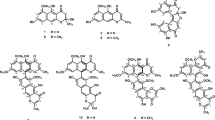
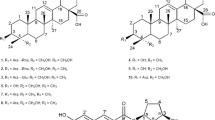
Abstract
Amyloid β (Aβ) is thought to be involved in the pathogenesis of Alzheimer’s disease (AD). Aβ aggregation in the brain is considered the cause of AD. Therefore, inhibiting Aβ aggregation and degrading existing Aβ aggregates is a promising approach for the treatment and prevention of the disease. In searching for inhibitors of Aβ42 aggregation, we found that meroterpenoids isolated from Sargassum macrocarpum possess potent inhibitory activities. Therefore, we searched for active compounds from this brown alga and isolated 16 meroterpenoids, which contain three new compounds. The structures of these new compounds were elucidated using two-dimensional nuclear magnetic resonance techniques. Thioflavin-T assay and transmission electron microscopy were used to reveal the inhibitory activity of these compounds against Aβ42 aggregation. All the isolated meroterpenoids were found to be active, and compounds with a hydroquinone structure tended to have stronger activity than those with a quinone structure.
Graphical abstract





Similar content being viewed by others
References
Yaari R, Corey-Bloom J (2007) Alzheimer’s disease. Semin Neurol 27:32–41
Hardy JA, Higgins GA (1992) Alzheimer’s disease: the amyloid cascade hypothesis. Science 256:184–185
Watson D, Castaño E, Kokjohn TA, Kuo YM, Lyubchenko Y, Pinsky D, Connolly ES Jr, Esh C, Luehrs DC, Stine WB, Rowse LM, Emmerling MR, Roher AE (2005) Physicochemical characteristics of soluble oligomeric Aβ and their pathologic role in Alzheimer’s disease. Neurol Res 27:869–881
Miyamae Y, Kurisu M, Murakami K, Han J, Isoda H, Irie K, Shigemori H (2012) Protective effects of caffeoylquinic acids on the aggregation and neurotoxicity of the 42-residue amyloid β-protein. Bioorg Med Chem 20:5844–5849
Kurisu M, Miyamae Y, Murakami K, Han J, Isoda H, Irie K, Shigemori H (2013) Inhibition of amyloid β aggregation by acteoside, a phenylethanoid glycoside. Biosci Biotechnol Biochem 77:1329–1332
Tsunoda T, Takase M, Shigemori H (2018) Structure-activity relationship of clovamide and its related compounds for the inhibition of amyloid β aggregation. Bioorg Med Chem 26:3202–3209
Nomoto D, Tsunoda T, Shigemori H (2021) Effects of clovamide and its related compounds on the aggregations of amyloid polypeptides. J Nat Med 75:299–307
Tanaka T, Betkekar VV, Ohmori K, Suzuki K, Shigemori H (2021) Evaluation of amyloid polypeptide aggregation inhibition and disaggregation activity of A-type procyanidins. Pharmaceuticals 14:1118
Kobori A, Hosaka K, Shigemori H (2022) Inhibition of amyloid β aggregation by p-terphenyl derivatives isolated from Boletopsis leucomelas. Heterocycles 104:925–933
Sampei T, Wu Y, Shigemori H (2022) Amyloid polypeptide disaggregation activity of passion-fruit-seed-derived polyphenol compounds. Nat Prod Commun 17:1–9
Kamei Y, Sueyoshi M, Hayashi K, Terada R, Nozaki H (2009) The novel anti-Propiobacterium acnes compound, Sargafuran, found in the marine brown alga Sargassum macrocarpum. J Antibiot 62:259–263
Choi YK, Kim J, Lee KM, Choi YJ, Ye BR, Kim MS, Ko SG, Lee SH, Kang DH, Heo SJ (2017) Tuberatolide B suppresses cancer progression by promoting ROS–mediated inhibition of STAT3 signaling. Mar Drugs 15:55
Kwon J, Lee K, Hwang H, Kim SH, Park SE, Durai P, Park K, Kim HS, Jang DS, Choi JS, Kwon HC (2022) New monocyclic terpenoid lactones from a brown algae Sargassum macrocarpum as monoamine oxidase inhibitors. Plants 11:1998
Niwa H, Kurimoto SI, Kubota T, Sekiguchi M (2021) Macrocarquinoids A-C, new meroterpenoids from Sargassum macrocarpum. J Nat Med 75:194–200
Shinoda S, Kurimoto SI, Kubota T, Sekiguchi M (2022) Macrocarquinoid D, new meroterpenoid from brown alga, Sargassum macrocarpum. Heterocycles 104:1141–1147
Seo Y, Park KE, Kim YA, Lee HJ, Yoo JS, Ahn JW, Lee BJ (2006) Isolation of tetraprenyltoluquinols from the brown alga Sargassum thunbergii. Chem Pharm Bull (Tokyo) 54:1730–1733
Sato A, Shindo T, Hasegawa K, Ushiyama S (1990) Prenylquinone and hydroquinone derivative. Kokai Tokkyo Koho Japan H02-290826
Brkljača R, Urban S (2014) Chemical profiling (HPLC-NMR & HPLC-MS), isolation, and identification of bioactive meroditerpenoids from the southern Australian marine brown alga Sargassum paradoxum. Mar Drugs 13:102–127
Seo Y, Lee HL, Park KE, Kim YA, Ahn JW, Yoo JS, Lee BJ (2004) Peroxynitrite-scavenging constituents from the brown alga Sargassum thunbergii. Biotechnol Bioprocess Eng 9:212–216
Kusumi T, Shibata Y, Ishitsuka M, Kinoshita T, Kakisawa H (1979) Structure of new plastoquinones from the brown alga Sargassum serratifolium. Chem Lett 8:277–278
Silva DH, Pereira FC, Zanoni MV, Yoshida M (2001) Lipophyllic antioxidants from Iryanthera juruensis fruits. Phytochem 57:437–442
Voutquenne L, Lavaud C, Massiot G, Sevenet T, Hadi HA (1999) Cytotoxic polyisoprenes and glycosides of long-chain fatty alcohols from Dimocarpus fumatus. Phytochem 50:63–69
Reddy P, Urban S (2009) Meroditerpenoids from southern Australian marine brown alga Sargassum fallax. Phytochem 70:250–255
Naiki H, Gejyo F (1999) Kinetic analysis of amyloid fibril formation. Methods Enzymol 309:305–318
Acknowledgements
The authors thank Mr. Takahiko Ikemori, Ishikawa Prefecture Fisheries Research Center, for his assistance with collection and classification of the alga.
Funding
This work was partially supported by Grants-in-Aids for Scientific Research from the Japan Society for the Promotion of Science (JSPS) KAKENHI under grant number 22K06664.
Author information
Authors and Affiliations
Contributions
MS and HS designed experiments and wrote the manuscript. SS isolated compounds. SS and SK measured physicochemical data including NMR and MS and elucidated the structures. YT conducted Th-T assay and TEM measurement. All authors have been read and approved the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest related to this report or the study it describes.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Shinoda, S., Tozawa, Y., Kurimoto, Si. et al. Three new meroterpenoids from Sargassum macrocarpum and their inhibitory activity against amyloid β aggregation. J Nat Med 77, 508–515 (2023). https://doi.org/10.1007/s11418-023-01693-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11418-023-01693-y