Abstract
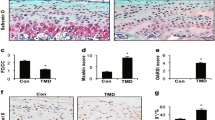
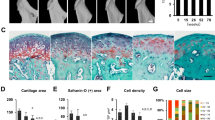
Osteoarthritis (OA) of the knee is closely associated with aging; however, little is known about the age-related degeneration in the mandibular condylar cartilage (MCC) of the TMJ. Our objective was to examine whether a correlation exists between aging and degeneration of the MCC of the TMJ. Thirty-two male C57BL/6J wild-type mice were aged to 2, 12, 18, and 25 months old. The mice were euthanized by CO2 inhalation and were dissected and examined by micro-CT and histology. Sagittal sections of the condyles were stained for tartrate-resistant alkaline phosphatase, alkaline phosphatase, safranin O, picrosirius red, and toluidine blue. In addition, immunostaining for BMP2, BMP4, BMP7, PRG4, and MMP13 was performed. Bone volume fraction and tissue density significantly increased with the age of the animals. There was a significant increase in the Osteoarthritis Research Society International histopathological score and mineralization of the noncalcified cartilage in the aged animals. There was a decrease in cartilage thickness, proteoglycan distribution, and cellularity in the aged animals. Additionally, we noted increased picrosirius red staining with the increase in the age of the animals. Our protein expression showed increased BMP2, BMP4, BMP7, and MMP13, whereas there was a decrease in PRG4 expression in the aged animals. As the animal ages, there is decreased proteoglycan secretion, decreased cellularity, decreased cartilage thickness, increased fibrillation, and increased proteolytic activity. A better understanding of the basic mechanisms underlying the degeneration of the MCC in the older animals could provide novel ways to slow the development of OA.




Similar content being viewed by others
References
Aigner T, Kurz B, Fukui N, Sandell L (2002) Roles of chondrocytes in the pathogenesis of osteoarthritis. Curr Opin Rheumatol 14:578–584
Aigner T, Soder S, Gebhard PM, McAlinden A, Haag J (2007) Mechanisms of disease: role of chondrocytes in the pathogenesis of osteoarthritis--structure, chaos and senescence. Nat Clin Pract Rheumatol 3:391–399. https://doi.org/10.1038/ncprheum0534
Bao JP, Chen WP, Wu LD (2011) Lubricin: a novel potential biotherapeutic approaches for the treatment of osteoarthritis. Mol Biol Rep 38:2879–2885. https://doi.org/10.1007/s11033-010-9949-9
Bechtold TE et al (2016a) Osteophyte formation and matrix mineralization in a TMJ osteoarthritis mouse model are associated with ectopic hedgehog signaling. Matrix Biol 52-54:339–354. https://doi.org/10.1016/j.matbio.2016.03.001
Bechtold TE et al (2016b) Excess BMP signaling in heterotopic cartilage forming in Prg4-null TMJ discs. J Dent Res 95:292–301. https://doi.org/10.1177/0022034515613508
DiLoreto R, Murphy CT (2015) The cell biology of aging. Mol Biol Cell 26:4524–4531. https://doi.org/10.1091/mbc.E14-06-1084
Flannery CR et al (2009) Prevention of cartilage degeneration in a rat model of osteoarthritis by intraarticular treatment with recombinant lubricin. Arthritis Rheum 60:840–847. https://doi.org/10.1002/art.24304
Gho WG, Choi Y, Park K-H, Huh J-K (2018) Expression of collagenases (matrix metalloproteinase-1, 8, 13) and tissue inhibitor of metalloproteinase-1 of retrodiscal tissue in temporomandibular joint disorder patients. J Korean Assoc Oral Maxillofac Surg 44:120–127. https://doi.org/10.5125/jkaoms.2018.44.3.120
Glasson SS, Chambers MG, Van Den Berg WB, Little CB (2010) The OARSI histopathology initiative - recommendations for histological assessments of osteoarthritis in the mouse osteoarthritis and cartilage / OARS. Osteoarthr Res Soc 18(Suppl 3):S17–S23. https://doi.org/10.1016/j.joca.2010.05.025
Hill A, Duran J, Purcell P (2014) Lubricin protects the temporomandibular joint surfaces from degeneration. PloS One 9:e106497. https://doi.org/10.1371/journal.pone.0106497
Koyama E et al (2014) Lubricin is required for the structural integrity and post-natal maintenance of TMJ. J Dent Res 93:663–670. https://doi.org/10.1177/0022034514535807
Kuroda S, Tanimoto K, Izawa T, Fujihara S, Koolstra JH, Tanaka E (2009) Biomechanical and biochemical characteristics of the mandibular condylar cartilage osteoarthritis and cartilage / OARS. Osteoarthr Res Soc 17:1408–1415. https://doi.org/10.1016/j.joca.2009.04.025
Loeser RF (2010) Age-related changes in the musculoskeletal system and the development of osteoarthritis. Clin Geriatr Med 26:371–386. https://doi.org/10.1016/j.cger.2010.03.002
Loeser RF, Gandhi U, Long DL, Yin W, Chubinskaya S (2014) Aging and oxidative stress reduce the response of human articular chondrocytes to insulin-like growth factor 1 and osteogenic protein 1. Arthritis Rheumatol 66:2201–2209. https://doi.org/10.1002/art.38641
Lories RJ, Luyten FP (2011) The bone-cartilage unit in osteoarthritis. Nat Rev Rheumatol 7:43–49. https://doi.org/10.1038/nrrheum.2010.197
Lotz M, Loeser RF (2012) Effects of aging on articular cartilage homeostasis. Bone 51:241–248. https://doi.org/10.1016/j.bone.2012.03.023
Luder HU (2002) Factors affecting degeneration in human temporomandibular joints as assessed histologically. Eur J Oral Sci 110:106–113
Martin JA, Buckwalter JA (2002) Human chondrocyte senescence and osteoarthritis. Biorheology 39:145–152
Mirahmadi F et al (2018) Mechanical stiffness of TMJ condylar cartilage increases after artificial aging by ribose. Arch Oral Biol 87:102–109. https://doi.org/10.1016/j.archoralbio.2017.12.010
Orajarvi M et al (2018) Changes in type I and type II collagen expression in rat mandibular condylar cartilage associated with aging and dietary loading. J Oral Facial Pain Headache 32:258–265. https://doi.org/10.11607/ofph.1581
Rahmati M, Nalesso G, Mobasheri A, Mozafari M (2017) Aging and osteoarthritis: central role of the extracellular matrix. Ageing Res Rev 40:20–30. https://doi.org/10.1016/j.arr.2017.07.004
Roemhildt ML, Beynnon BD, Gardner-Morse M (2012) Mineralization of articular cartilage in the Sprague-Dawley rat: characterization and mechanical analysis osteoarthritis and cartilage / OARS. Osteoarthr Res Soc 20:796–800. https://doi.org/10.1016/j.joca.2012.04.011
Sandell LJ, Aigner T (2001) Articular cartilage and changes in arthritis. An introduction: cell biology of osteoarthritis. Arthritis Res 3:107–113. https://doi.org/10.1186/ar148
Schmitz N, Laverty S, Kraus VB, Aigner T (2010) Basic methods in histopathology of joint tissues osteoarthritis and cartilage / OARS. Osteoarthr Res Soc 18(Suppl 3):S113–S116. https://doi.org/10.1016/j.joca.2010.05.026
Shiomi T, Lemaitre V, D'Armiento J, Okada Y (2010) Matrix metalloproteinases, a disintegrin and metalloproteinases, and a disintegrin and metalloproteinases with thrombospondin motifs in non-neoplastic diseases. Pathol Int 60:477–496. https://doi.org/10.1111/j.1440-1827.2010.02547.x
Teeple E, Elsaid KA, Fleming BC, Jay GD, Aslani K, Crisco JJ, Mechrefe AP (2008) Coefficients of friction, lubricin, and cartilage damage in the anterior cruciate ligament-deficient Guinea pig knee. J Orthop Res 26:231–237. https://doi.org/10.1002/jor.20492
Thielen NGM, van der Kraan PM, van Caam APM (2019) TGFbeta/BMP signaling pathway in cartilage homeostasis cells 8. https://doi.org/10.3390/cells8090969
Wadhwa S, Kapila S (2008) TMJ disorders: future innovations in diagnostics and therapeutics. J Dent Educ 72:930–947
Wang M, Sampson ER, Jin H, Li J, Ke QH, Im HJ, Chen D (2013) MMP13 is a critical target gene during the progression of osteoarthritis. Arthritis Res Ther 15:R5. https://doi.org/10.1186/ar4133
Wu L, Huang X, Li L, Huang H, Xu R, Luyten W (2012) Insights on biology and pathology of HIF-1alpha/-2alpha, TGFbeta/BMP, Wnt/beta-catenin, and NF-kappaB pathways in osteoarthritis. Curr Pharm Des 18:3293–3312. https://doi.org/10.2174/1381612811209023293
Wu Y et al (2019) Effect of sustained joint loading on TMJ disc nutrient environment. J Dent Res 98:888–895. https://doi.org/10.1177/0022034519851044
Yadav S, Yang Y, Dutra EH, Robinson JL, Wadhwa S (2018) Temporomandibular joint disorders in older adults. J Am Geriatr Soc 66:1213–1217. https://doi.org/10.1111/jgs.15354
Zhang W, Robertson WB, Zhao J, Chen W, Xu J (2019) Emerging trend in the pharmacotherapy of osteoarthritis. Front Endocrinol (Lausanne) 10:431. https://doi.org/10.3389/fendo.2019.00431
Acknowledgments
Research reported in this publication was supported by the National Institute of Dental and Craniofacial Research of the National Institute of Health under the award number KO8DE025914 and by the American Association of Orthodontic Foundation and startup funds provided to SY.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
About this article
Cite this article
Chen, PJ., Dutra, E.H., Mehta, S. et al. Age-related changes in the cartilage of the temporomandibular joint. GeroScience 42, 995–1004 (2020). https://doi.org/10.1007/s11357-020-00160-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11357-020-00160-w