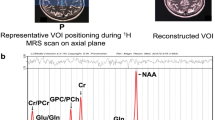
Abstract
Aging is a major risk for cognitive decline and transition to dementia. One well-known age-related change involves decreased brain efficiency and energy production, mediated in part by changes in mitochondrial function. Damaged or dysfunctional mitochondria have been implicated in the pathogenesis of age-related neurodegenerative conditions like Alzheimer’s disease (AD). The aim of the current study was to investigate mitochondrial function over frontal and temporal regions in a sample of 70 cognitively normal older adults with subjective memory complaints and a first-degree family history of AD. We hypothesized cerebral mitochondrial function and energy metabolism would be greater in temporal as compared to frontal regions based on the high energy consumption in the temporal lobes (i.e., hippocampus). To test this hypothesis, we used phosphorous (31P) magnetic resonance spectroscopy (MRS) which is a non-invasive and powerful method for investigating in vivo mitochondrial function via high energy phosphates and phospholipid metabolism ratios. We used a single voxel method (left temporal and bilateral prefrontal) to achieve optimal sensitivity. Results of separate repeated measures analyses of variance showed 31P MRS ratios of static energy, energy reserve, energy consumption, energy demand, and phospholipid membrane metabolism were greater in the left temporal than bilateral prefrontal voxels. Our findings that all 31P MRS ratios were greater in temporal than bifrontal regions support our hypothesis. Future studies are needed to determine whether findings are related to cognition in older adults.



Similar content being viewed by others
Data Availability
Data are managed under the data sharing agreement established with NIA and the parent R01 clinical trial Data Safety and Monitoring Board (DSMB) in the context of a phase II clinical trial (REVITALIZE, R01AG064587). All trial data will be made publicly available 2 years after completion of the parent clinical trial, per NIA and DSMB agreement.
References
Bratic I, Trifunovic A. Mitochondrial energy metabolism and ageing. Biochim Biophys Acta (BBA)-Bioenergetics. 2010;1797(6–7):961–7.
Bertoni-Freddari C, Fattoretti P, Giorgetti B, Solazzi M, Balietti M, Meier-Ruge W. Role of mitochondrial deterioration in physiological and pathological brain aging. Gerontology. 2004;50(3):187–92.
Kadenbach B, Ramzan R, Vogt S. Degenerative diseases, oxidative stress and cytochrome c oxidase function. Trends Mol Med. 2009;15(4):139–47.
Dienel GA. Brain glucose metabolism: integration of energetics with function. Physiol Rev. 2019;99(1):949–1045.
Beal MF. Mitochondria take center stage in aging and neurodegeneration. Ann Neurol. 2005;58(4):495–505.
Sharma C, Kim S, Nam Y, Jung UJ, Kim SR. Mitochondrial dysfunction as a driver of cognitive impairment in Alzheimer’s disease. Int J Mol Sci. 2021;22(9):4850.
Mani S, Sevanan M, Krishnamoorthy A, Sekar S. A systematic review of molecular approaches that link mitochondrial dysfunction and neuroinflammation in Parkinson’s disease. Neurol Sci. 2021;42:4459–69.
Anoar S, Woodling NS, Niccoli T. Mitochondria dysfunction in frontotemporal dementia/amyotrophic lateral sclerosis: lessons from Drosophila models. Front Neurosci. 2021;15:786076.
Buchli R, Martin E, Boesiger P, Rumpel H. Developmental changes of phosphorus metabolite concentrations in the human brain: a 31P magnetic resonance spectroscopy study in vivo. Pediatr Res. 1994;35(4):431–5.
Ross AJ, Sachdev PS. Magnetic resonance spectroscopy in cognitive research. Brain Res Rev. 2004;44(2–3):83–102.
Chaumeil MM, Valette J, Guillermier M, Brouillet E, Boumezbeur F, Herard AS, Bloch G, Hantraye P, Lebon V. Multimodal neuroimaging provides a highly consistent picture of energy metabolism, validating 31P MRS for measuring brain ATP synthesis. Proc Natl Acad Sci. 2009;106(10):3988–93.
Cady EB. In vivo cerebral 31P magnetic resonance spectroscopy. Neural Metab Vivo. 2012;149–79.
Purdon AD, Rapoport SI. Energy requirements for two aspects of phospholipid metabolism in mammalian brain. Biochem J. 1998;335(2):313–8.
Hnilicova P, Richterova R, Zelenak K, Kolarovszki B, Majercikova Z, Hatok J. Noninvasive study of brain tumours metabolism using phosphorus-31 magnetic resonance spectroscopy. Bratislava Medical Journal/Bratislavske Lekarske Listy. 2020;121(7).
Walchhofer LM, Steiger R, Rietzler A, Kerschbaumer J, Freyschlag CF, Stockhammer G, Gizewski ER, Grams AE. Phosphorous magnetic resonance spectroscopy to detect regional differences of energy and membrane metabolism in naïve glioblastoma multiforme. Cancers. 2021;13(11):2598.
Pettegrew JW, Panchalingam K, Moossy J, Martinez J, Rao G, Boller F. Correlation of phosphorus-31 magnetic resonance spectroscopy and morphologic findings in Alzheimer’s disease. Arch Neurol. 1988;45(10):1093–6.
Brown GG, Levine SR, Gorell JM, Pettegrew JW, Gdowski JW, Bueri JA, Helpern JA, Welch KM. In vivo 31P NMR profiles of Alzheimer’s disease and multiple subcortical infarct dementia. Neurology. 1989;39(11):1423-.
Pettegrew JW, Panchalingam K, Klunk WE, McClure RJ, Muenz LR. Alterations of cerebral metabolism in probable Alzheimer’s disease: a preliminary study. Neurobiol Aging. 1994;15(1):117–32.
González RG, Guimaraes AR, Moore GJ, Crawley A, Cupples LA, Growdon JH. Quantitative in vivo 31P magnetic resonance spectroscopy of Alzheimer disease. Alzheimer Dis Assoc Disord. 1996;10(1):46–52.
Forlenza OV, Wacker P, Nunes PV, Yacubian J, Castro CC, Otaduy MC, Gattaz WF. Reduced phospholipid breakdown in Alzheimer’s brains: a 31P spectroscopy study. Psychopharmacology. 2005;180:359–65.
Rijpma A, van der Graaf M, Meulenbroek O, Rikkert MG, Heerschap A. Altered brain high-energy phosphate metabolism in mild Alzheimer’s disease: a 3-dimensional 31P MR spectroscopic imaging study. NeuroImage: Clinical. 2018;18:254–61.
Das N, Ren J, Spence JS, Rackley A, Chapman SB. Relationship of parieto-occipital brain energy phosphate metabolism and cognition using 31P MRS at 7-Tesla in amnestic mild cognitive impairment. Front Aging Neurosci. 2020;12:222.
Das N, Ren J, Spence J, Chapman SB. phosphate brain energy metabolism and cognition in Alzheimer’s disease: a spectroscopy study using whole-brain volume-coil 31Phosphorus magnetic resonance spectroscopy at 7Tesla. Front Neurosci. 2021;15:641739.
Jett S, Boneu C, Zarate C, Carlton C, Kodancha V, Nerattini M, Battista M, Pahlajani S, Williams S, Dyke JP, Mosconi L. Systematic review of 31P-magnetic resonance spectroscopy studies of brain high energy phosphates and membrane phospholipids in aging and Alzheimer’s disease. Front Aging Neurosci. 2023;15:1183228.
Forester BP, Berlow YA, Harper DG, Jensen JE, Lange N, Froimowitz MP, Ravichandran C, Iosifescu DV, Lukas SE, Renshaw PF, Cohen BM. Age-related changes in brain energetics and phospholipid metabolism. NMR Biomed. 2010;23(3):242–50.
Schmitz B, Wang X, Barker PB, Pilatus U, Bronzlik P, Dadak M, Kahl KG, Lanfermann H, Ding XQ. Effects of aging on the human brain: a proton and phosphorus MR spectroscopy study at 3T. J Neuroimaging. 2018;28(4):416–21.
Cuenoud B, Ipek Ö, Shevlyakova M, Beaumont M, Cunnane SC, Gruetter R, Xin L. Brain NAD is associated with ATP energy production and membrane phospholipid turnover in humans. Front Aging Neurosci. 2020;12:609517.
Rietzler A, Steiger R, Mangesius S, Walchhofer LM, Gothe RM, Schocke M, Gizewski ER, Grams AE. Energy metabolism measured by 31P magnetic resonance spectroscopy in the healthy human brain. J Neuroradiol. 2022;49(5):370–9.
Jett S, Dyke JP, Andy C, Schelbaum E, Jang G, Boneu Yepez C, Pahlajani S, Diaz I, Diaz Brinton R, Mosconi L. Sex and menopause impact 31P-magnetic resonance spectroscopy brain mitochondrial function in association with 11C-PiB PET amyloid-beta load. Sci Rep. 2022;12(1):22087.
Hu Y, Wilson GS. A temporary local energy pool coupled to neuronal activity: fluctuations of extracellular lactate levels in rat brain monitored with rapid-response enzyme-based sensor. J Neurochem. 1997;69(4):1484–90.
Lepage M, Habib R, Tulving E. Hippocampal PET activations of memory encoding and retrieval: the HIPER model. Hippocampus. 1998;8(4):313–22.
Schacter DL, Wagner AD. Medial temporal lobe activations in fMRI and PET studies of episodic encoding and retrieval. Hippocampus. 1999;9(1):7–24.
Choi IY, Gruetter R (editors). Neural metabolism in vivo. 4th ed. Springer Science & Business Media; 2012.
Risacher SL, Kim S, Nho K, Foroud T, Shen L, Petersen RC, Jack CR Jr, Beckett LA, Aisen PS, Koeppe RA, Jagust WJ. APOE effect on Alzheimer’s disease biomarkers in older adults with significant memory concern. Alzheimer’s Dementia. 2015;11(12):1417–29.
Morris JC, Weintraub S, Chui HC, Cummings J, DeCarli C, Ferris S, Foster NL, Galasko D, Graff-Radford N, Peskind ER, Beekly D. The Uniform Data Set (UDS): clinical and cognitive variables and descriptive data from Alzheimer Disease Centers. Alzheimer Dis Assoc Disord. 2006;20(4):210–6.
Vanhamme L, van den Boogaart A, Van Huffel S. Improved method for accurate and efficient quantification of MRS data with use of prior knowledge. J Magn Reson. 1997;129(1):35–43.
Stefan DD, Di Cesare F, Andrasescu A, Popa E, Lazariev A, Vescovo E, Strbak O, Williams S, Starcuk Z, Cabanas M, Van Ormondt D. Quantitation of magnetic resonance spectroscopy signals: the jMRUI software package. Meas Sci Technol. 2009;20(10):104035.
Santos-Díaz A, Noseworthy MD. Phosphorus magnetic resonance spectroscopy and imaging (31P-MRS/MRSI) as a window to brain and muscle metabolism: A review of the methods. Biomed Signal Process Control. 2020;60:101967.
Zhang Y, Brady M, Smith S. Segmentation of brain MR images through a hidden Markov random field model and the expectation-maximization algorithm. IEEE Trans Med Imaging. 2001;20(1):45–57.
Smith SM, Jenkinson M, Woolrich MW, Beckmann CF, Behrens TE, Johansen-Berg H, Bannister PR, De Luca M, Drobnjak I, Flitney DE, Niazy RK. Advances in functional and structural MR image analysis and implementation as FSL. Neuroimage. 2004;23:S208–19.
Ashburner J, Friston KJ. Unified segmentation. Neuroimage. 2005;26(3):839–51.
Quadrelli S, Mountford C, Ramadan S. Hitchhiker’s guide to voxel segmentation for partial volume correction of in vivo magnetic resonance spectroscopy. Magn Reson Insights. 2016;9:MRI-S32903.
Sassani M, Alix JJ, McDermott CJ, Baster K, Hoggard N, Wild JM, Mortiboys HJ, Shaw PJ, Wilkinson ID, Jenkins TM. Magnetic resonance spectroscopy reveals mitochondrial dysfunction in amyotrophic lateral sclerosis. Brain. 2020;143(12):3603–18.
McNay EC, Fries TM, Gold PE. Decreases in rat extracellular hippocampal glucose concentration associated with cognitive demand during a spatial task. Proc Natl Acad Sci. 2000;97(6):2881–5.
Calabrese V, Scapagnini G, Ravagna A, Fariello RG, Giuffrida Stella AM, Abraham NG. Regional distribution of heme oxygenase, HSP70, and glutathione in brain: relevance for endogenous oxidant/antioxidant balance and stress tolerance. J Neurosci Res. 2002;68(1):65–75.
Moeller JR, Ishikawa T, Dhawan V, Spetsieris P, Mandel F, Alexander GE, Grady C, Pietrini P, Eidelberg D. The metabolic topography of normal aging. J Cereb Blood Flow Metab. 1996;16(3):385–98.
Petit-Taboue MC, Landeau B, Desson JF, Desgranges B, Baron JC. Effects of healthy aging on the regional cerebral metabolic rate of glucose assessed with statistical parametric mapping. Neuroimage. 1998;7(3):176–84.
Bartzokis G, Beckson M, Lu PH, Nuechterlein KH, Edwards N, Mintz J. Age-related changes in frontal and temporal lobe volumes in men: a magnetic resonance imaging study. Arch Gen Psychiatry. 2001;58(5):461–5.
Jett S, Dyke JP, Boneu Yepez C, Zarate C, Carlton C, Schelbaum E, Jang G, Pahlajani S, Williams S, Diaz Brinton R, Mosconi L. Effects of sex and APOE ε4 genotype on brain mitochondrial high-energy phosphates in midlife individuals at risk for Alzheimer’s disease: a 31Phosphorus MR spectroscopy study. PLoS ONE. 2023;18(2):e0281302.
Meyerspeer M, Boesch C, Cameron D, Dezortová M, Forbes SC, Heerschap A, Jeneson JA, Kan HE, Kent J, Layec G, Prompers JJ. 31P magnetic resonance spectroscopy in skeletal muscle: experts’ consensus recommendations. NMR Biomed. 2021;34(5):e4246.
Ren J, Shang T, Sherry AD, Malloy CR. Unveiling a hidden 31P signal coresonating with extracellular inorganic phosphate by outer-volume-suppression and localized 31P MRS in the human brain at 7T. Magn Reson Med. 2018;80(4):1289–97.
Acknowledgements
We would like to thank all the research participants at the McKnight Brain Institutes of the Universities of Florida (UF) and Arizona (UA), who generously volunteered their time and effort to help make this manuscript possible. We want to also thank to all research team members at the UF-Cognitive Neuroscience Lab and UA-Brain Imaging, Behavior and Aging Lab for their contributions to this project.
Funding
This study was supported by the National Institutes of Health (R01AG64587, F31AG071264, T32NS082168), the University of Florida and Arizona Evelyn F. McKnight Brain Institute, and University of Florida Norman Fixel Institute for Neurological Diseases.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
About this article
Cite this article
Lopez, F.V., O’Shea, A., Huo, Z. et al. Frontal–temporal regional differences in brain energy metabolism and mitochondrial function using 31P MRS in older adults. GeroScience 46, 3185–3195 (2024). https://doi.org/10.1007/s11357-023-01046-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11357-023-01046-3