Abstract
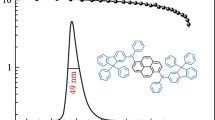
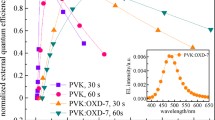
The development of high-performance solution-processed red organic light-emitting diodes (OLEDs) remains a challenge, particularly in terms of maintaining efficiency at high luminance. Here, we designed and synthesized four novel orange-red thermally activated delayed fluorescence (TADF) dendrimers that are solution-processable: 2GCzBP, 2DPACzBP, 2FBP2GCz and 2FBP2DPACz. We systematically investigated the effect of substitution position and strength of donors on the optoelectronic properties. The reverse intersystem crossing rate constant (kRISC) of the emitters having donors substituted at positions 11 and 12 of the dibenzo[a,c]phenazine (BP) is more than 10-times faster than that of compounds substituted having donors substituted at positions 3 and 6. Compound 2DPACzBP, containing stronger donors than 2GCzBP, exhibits a red-shifted emission and smaller singlet-triplet energy splitting, ΔEST, of 0.01 eV. The solution-processed OLED with 10 wt% 2DPACzBP doped in mCP emitted at 640 nm and showed a maximum external quantum efficiency (EQEmax) of 7.8%, which was effectively maintained out to a luminance of 1,000 cd m−2. Such a device∙s performance at relevant display luminance is among the highest for solution-processed red TADF OLEDs. The efficiency of the devices was improved significantly by using 4CzIPN as an assistant dopant in a hyperfluorescence (HF) configuration, where the 2DPACzBP HF device shows an EQEmax of 20.0% at λEL of 605 nm and remains high at 11.8% at a luminance of 1,000 cd m−2, which makes this device one of the highest efficiency orange-to-red HF SP-OLEDs to date.

Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Sun D, Si C, Wang T, Zysman-Colman E. Adv Photon Res, 2022, 3: 2200203
Zou SJ, Shen Y, Xie FM, Chen JD, Li YQ, Tang JX. Mater Chem Front, 2020, 4: 788–820
Huang T, Jiang W, Duan L. J Mater Chem C, 2018, 6: 5577–5596
Wong MY, Zysman-Colman E. Adv Mater, 2017, 29: 1605444
Uoyama H, Goushi K, Shizu K, Nomura H, Adachi C. Nature, 2012, 492: 234–238
Thejo Kalyani N, Dhoble SJ. Renew Sustain Energy Rev, 2012, 16: 2696–2723
Wang S, Zhang H, Zhang B, Xie Z, Wong WY. Mater Sci Eng-R-Rep, 2020, 140: 100547
Xie Y, Li Z. J Polym Sci Part A-Polym Chem, 2017, 55: 575–584
Sun D, Duda E, Fan X, Saxena R, Zhang M, Bagnich S, Zhang X, Köhler A, Zysman-Colman E. Adv Mater, 2022, 34: 2110344
Sun D, Saxena R, Fan X, Athanasopoulos S, Duda E, Zhang M, Bagnich S, Zhang X, Zysman-Colman E, Kohler A. Adv Sci, 2022, 9: 2201470
Abd-El-Aziz AS, Abdelghani AA, Wagner BD, Bissessur R. Macromol Rapid Commun, 2019, 40: 1800711
Albrecht K, Matsuoka K, Fujita K, Yamamoto K. Angew Chem Int Ed, 2015, 54: 5677–5682
Burn PL, Lo SC, Samuel IDW. Adv Mater, 2007, 19: 1675–1688
Zhao G, Liu D, Wang P, Huang X, Chen H, Zhang Y, Zhang D, Jiang W, Sun Y, Duan L. Angew Chem Int Ed, 2022, 61: e202212861
Zeng S, Xiao C, Zhou J, Dong Q, Li Q, Lim J, Ma H, Lee JY, Zhu W, Wang Y. Adv Funct Mater, 2022, 32: 2113183
Liu J, Chen L, Wang X, Yang Q, Zhao L, Tong C, Wang S, Shao S, Wang L. Macromol Rapid Commun, 2022, 43: 2200079
Kim YH, Wolf C, Cho H, Jeong SH, Lee TW. Adv Mater, 2016, 28: 734–741
Wada Y, Kubo S, Kaji H. Adv Mater, 2018, 30: 1705641
Wilson JS, Chawdhury N, Al-Mandhary MRA, Younus M, Khan MS, Raithby PR, Köhler A, Friend RH. J Am Chem Soc, 2001, 123: 9412–9417
Caspar JV, Kober EM, Sullivan BP, Meyer TJ. J Am Chem Soc, 1982, 104: 630–632
Yu Y, Hu Y, Yang S, Luo W, Yuan Y, Peng C, Liu J, Khan A, Jiang Z, Liao L. Angew Chem Int Ed, 2020, 59: 21578–21584
Li X, Wang K, Shi YZ, Zhang M, Dai GL, Liu W, Zheng CJ, Ou XM, Zhang XH. J Mater Chem C, 2018, 6: 9152–9157
Zhang Y, Wu J, Song J, Chen Z, He J, Wang X, Liu H, Chen S, Qu J, Wong W. Adv Elect Mater, 2019, 5: 1800677
Wang B, Yang H, Zhang Y, Xie G, Ran H, Wang T, Fu Q, Ren Y, Sun N, Zhao G, Hu JY, Wang Q. J Mater Chem C, 2019, 7: 12321–12327
Zeng W, Zhou T, Ning W, Zhong C, He J, Gong S, Xie G, Yang C. Adv Mater, 2019, 31: 1901404
Chen J, Xiao Y, Wang K, Sun D, Fan X, Zhang X, Zhang M, Shi Y, Yu J, Geng F, Lee C, Zhang X. Angew Chem Int Ed, 2021, 60: 2478–2484
Sharma S, Pal AK. J Mater Chem C, 2022, 10: 15681–15707
Kim JH, Yun JH, Lee JY. Adv Opt Mater, 2018, 6: 1800255
Adamo C, Barone V. J Chem Phys, 1999, 110: 6158–6170
Petersson GA, Tensfeldt TG, Montgomery J. J Chem Phys, 1991, 94: 6091–6101
Kerwin SM. J Am Chem Soc, 2010, 132: 2466–2467
Hirata S, Head-Gordon M. Chem Phys Lett, 1999, 314: 291–299
Connelly NG, Geiger WE. Chem Rev, 1996, 96: 877–910
Duda E, Hall D, Bagnich S, Carpenter-Warren CL, Saxena R, Wong MY, Cordes DB, Slawin AMZ, Beljonne D, Olivier Y, Zysman-Colman E, Köhler A. J Phys Chem B, 2022, 126: 552–562
Englman R, Jortner J. Mol Phys, 1970, 18: 145–164
Xue J, Liang Q, Wang R, Hou J, Li W, Peng Q, Shuai Z, Qiao J. Adv Mater, 2019, 31: 1808242
Wu L, Huang C, Emery BP, Sedgwick AC, Bull SD, He XP, Tian H, Yoon J, Sessler JL, James TD. Chem Soc Rev, 2020, 49: 5110–5139
Liu Y, Chen Y, Li H, Wang S, Wu X, Tong H, Wang L. ACS Appl Mater Interfaces, 2020, 12: 30652–30658
Xie FM, Li HZ, Dai GL, Li YQ, Cheng T, Xie M, Tang JX, Zhao X. ACS Appl Mater Interfaces, 2019, 11: 26144–26151
Zeng X, Zhou J, Zou S, Tang Y, Li H, He Y, Li Y, Wang W, Tang J. Adv Opt Mater, 2022, 10: 2200277
Chan CY, Tanaka M, Lee YT, Wong YW, Nakanotani H, Hatakeyama T, Adachi C. Nat Photon, 2021, 15: 203–207
Sun JW, Lee JH, Moon CK, Kim KH, Shin H, Kim JJ. Adv Mater, 2014, 26: 5684–5688
Wu S, Kumar Gupta A, Yoshida K, Gong J, Hall D, Cordes DB, Slawin AMZ, Samuel IDW, Zysman-Colman E. Angew Chem Int Ed, 2022, 61: e202213697
Kim HS, Park SR, Suh MC. J Phys Chem C, 2017, 121: 13986–13997
Chen D, Cai X, Li XL, He Z, Cai C, Chen D, Su SJ. J Mater Chem C, 2017, 5: 5223–5231
Woo JY, Park MH, Jeong SH, Kim YH, Kim B, Lee TW, Han TH. Adv Mater, 2023, 35: 2207454
Murawski C, Leo K, Gather MC. Adv Mater, 2013, 25: 6801–6827
Oda S, Kawakami B, Yamasaki Y, Matsumoto R, Yoshioka M, Fu-kushima D, Nakatsuka S, Hatakeyama T. J Am Chem Soc, 2022, 144: 106–112
Hasan M, Saggar S, Shukla A, Bencheikh F, Sobus J, McGregor SKM, Adachi C, Lo SC, Namdas EB. Nat Commun, 2022, 13: 254
Acknowledgements
Changfeng Si thanks the China Scholarship Council (201806890001). Dianming Sun acknowledges support from the Royal Academy of Engineering Enterprise Fellowship (EF2122-13106). We thank EPSRC (EP/W015137/1, EP/W524505/1) for financial support.
Funding
Funding note Open Access funding enabled and organized by Projekt DEAL.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest The authors declare no conflict of interest.
Additional information
Supporting information The supporting information is available online at chem.scichina.com and link.springer.com/journal/11426. The supporting materials are published as submitted, without typesetting or editing. The responsibility for scientific accuracy and content remains entirely with the authors. The research data underpinning this publication can be accessed at https://doi.org/10.17630/e89b2529-b213-4a5f-9102-5ed7a2a30ebc
Supporting Information
11426_2023_1945_MOESM1_ESM.pdf
Rational Molecular Design of Efficient Yellow-Red Dendrimer TADF for Solution-Processed OLEDs: A Combined Effect of Substitution Position and Strength of the Donors
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article∙s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article∙s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Si, C., Sun, D., Matulaitis, T. et al. Rational molecular design of efficient yellow-red dendrimer TADF for solution-processed OLEDs: a combined effect of substitution position and strength of the donors. Sci. China Chem. 67, 1613–1623 (2024). https://doi.org/10.1007/s11426-023-1945-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11426-023-1945-8