randomised controlled trial. BJOG 126, 1259–1266.
Singh, N., Mohanty, S., Seth, T., Shankar, M., Dharmendra, S., and Bhaskaran, S. (2014). Autologous stem cell transplantation in refractory Asherman’s syndrome: a novel cell based therapy. J Hum Reprod Sci 7, 93–98.
Singh, N., Shekhar, B., Mohanty, S., Kumar, S., Seth, T., and Girish, B. (2020). Autologous bone marrow-derived stem cell therapy for Asherman’s syndrome and endometrial atrophy: a 5-year follow-up study. J Hum Reprod Sci 13, 31–37.
Sroussi, J., Bourret, A., Pourcelot, A.G., Thubert, T., Lesavre, M., Legendre, G., Tuffet, S., Rousseau, A., and Benifla, J.L. (2022). Does hyaluronic acid gel reduce intrauterine adhesions after dilation and curettage in women with miscarriage.? A Multicentric randomized controlled trial (HYFACO Study). Am J Obstet Gynecol 227, 597.e1–597.e8.
Su, J., Ding, L., Cheng, J., Yang, J., Li, X., Yan, G., Sun, H., Dai, J., and Hu, Y. (2016). Transplantation of adipose-derived stem cells combined with collagen scaffolds restores ovarian function in a rat model of premature ovarian insufficiency. Hum Reprod 31, 1075–1086.
Trinh, T.T., Nguyen, K.D., Pham, H.V., Ho, T.V., Nguyen, H.T., O’Leary, S., Le, H.T.T., and Pham, H.M. (2022). Effectiveness of hyaluronic acid gel and intrauterine devices in prevention of intrauterine adhesions after hysteroscopic adhesiolysis in infertile women. J Minim Invasive Gynecol 29, 284–290.
Tang, F., Tang, J., Zhao, Y., Zhang, J., Xiao, Z., Chen, B., Han, G., Yin, N., Jiang, X., Zhao, C., et al. (2022). Long-term clinical observation of patients with acute and chronic complete spinal cord injury after transplantation of NeuroRegen scaffold. Sci China Life Sci 65, 909–926.
Vitale, S.G., Riemma, G., Carugno, J., Perez-Medina, T., Alonso Pacheco, L., Haimovich, S., Parry, J.P., Di Spiezio Sardo, A., and De Franciscis, P. (2022). Postsurgical barrier strategies to avoid the recurrence of intrauterine adhesion formation after hysteroscopic adhesiolysis: a network meta-analysis of randomized controlled trials. Am J Obstet Gynecol 226, 487–498.e8.
Wang, Q., He, X., Wang, B., Pan, J., Shi, C., Li, J., Wang, L., Zhao, Y., Dai, J., and Wang, D. (2021). Injectable collagen scaffold promotes swine myocardial infarction recovery by long-term local retention of transplanted human umbilical cord mesenchymal stem cells. Sci China Life Sci 64, 269–281.
Wang, Y., Zhao, Y., Ge, Y., Cen, J., Liao, Y., and Xu, G. (2022). Reproductive outcomes and reproductive tract microbiota shift in women with moderate-to-severe intrauterine adhesions following 30-day post-hysteroscopic placement of balloon stents or intrauterine contraceptive devices: a randomized controlled trial. Eclinicalmedicine 43, 101200.
Xu, L., Ding, L., Wang, L., Cao, Y., Zhu, H., Lu, J., Li, X., Song, T., Hu, Y., and Dai, J. (2017). Umbilical cord-derived mesenchymal stem cells on scaffolds facilitate collagen degradation via upregulation of MMP-9 in rat uterine scars. Stem Cell Res Ther 8, 84.
Yu, D., Wong, Y.M., Cheong, Y., Xia, E., and Li, T.C. (2008). Asherman syndrome-one century later. Fertil Steril 89, 759–779.
Zhao, G., Cao, Y., Zhu, X., Tang, X., Ding, L., Sun, H., Li, J., Li, X., Dai, C., Ru, T., et al. (2017). Transplantation of collagen scaffold with autologous bone marrow mononuclear cells promotes functional endometrium reconstruction via down-regulating ΔNp63 expression in Asherman’s syndrome. Sci China Life Sci 60, 404–416.
Zhao, G., Li, R., Cao, Y., Song, M., Jiang, P., Wu, Q., Zhou, Z., Zhu, H., Wang, H., Dai, C., et al. (2020). ΔNp63α-induced DUSP4/GSK3β/SNAI1 pathway in epithelial cells drives endometrial fibrosis. Cell Death Dis 11, 449.
Acknowledgement
This work was supported by the Strategic Priority Research Program of the Chinese Academy of Sciences (XDA16040302, XDA01030505), the National Natural Science Foundation of China (81971336), Jiangsu Provincial Key Medical Center (YXZXB2016004), and Jiangsu Provincial Obstetrics and Gynecology Innovation Center (CXZX202229). The funding sources had no role in the study design, data collection and analysis, decision to publish, or preparation of the manuscript. We thank Yaling Li from Department of Obstetrics and Gynecology, Nanjing Drum Tower Hospital, Affiliated Hospital of Medical School, Nanjing University for assistance in participants care, Biyun Xu from Department of Biostatistics Nanjing Drum Tower Hospital, Affiliated Hospital of Medical School, Nanjing University for her help in randomization, Jian Dai from Clinical Trial Institution Office of Nanjing Drum Tower Hospital, Affiliated Hospital of Medical School, Nanjing University for monitoring the process of the clinical trial, Yihua Zhou from Departments of Laboratory Medicine and Infectious Diseases, Nanjing Drum Tower Hospital, Affiliated Hospital of Medical School, Nanjing University for his help in polishing the language, and all the dedicated staff in the group and participants included.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
The author(s) declare that they have no conflict of interest. For studies of human participants, we conformed with the Helsinki Declaration of 1975 (as revised in 2008) concerning Human Rights, and that we followed out policy concerning Informed Consent as shown on Springer.com.
Electronic Supplementary Material
Rights and permissions
About this article
Cite this article
Zhu, H., Li, T., Xu, P. et al. Effect of autologous bone marrow stem cells-scaffold transplantation on the ongoing pregnancy rate in intrauterine adhesion women: a randomized, controlled trial. Sci. China Life Sci. 67, 113–121 (2024). https://doi.org/10.1007/s11427-023-2403-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11427-023-2403-7