Abstract
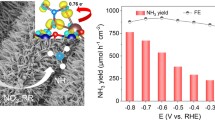
Ammonia production via electrochemical nitrate reduction is essential for environmental protection and the emerging hydrogen economy. Complex nitrate wastewater with a wide pH range calls for flexible catalysts with high selectivity. A high Faradaic efficiency (FE) of NH3 cannot be obtained under strong acid or alkaline conditions due to the uncontrollable adsorption energy and coverage of hydrogen species (H*) on active sites. This article describes the design and fabrication of a copper-palladium (Cu-Pd) alloy nanocrystal catalyst that inhibits H2 and nitrite generation in electrolytes with different nitrate concentrations and varied pH. The interfacial sites of Cu-Pd alloys could enhance the adsorption energy and coverage of H* while increasing the reaction rate constant of NO2*-to-NO*, which achieves a rapid conversion of NO2* along with a decreased FE of NO2−. Under ambient conditions, optimal FE(NH3) is close to 100% at a wide pH range, with the solar-to-chemical conversion efficiency approaching 4.29%. The combination of thermodynamics and kinetics investigations would offer new insights into the reduction mechanism of NO2* for further development of nitrate reduction.

Similar content being viewed by others
References
Daiyan R, MacGill I, Amal R. ACS Energy Lett, 2020, 5: 3843–3847
Ling Y, Ma Q, Yu Y, Zhang B. Trans Tianjin Univ, 2021, 27: 180–200
Rosca V, Duca M, de Groot MT, Koper MTM. Chem Rev, 2009, 109: 2209–2244
Battino R, Rettich TR, Tominaga T. J Phys Chem Reference Data, 1984, 13: 563–600
Xue ZH, Zhang SN, Lin YX, Su H, Zhai GY, Han JT, Yu QY, Li XH, Antonietti M, Chen JS. J Am Chem Soc, 2019, 141: 14976–14980
Luo Y, Chen GF, Ding L, Chen X, Ding LX, Wang H. Joule, 2019, 3: 279–289
Li C, Wang T, Gong J. Trans Tianjin Univ, 2020, 26: 67–91
Xu T, Ma B, Liang J, Yue L, Liu Q, Li T, Zhao H, Luo Y, Lu S, Sun X. Acta Phys Chim Sin, 2020, 0: 2009043-
Hirakawa H, Hashimoto M, Shiraishi Y, Hirai T. ACS Catal, 2017, 7: 3713–3720
Zhang X, Wang Y, Liu C, Yu Y, Lu S, Zhang B. Chem Eng J, 2021, 403: 126269
Jia R, Wang Y, Wang C, Ling Y, Yu Y, Zhang B. ACS Catal, 2020, 10: 3533–3540
Yu Y, Wang C, Yu Y, Wang Y, Zhang B. Sci China Chem, 2020, 63: 1469–1476
Chen GF, Yuan Y, Jiang H, Ren SY, Ding LX, Ma L, Wu T, Lu J, Wang H. Nat Energy, 2020, 5: 605–613
Li J, Zhan G, Yang J, Quan F, Mao C, Liu Y, Wang B, Lei F, Li L, Chan AWM, Xu L, Shi Y, Du Y, Hao W, Wong PK, Wang J, Dou SX, Zhang L, Yu JC. J Am Chem Soc, 2020, 142: 7036–7046
Wu ZY, Karamad M, Yong X, Huang Q, Cullen DA, Zhu P, Xia C, Xiao Q, Shakouri M, Chen FY, Kim JYT, Xia Y, Heck K, Hu Y, Wong MS, Li Q, Gates I, Siahrostami S, Wang H. Nat Commun, 2021, 12: 2870
van Langevelde PH, Katsounaros I, Koper MTM. Joule, 2021, 5: 290–294
Su L, Li K, Zhang H, Fan M, Ying D, Sun T, Wang Y, Jia J. Water Res, 2017, 120: 1–11
Vetter KJ. Electrochemical Kinetics: Theoretical Aspects. New York: Elsevier, 2013
Liu H, Park J, Chen Y, Qiu Y, Cheng Y, Srivastava K, Gu S, Shanks BH, Roling LT, Li W. ACS Catal, 2021, 11: 8431–8442
Pérez-Gallent E, Figueiredo MC, Katsounaros I, Koper MTM. Electrochim Acta, 2017, 227: 77–84
Li L, Wang P, Shao Q, Huang X. Chem Soc Rev, 2020, 49: 3072–3106
Liang Z, Ahn HS, Bard AJ. J Am Chem Soc, 2017, 139: 4854–4858
Duca M, Koper MTM. Energy Environ Sci, 2012, 5: 9726–9742
Kunimatsu K, Senzaki T, Tsushima M, Osawa M. Chem Phys Lett, 2005, 401: 451–454
Krstajić N, Popović M, Grgur B, Vojnović M, Šepa D. J Electroanal Chem, 2001, 512: 16–26
Ji L, Peng X, Wang Z. Trans Tianjin Univ, 2020, 26: 373–381
Li M, Zheng X, Li L, Wei Z. Acta Phys-Chim Sin, 2020, 37: 2007050–2007054
Chen J, Pan A, Zhang W, Cao X, Lu R, Liang S, Cao G. Sci China Mater, 2021, 64: 1150–1158
Chauhan R, Srivastava VC. Chem Eng J, 2020, 386: 122065
Feng JX, Wu JQ, Tong YX, Li GR. J Am Chem Soc, 2018, 140: 610–617
Wang M, Cheng X, Ni Y. Dalton Trans, 2019, 48: 823–832
Kannimuthu K, Sangeetha K, Sam Sankar S, Karmakar A, Madhu R, Kundu S. Inorg Chem Front, 2021, 8: 234–272
Zeradjanin AR, Vimalanandan A, Polymeros G, Topalov AA, Mayrhofer KJJ, Rohwerder M. Phys Chem Chem Phys, 2017, 19: 17019–17027
Ferrin P, Kandoi S, Nilekar AU, Mavrikakis M. Surf Sci, 2012, 606: 679–689
Nørskov JK, Bligaard T, Logadottir A, Kitchin JR, Chen JG, Pandelov S, Stimming U. J Electrochem Soc, 2005, 152: J23
Watson GW, Wells RPK, Willock DJ, Hutchings GJ. J Phys Chem B, 2000, 105: 4889–4894
Luc W, Rosen J, Jiao F. Catal Today, 2017, 288: 79–84
Dima GE, de Vooys ACA, Koper MTM. J Electroanal Chem, 2003, 554–555: 15–23
Queffélec C, Forato F, Bujoli B, Knight DA, Fonda E, Humbert B. Phys Chem Chem Phys, 2020, 22: 2193–2199
Torreggiani A, Esposti AD, Tamba M, Marconi G, Fini G. J Raman Spectrosc, 2006, 37: 291–298
Henson MJ, Vance MA, Zhang CX, Liang HC, Karlin KD, Solomon EI. J Am Chem Soc, 2003, 125: 5186–5192
Thamann TJ, Frank P, Willis LJ, Loehr TM. Proc Natl Acad Sci USA, 1982, 79: 6396–6400
Fujisawa K, Lehnert N, Ishikawa Y, Okamoto K. Angew Chem, 2004, 116: 5052–5055
Wang Y, Xu A, Wang Z, Huang L, Li J, Li F, Wicks J, Luo M, Nam DH, Tan CS, Ding Y, Wu J, Lum Y, Dinh CT, Sinton D, Zheng G, Sargent EH. J Am Chem Soc, 2020, 142: 5702–5708
Chen FY, Wu ZY, Gupta S, Rivera DJ, Lambeets SV, Pecaut S, Kim JYT, Zhu P, Finfrock YZ, Meira DM, King G, Gao G, Xu W, Cullen DA, Zhou H, Han Y, Perea DE, Muhich CL, Wang H. Nat Nanotechnol, 2022, 17: 759–767
Niu H, Zhang Z, Wang X, Wan X, Kuai C, Guo Y. Small, 2021, 17: 2102396
Liu JX, Richards D, Singh N, Goldsmith BR. ACS Catal, 2019, 9: 7052–7064
Machado SAS, Avaca LA. Electrochim Acta, 1994, 39: 1385–1391
Rebollar L, Intikhab S, Snyder JD, Tang MH. J Electrochem Soc, 2018, 165: J3209–J3221
Tian X, Zhao P, Sheng W. Adv Mater, 2019, 31: 1808066
Acknowledgements
This work was supported by the National Key R&D Program of China (2021YFA1500804), the National Natural Science Foundation of China (22121004, 51861125104), the Natural Science Foundation of Tianjin City (18JCJQJC47500), Haihe Laboratory of Sustainable Chemical Transformations, the Program of Introducing Talents of Discipline to Universities (BP0618007) and the Xplorer Prize.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest The authors declare no conflict of interest.
Additional information
Supporting information The supporting information is available online at https://chem.scichina.com and https://link.springer.com/journal/11426. The supporting materials are published as submitted, without typesetting or editing. The responsibility for scientific accuracy and content remains entirely with the authors.
Supporting Information
Rights and permissions
About this article
Cite this article
Wang, Y., Zhang, P., Lin, X. et al. Wide-pH-range adaptable ammonia electrosynthesis from nitrate on Cu-Pd interfaces. Sci. China Chem. 66, 913–922 (2023). https://doi.org/10.1007/s11426-022-1411-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11426-022-1411-0