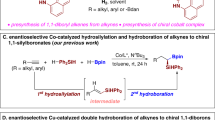
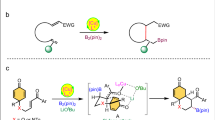
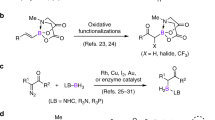
Abstract
Functionalized bisboryl compounds have recently emerged as a new class of synthetically useful building blocks in organic synthesis. Herein, we report an efficient strategy to synthesize β-geminal-diboryl ketones enabled by a Cu/Pd-catalyzed borocarbonylative trifunctionalization of readily available alkynes and allenes. This reaction promises to be a useful method for the synthesis of functionalized β-geminal-diboryl ketones with broad functional group tolerance. Mechanistic studies suggest that the reaction proceeds through borocarbonylation/hydroboration cascade of both alkynes and allenes.

Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Zeni G, Larock RC. Chem Rev, 2004, 104: 2285–2310
Cresswell AJ, Eey STC, Denmark SE. Angew Chem Int Ed, 2015, 54: 15642–15682
Hoffmann-Röder A, Krause N. Angew Chem Int Ed, 2004, 43: 1196–1216
Yu S, Ma S. Angew Chem Int Ed, 2012, 51: 3074–3112
Yin G, Mu X, Liu G. Acc Chem Res, 2016, 49: 2413–2423
Derosa J, Tran VT, van der Puyl VA, Engle KM. Aldrichimica Acta, 2018, 51: 21–32
Ma S. Chem Rev, 2005, 105: 2829–2872
Yang B, Qiu Y, Bäckvall JE. Acc Chem Res, 2018, 51: 1520–1531
Cantrell CL, Dayan FE, Duke SO. J Nat Prod, 2012, 75: 1231–1242
He J, Qiu D, Li Y. Acc Chem Res, 2020, 53: 508–519
Cheng LJ, Mankad NP. J Am Chem Soc, 2017, 139: 10200–10203
Gao DW, Gao Y, Shao H, Qiao TZ, Wang X, Sanchez BB, Chen JS, Liu P, Engle KM. Nat Catal, 2020, 3: 23–29
Hemming D, Fritzemeier R, Westcott SA, Santos WL, Steel PG. Chem Soc Rev, 2018, 47: 7477–7494
Talbot FJT, Dherbassy Q, Manna S, Shi C, Zhang S, Howell GP, Perry GJP, Procter DJ. Angew Chem Int Ed, 2020, 59: 20278–20289
Perry GJP, Jia T, Procter DJ. ACS Catal, 2020, 10: 1485–1499
Pulis AP, Yeung K, Procter DJ. Chem Sci, 2017, 8: 5240–5247
Neeve EC, Geier SJ, Mkhalid IAI, Westcott SA, Marder TB. Chem Rev, 2016, 116: 9091–9161
Collins BSL, Wilson CM, Myers EL, Aggarwal VK. Angew Chem Int Ed, 2017, 56: 11700–11733
Jia T, Cao P, Wang B, Lou Y, Yin X, Wang M, Liao J. J Am Chem Soc, 2015, 137: 13760–13763
Yang K, Song Q. Org Lett, 2016, 18: 5460–5463
Logan KM, Brown MK. Angew Chem Int Ed, 2017, 56: 851–855
Liu Z, Chen J, Lu HX, Li X, Gao Y, Coombs JR, Goldfogel MJ, Engle KM. Angew Chem Int Ed, 2019, 58: 17068–17073
Liao Y, Yin X, Wang X, Yu W, Fang D, Hu L, Wang M, Liao J. Angew Chem Int Ed, 2020, 59: 1176–1180
Whyte A, Torelli A, Mirabi B, Zhang A, Lautens M. ACS Catal, 2020, 10: 11578–11622
Wu FP, Holz J, Yuan Y, Wu XF. CCS Chem, 2020, 2: 2643–2654
Wu FP, Yuan Y, Schünemann C, Kamer PCJ, Wu XF. Angew Chem Int Ed, 2020, 59: 10451–10455
He T, Li B, Liu LC, Wang J, Ma WP, Li GY, Zhang QW, He W. Chem Eur J, 2019, 25: 966–970
Ghosh S, Lai D, Hajra A. Org Biomol Chem, 2020, 18: 7948–7976
Yang Y, Daniliuc CG, Studer A. Angew Chem Int Ed, 2021, 60: 2145–2148
Tani Y, Fujihara T, Terao J, Tsuji Y. J Am Chem Soc, 2014, 136: 17706–17709
Rae J, Yeung K, McDouall JJW, Procter DJ. Angew Chem Int Ed, 2016, 55: 1102–1107
Zhou Y, You W, Smith KB, Brown MK. Angew Chem Int Ed, 2014, 53: 3475–3479
Lesieur M, Bidal YD, Lazreg F, Nahra F, Cazin CSJ. ChemCatChem, 2015, 7: 2108–2112
Semba K, Yoshizawa M, Ohtagaki Y, Nakao Y. BCSJ, 2017, 90: 1340–1343
Cheng LJ, Mankad NP. Angew Chem Int Ed, 2018, 57: 10328–10332
Yuan Y, Wu FP, Xu JX, Wu XF. Angew Chem Int Ed, 2020, 59: 17055–17061
Takaya J, Iwasawa N. ACS Catal, 2012, 2: 1993–2006
Miralles N, Maza RJ, Fernández E. Adv Synth Catal, 2018, 360: 1306–1327
Deposition Numbers 2064555 (for 3a), 2042154 (for 3n), and 2049314 (for 6b) contain the supplementary crystallographic data for this paper. These data are provided free of charge by the joint Cambridge Crystallographic Data Centre and Fachinformationszentrum Karlsruhe Access Structures service http://www.ccdc.cam.ac.uk/structures
Laitar DS, Müller P, Sadighi JP. J Am Chem Soc, 2005, 127: 17196–17197
Krause N, Hashmi ASK. Modern Allene Chemistry. Weinheim: Wiley-VCH, 2004
Acknowledgements
This work was supported by the Chinese Scholarship Council (CSC). We also thank the analytical team of LIKAT for their very kind support.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest The authors declare no conflict of interest.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Yuan, Y., Wu, FP., Spannenberg, A. et al. Cu/Pd-catalyzed borocarbonylative trifunctionalization of alkynes and allenes: synthesis of β-geminal-diboryl ketones. Sci. China Chem. 64, 2142–2153 (2021). https://doi.org/10.1007/s11426-021-1054-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11426-021-1054-4