Abstract
Purpose
Despite the benefits in human health from pharmaceuticals, their production has simultaneously given rise to severe environmental pollution. Using ciprofloxacin hydrochloride production as an example, the objectives of this study were to determine the key materials and stages in the life cycle of pharmaceutical production and to develop methods to prevent high materials/energy consumption and pollution.
Methods
The environmental impacts were evaluated by the Eco-indicator 99 (EI 99) method built into the Ecoinvent database of the Simapro software. The functional unit was set to the annual production of 280 million ciprofloxacin hydrochloridet tablets. The data in this study regarding the process parameters, raw materials energy consumption, and the emissions of pollutants were provided by a pharmaceutical enterprise located in the Zhejiang province, China. The complete production process was classified into three stages, active pharmaceutical ingredient (API) synthesis, galenic formulation, and packaging following the product category rules (PCR) for pharmaceuticals.
Results and discussion
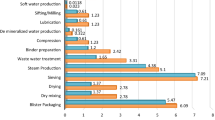
API synthesis had the largest environmental contribution, followed by galenic formulation, and packaging, which accounted for 42.9%, 41.9%, and 15.2%, respectively. The most important influencing factors of the first two stages were polyols which represent solvents or reaction media used and electricity (coal-fired generation). The damage categories of both factors were ranked from high to low as follows: human health, resources, and ecosystems. The ecological index points of human health in the API synthesis decreased by 8 k point (kPt) after solvent replacement, by 4 kPt after coal-based electricity generation was replaced with natural gas, and by 12 kPt after simultaneous optimization.
Conclusions
The proposed improvements for cleaner production all proved the feasibility of environmental sustainability in pharmaceutical production. Compared with energy optimization, the solvent replacement was a more effective and convenient method to reduce the environmental impact of ciprofloxacin hydrochloride production.




Similar content being viewed by others
References
Alvarez-Hess PS, Little SM, Moate PJ et al (2019) A partial life cycle assessment of the greenhouse gas mitigation potential of feeding 3-nitrooxypropanol and nitrate to cattle. Agr Syst 169:14–23
Alviz PLA, Patricia L, Alvarez AJ (2017) Comparative life cycle assessment of the use of an ionic liquid ([Bmim]Br) versus a volatile organic solvent in the production of acetylsalicylic acid. J Clean Prod 168:1614–1624
Bielen A, Simatovic A, Kosic-Vuksic J et al (2017) Negative environmental impacts of antibiotic-contaminated effluents from pharmaceutical industries. Water Res 126:79–87
Bourtsalas AC, Zhang J, Castaldi MJ et al (2018) Use of non-recycled plastics and paper as alternative fuel in cement production. J Clean Prod 181:8–16
Crenna E, Sozzo S, Sala S (2018) Natural biotic resources in LCA: towards an impact assessment model for sustainable supply chain management. J Clean Prod 172:3669–3684
Cymes BA, Almquist CB, Krekeler MPS et al (2020) Europium-doped cryptomelane: multi-pathway synthesis, characterization, and evaluation for the gas phase catalytic oxidation of ethanol. Appl Catal A-Gen 589:117310
De Soete W, Debaveye S, De Meester S et al (2014) Environmental sustainability assessments of pharmaceuticals: an emerging need for simplification in life cycle assessments. Environ Sci Technol 48(20):12247–12255
Dones R, Heck T, Emmenegger MF et al (2018) Life cycle inventories for the nuclear and natural gas energy systems, and examples of uncertainty analysis. Int J Life Cycle Assess 10(1):10–23
Emara Y, Lehmann A, Siegert MW et al (2019) Modeling pharmaceutical emissions and their toxicity-related effects in life cycle assessment (LCA): a review. Integr Environ Assess 15:6–18
Emara Y, Siegert MW, Lehmann A, et al (2018) Life cycle management in the pharmaceutical industry using an applicable and robust LCA-based environmental sustainability assessment approach. Designing Sustainable Technologies, Products and Policies 79–88.
Feng YX, Gao YC, Tian GD et al (2019) Flexible process planning and end-of-life decision-making for product recovery optimization based on hybrid disassembly. IEEE T Autom Sci Eng 16(1):311–326
Gamiz AGR, De Soete W, Heirman B et al (2019) Environmental sustainability assessment of the manufacturing process of a biological active pharmaceutical ingredient. J Chem Technol Biotechnol 94(6):1937–1944
Guo XP, Ren DF, Guo XD (2018) A system dynamics model of China’s electric power structure adjustment with constraints of PM10 emission reduction. Environ Sc Pollut R 25(18):17540–17552
Guo Z, Ma LW, Liu P et al (2017) A long-term multi-region load-dispatch model based on grid structures for the optimal planning of China’s power sector. Comput Chem Eng 102:52–63
Hischier R, Baudin I (2018) LCA study of a plasma television device. Int J Life Cycle Assess 15(5):428–438
ISO14040 (2006) Environmental management - Life cycle assessment - Principles and framework. International Organization for Standardization, Geneva.
Environmental Management - Life Cycle Assessment - Requirements and Guidelines. International Organization for Standardization, Geneva.
Jiménez-González C, Overcash MR (2014) The evolution of life cycle assessment in pharmaceutical and chemical applications—a perspective. Green Chem 16(7):3392–3400
Kuksa L, Barkane L, Hittel N et al (2017) Final treatment outcomes of multidrug- and extensively drug-resistant tuberculosis patients in Latvia receiving delamanid-containing regimens. Eur Respir J 50(5):1701105
Lee CK, Khoo HH, Reginald BHT (2015) LCA-based environmental performance comparison of batch and continuous processing: a case of 4-D-Erythronolactone synthesis. Org Process Res Dev 20:1937–1948
Lesage P, Mutel C, Schenker U et al (2018) Uncertainty analysis in LCA using precalculated aggregated datasets. Int J Life Cycle Assess 23(11):2248–2265
Lin HK, Dai CH, Jamison TF et al (2017) A rapid total synthesis of ciprofloxacin hydrochloride in continuous flow. Angew Chem int Edit 56(30):8870–8873
McElroy CR, Constantinou A, Jones LC et al (2015) Towards a holistic approach to metrics for the 21st century pharmaceutical industry. Green Chem 17(5):3111–3121
Rosenthal K, Lutz S (2018) Recent developments and challenges of biocatalytic processes in the pharmaceutical industry. Curr Opin Gree Sust Chem 11:58–64
Siegert MW, Lehmann A, Emara Y et al (2018) Harmonized rules for future LCAs on pharmaceutical products and processes. Int J Life Cycle Ass 24(6):1040–1057
Siegert MW, Lehmann A, Emara Y et al (2020) Correction to: Addressing the use and end-of-life phase of pharmaceutical products in life cycle assessment. Int J Life Cycle Ass 25(8):1633–1634
Thiebault T, Chassiot L, Fougeere L et al (2017) Record of pharmaceutical products in river sediments: a powerful tool to assess the environmental impact of urban management. Anthropocene 18:47–56
Wang H, Al-Saadi I, Lu P et al (2020) Quantifying greenhouse asphalt pavement preservation at construction and use stages using life-cycle assessment. Int J Sustain Transp 14(1):25–34
Yang XN, Hu MM, Wu JB et al (2018) Building information modeling enabled life cycle assessment, a case study on carbon footprint accounting for a residential building in China. J Clean Prod 183:729–743
Yang Y (2017) Does hybrid LCA with a complete system boundary yield adequate results for product promotion? Int J Life Cycle Assess 22(3):456–460
Funding
This work was financially supported by the National Key Research and Development Program (2018YFC0214103) and the Subsidized Project for Postgraduates' Innovative Fund in Scientific Research of Huaqiao University.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Matthias Finkbeiner.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Yang, K., Lv, B., Shen, H. et al. Life cycle assessment of pharmaceuticals: the ciprofloxacin hydrochloride case. Int J Life Cycle Assess 26, 64–75 (2021). https://doi.org/10.1007/s11367-020-01841-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11367-020-01841-6